缺氧诱导因子1α对肝癌细胞HepG2干细胞特性及表阿霉素敏感性的影响
DOI: 10.3969/j.issn.1001-5256.2021.02.021
利益冲突声明:本研究不存在研究者、伦理委员会、受试者监护人以及与公开研究成果有关的利益冲突,特此声明。
作者贡献声明:赵金金负责课题设计,资料分析,撰写论文;张海光负责实验操作,参与数据分析;崔非非、汪磊参与实验操作;莫清江参与收集数据,修改论文;焦路阳负责拟定写作思路,指导撰写论文并最后定稿。
Effect of hypoxia-inducible factor-1α on stemness and epirubicin sensitivity of HepG2 hepatoma cells
-
摘要:
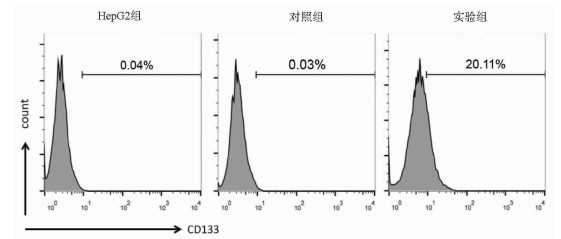
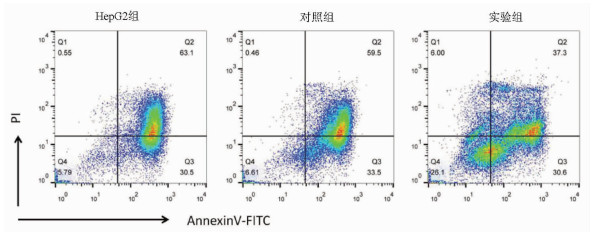
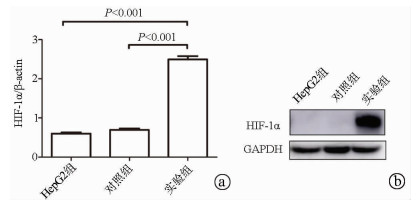
目的 探讨缺氧诱导因子(HIF)1α对肝癌细胞HepG2干细胞特性及表阿霉素敏感性的影响。 方法 以肝癌细胞为研究对象,肝癌细胞HepG2脂质体转染过表达HIF-1α的质粒作为实验组,转染pcDNA3.1空质粒作为对照组,单独HepG2细胞为HepG2组。实时荧光定量PCR检测HIF-1α mRNA表达,Western Blot检测HIF-1α蛋白表达;流式细胞术检测细胞表面CD133表达。不同浓度表阿霉素(0、6.25、12.5、25、50 μmol/L)作用3组细胞24 h,MTT法检测细胞活性,流式细胞术检测表阿霉素(50 μmol/L)处理后细胞凋亡情况。计量资料多组间比较采用单因素方差分析,进一步两两比较采用t检验。 结果 相较于HepG2组及对照组,实验组HIF-1α mRNA的表达水平明显升高,差异具有统计学意义(P值均<0.001);Western Blot结果显示实验组HIF-1α蛋白高表达。HepG2组、对照组和实验组细胞的CD133比例分别为0.040%±0.003%、0.030%±0.010%、20.110%±0.600%,实验组的CD133阳性率显著高于HepG2组和对照组(P值均<0.001)。表阿霉素浓度为25、50 μmol/L时,HepG2组和对照组的细胞活性明显受到抑制,显著低于实验组(P值均<0.05)。50 μmol/L表阿霉素作用48 h后,实验组的细胞凋亡率(67.9%±2.5%)较HepG2组(93.6%±1.5%)和对照组(93.0%±1.2%)明显降低(P值均<0.001)。 结论 过表达HIF-1α的质粒成功转染至HepG2细胞,HIF-1α可提高肝癌细胞干细胞比例使其对表阿霉素耐药。 -
关键词:
- 肝肿瘤, 实验性 /
- 缺氧诱导因子1, α亚基 /
- 表柔比星 /
- 干细胞 /
- 抗药性, 肿瘤
Abstract:Objective To investigate the effect of hypoxia-inducible factor-1α (HIF-1α) on the stemness and epirubicin sensitivity of hepatoma cells. Methods Hepatoma cells were selected for experiment. HepG2 hepatoma cells transfected with HIF-1α overexpression plasmid were selected as experimental group, and those transfected with pcDNA3.1 empty plasmid were selected as control group; HepG2 cells alone were selected as HepG2 group. Quantitative real-time PCR was used to measure the mRNA expression of HIF-1α; Western blot was used to measure the protein expression of HIF-1α; flow cytometry was used to measure the expression of CD133 on the surface of hepatoma cells. The three groups of cells were treated with epirubicin at different concentrations (0, 6.25, 12.5, 25, and 50 μmol/L) for 24 hours; MTT assay was used to measure cell viability, and flow cytometry was used to measure apoptosis after treatment with epirubicin (50 μmol/L). A one-way analysis of variance was used for comparison of continuous data between multiple groups, and the t-test was used for further comparison between two groups. Results Compared with the HepG2 group and the control group, the experimental group had a significant increase in the mRNA expression of HIF-1α (both P < 0.001), and Western blot showed high expression of HIF-1α in the experimental group. The percentage of CD133 cells was 0.040%±0.003% in the HepG2 group, 0.030%±0.010% in the control group, and 20.110%±0.600% in the experimental group, and the experimental group had a significantly higher positive rate of CD133+ than the HepG2 group and the control group (both P < 0.001). At an epirubicin concentration of 25 and 50 μmol/L, the HepG2 group and the control group had significantly inhibited cell viability and a significantly lower cell viability than the experimental group (both P < 0.05). After the treatment with 50 μmol/L epirubicin for 48 hours, the experimental group had a significantly lower cell apoptosis rate than the HepG2 group (67.9%±2.5% vs 93.6%±1.5%, P < 0.001) and the control group (67.9%±2.5% vs 93.0%±1.2%, P < 0.001). Conclusion HepG2 cells are successfully transfected with HIF-1α overexpression plasmid, and HIF-1α can increase the percentage of liver cancer stem cells and improve their resistance to epirubicin. -
表 1 不同浓度表阿霉素作用24 h的细胞活性
组别 表阿霉素浓度(μmol/L) 0 6.25 12.5 25 50 HepG2组(%) 100 96.33±1.21 85.47±2.28 67.10±3.56 42.27±0.67 对照组(%) 100 92.60±1.54 86.80±2.40 59.03±2.60 45.50±0.56 实验组(%) 100 92.50±0.35 85.53±0.81 84.10±1.181)2) 72.30±2.071)2) F值 3.618 0.696 23.570 162.200 P值 0.093 0.535 0.001 <0.001 注:与对照组比较,1)P<0.05;与HepG2组比较,2)P<0.05。 -
[1] SIA D, VILLANUEVA A, FRIEDMAN SL, et al. Liver cancer cell of origin, molecular class, and effects on patient prognosis[J]. Gastroenterology, 2017, 152(4): 745-761. DOI: 10.1053/j.gastro.2016.11.048 [2] JIN J. Stem cell treatments[J]. JAMA, 2017, 317(3): 330. DOI: 10.1001/jama.2016.17822 [3] SUN JH, LUO Q, LIU LL, et al. Liver cancer stem cell markers: Progression and therapeutic implications[J]. World J Gastroenterol, 2016, 22(13): 3547-3557. DOI: 10.3748/wjg.v22.i13.3547 [4] HUANG Y, LIN D, TANIGUCHI CM. Hypoxia inducible factor (HIF) in the tumor microenvironment: Friend or foe?[J]. Sci China Life Sci, 2017, 60(10): 1114-1124. DOI: 10.1007/s11427-017-9178-y [5] QIAN J, RANKIN EB. Hypoxia-induced phenotypes that mediate tumor heterogeneity[J]. Adv Exp Med Biol, 2019, 1136: 43-55. [6] SAMANTA D, PARK Y, NI X, et al. Chemotherapy induces enrichment of CD47+/CD73+/PDL1+ immune evasive triple-negative breast cancer cells[J]. Proc Natl Acad Sci U S A, 2018, 115(6): e1239-e1248. DOI: 10.1073/pnas.1718197115 [7] LU H, SAMANTA D, XIANG L, et al. Chemotherapy triggers HIF-1-dependent glutathione synthesis and copper chelation that induces the breast cancer stem cell phenotype[J]. Proc Natl Acad Sci U S A, 2015, 112(33): e4600-e4609. DOI: 10.1073/pnas.1513433112 [8] LAN J, LU H, SAMANTA D, et al. Hypoxia-inducible factor 1-dependent expression of adenosine receptor 2B promotes breast cancer stem cell enrichment[J]. Proc Natl Acad Sci U S A, 2018, 115(41): e9640-e9648. DOI: 10.1073/pnas.1809695115 [9] SEMENZA GL. The genomics and genetics of oxygen homeostasis[J]. Annu Rev Genomics Hum Genet, 2020, 21: 183-204. DOI: 10.1146/annurev-genom-111119-073356 [10] MARIE-EGYPTIENNE DT, LOHSE I, HILL RP. Cancer stem cells, the epithelial to mesenchymal transition (EMT) and radioresistance: Potential role of hypoxia[J]. Cancer Lett, 2013, 341(1): 63-72. DOI: 10.1016/j.canlet.2012.11.019 [11] NOZAWA-SUZUKI N, NAGASAWA H, OHNISHI K, et al. The inhibitory effect of hypoxic cytotoxin on the expansion of cancer stem cells in ovarian cancer[J]. Biochem Biophys Res Commun, 2015, 457(4): 706-711. DOI: 10.1016/j.bbrc.2015.01.053 [12] BEN-PORATH I, THOMSON MW, CAREY VJ, et al. An embryonic stem cell-like gene expression signature in poorly differentiated aggressive human tumors[J]. Nat Genet, 2008, 40(5): 499-507. DOI: 10.1038/ng.127 [13] ALDERSON D, CUNNINGHAM D, NANKIVELL M, et al. Neoadjuvant cisplatin and fluorouracil versus epirubicin, cisplatin, and capecitabine followed by resection in patients with oesophageal adenocarcinoma (UK MRC OE05): An open-label, randomised phase 3 trial[J]. Lancet Oncol, 2017, 18(9): 1249-1260. DOI: 10.1016/S1470-2045(17)30447-3 [14] SONG B, BIAN Q, SHAO CH, et al. Ulinastatin reduces the resistance of liver cancer cells to epirubicin by inhibiting autophagy[J]. PLoS One, 2015, 10(3): e0120694. DOI: 10.1371/journal.pone.0120694 [15] LI J, DUAN B, GUO Y, et al. Baicalein sensitizes hepatocellular carcinoma cells to 5-FU and Epirubicin by activating apoptosis and ameliorating P-glycoprotein activity[J]. Biomed Pharmacother, 2018, 98: 806-812. DOI: 10.1016/j.biopha.2018.01.002 [16] PLAKS V, KONG N, WERB Z. The cancer stem cell niche: How essential is the niche in regulating stemness of tumor cells?[J]. Cell Stem Cell, 2015, 16(3): 225-238. DOI: 10.1016/j.stem.2015.02.015 -



 PDF下载 ( 2169 KB)
PDF下载 ( 2169 KB)

 下载:
下载: