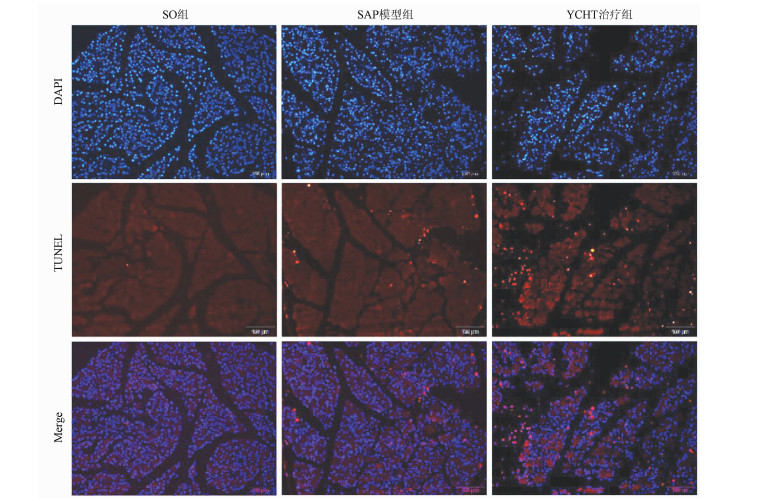
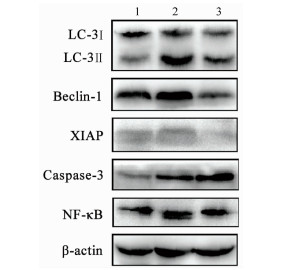
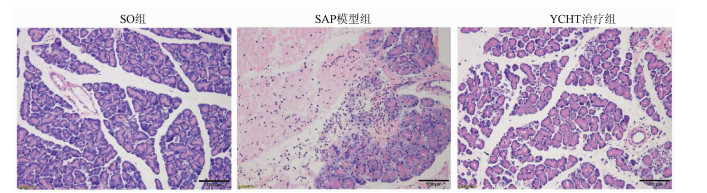
茵陈蒿汤调控lncRNA PVT1/miRNA-30a-5p信号通路对重症急性胰腺炎大鼠模型的保护作用
DOI: 10.3969/j.issn.1001-5256.2021.01.029
Artemisia capillaris Thunb. decoction exerts a protective effect on rats with severe acute pancreatitis by regulating the lncRNA PVT1/miRNA-30a-5p signaling pathway
-
摘要:
目的 探讨清热利胆的经典中药方剂——茵陈蒿汤(YCHT)对牛磺胆酸钠诱导的大鼠重症急性胰腺炎(SAP)的保护作用及其机制。 方法 30只SD大鼠随机分成假手术(SO)组、SAP模型组和YCHT (4.0 g/kg)治疗组,每组10只。造模成功24 h后,留取大鼠胰腺组织和血浆待检测。HE染色观察胰腺病理损伤情况; ELISA法检测血浆淀粉酶、TNFα和IL-1β水平; 免疫荧光染色检测LC-3蛋白的荧光强度,TUNEL检测细胞凋亡情况。Western Blot检测胰腺组织LC-3、Beclin-1、X连锁凋亡抑制蛋白(XIAP)、Caspase-3和NF-κB蛋白表达。实时荧光定量PCR检测lncRNA PVT1和miRNA-30a-5p的表达水平。采用单因素方差分析和Tukey’s检验用于分析多个独立样本之间的差异。 结果 YCHT能明显减轻SAP大鼠胰腺组织水肿、坏死、出血及炎性细胞浸润等病理学损伤。与SO组相比,SAP模型组大鼠血浆淀粉酶、炎性因子TNFα和IL-1β水平显著升高,YCHT治疗后血浆淀粉酶及TNFα和IL-1β水平显著降低(P值均<0.05)。与SO组相比,SAP模型组LC-3Ⅱ/LC-3Ⅰ比值及Beclin-1、XIAP、Caspase-3和NF-κB蛋白表达明显上调,YCHT治疗组LC-3Ⅱ/LC-3Ⅰ、Beclin-1、XIAP和NF-κB的表达水平较SAP模型组显著降低,Caspase-3水平显著升高(P值均<0.05)。与SO组相比,SAP模型组大鼠胰腺lncRNA PVT1表达显著升高,miRNA-30a-5p表达显著降低(P值均<0.05);与SAP模型组相比,YCHT显著降低了lncRNA PVT1的表达,增加了miRNA-30a-5p的表达(P值均<0.05)。 结论 lncRNA PVT1/miRNA-30a-5p介导的细胞自噬和凋亡可能是YCHT治疗SAP的一个药物靶点,这为进一步开发中药方剂YCHT治疗SAP提供了实验基础和理论依据。 -
关键词:
- 胰腺炎 /
- 大鼠,Sprague-Dawley /
- 茵陈蒿汤 /
- RNA,长链非编码 /
- 微RNAs
Abstract:Objective To investigate the protective effect and mechanism of Artemisia capillaris Thunb. decoction (YCHT), a classic heat-clearing and cholagogic traditional Chinese medicine (TCM) prescription, on rats with severe acute pancreatitis (SAP) induced by sodium taurocholate. Methods A total of 30 Sprague-Dawley rats were randomly divided into sham-operation (SO) group, SAP model group, and YCHT (4.0 g/kg) treatment group, with 10 rats in each group. At 24 hours after successful modeling, pancreatic tissue and plasma samples were collected for analysis. HE staining was used to observe pathological injury of the pancreas; ELISA was used to measure the plasma levels of amylase, tumor necrosis factor-α (TNFα), and interleukin-1β (IL-1β); immunofluorescent staining was used to measure the fluorescence intensity of LC-3 protein, and TUNEL was used to measure cell apoptosis. Western blot was used to measure the protein expression of LC-3, Beclin-1, X-linked inhibitor of apoptosis protein (XIAP), caspase-3, and nuclear factor-kappa B (NF-κB) in the pancreas, and quantitative real-time PCR was used to measure the expression levels of lncRNA PVT1 and miRNA-30a-5p. A one-way analysis of variance and the Tukey's test were used to analyze the differences between multiple independent samples. Results YCHT significantly alleviated the pathological injury of the pancreas of SAP rats, such as edema, necrosis, hemorrhage, and inflammatory cell infiltration. Compared with the SO group, the SAP group had significant increases in the plasma levels of amylase and the inflammatory factors TNFα and IL-1β, and there were significant reductions in the plasma levels of amylase, TNFα, and IL-1β after YCHT treatment (all P < 0.05). Compared with the SO group, the SAP group had significant increases in LC-3II/LC-3I ratio and the protein expression of Beclin-1, XIAP, caspase-3, and NF-κB, and compared with the SAP group, the YCHT group had significant reductions in LC-3II/LC-3I ratio and the protein expression of Beclin-1, XIAP, and NF-κB (all P < 0.05). Compared with the SO group, the SAP group had a significant increase in the expression of lncRNA PVT1 and a significant reduction in the expression of miRNA-30a-5p in the pancreas (both P < 0.05), and compared with the SAP group, the YCHT group had a significant reduction in the expression of lncRNA PVT1 and a significant increase in the expression of miRNA-30a-5p (both P < 0.05). Conclusion Cell autophagy and apoptosis mediated by lncRNA PVT1/miRNA-30a-5p may be a drug target for YCHT treatment of SAP, which provides experimental and theoretical bases for further development of the TCM prescription YCHT for the treatment of SAP. -
表 1 引物序列
基因 引物序列 lncRNA PVT1 R:5′-ACCAGGATATAAACCTAGTGATAAA-3′ F:5′-AAGCACCCAATGCAGAATAG-3′ GAPDH R:5′-ATGTTCCAGTATGACTCTA-3′ F:5′-CACCCCATTTGATGTTAG-3′ U6 R:5′-GGAACGATACAGAGAAGATTAGC-3′ F:5′-TGGAACGCTTCACGAATTTGCG-3′ 表 2 各组大鼠胰腺组织病理学评分以及血浆淀粉酶、TNFα、IL-1β水平的比较
组别 大鼠数(只) 病理学评分 淀粉酶(μmol/L) TNFα(ng/L) IL-1β(ng/L) SO组 10 1.667±0.577 2.259±0.935 4.444±1.684 2.129±0.931 SAP模型组 10 13.000±1.0001) 16.156±3.9161) 42.791±10.5021) 11.054±0.8501) YCHT治疗组 10 5.000±1.0002) 4.400±1.0892) 14.250±2.8212) 4.739±0.7272) F值 130.90 95.61 98.13 298.50 P值 <0.001 <0.001 <0.001 <0.001 注:与SO组相比,1)P<0.05;与SAP模型组相比,2)P<0.05。 表 3 各组自噬和凋亡标记蛋白相对表达量的比较
组别 大鼠数(只) LC-3Ⅱ/LC-3Ⅰ Beclin-1 XIAP Caspase-3 NF-κB SO组 10 0.360±0.049 0.767±0.017 0.227±0.002 0.264±0.035 0.701±0.007 SAP模型组 10 3.945±0.0931) 1.289±0.0131) 0.254±0.0041) 0.618±0.0351) 0.754±0.0001) YCHT治疗组 10 1.667±0.0622) 0.310±0.0212) 0.102±0.0162) 1.278±0.0612) 0.682±0.0142) F值 1498.0 2482.0 204.4 272.4 48.9 P值 <0.001 <0.001 <0.001 <0.001 <0.001 注:与SO组相比,1)P<0.05;与SAP模型组相比,2)P<0.05。 表 4 YCHT对胰腺组织中lncRNA-PVT1和miRNA-30a-5p相对表达量的影响
组别 大鼠数(只) lncRNA-PVT1 miRNA-30a-5p SO组 10 1.003±0.019 1.003±0.089 SAP模型组 10 1.626±0.0601) 0.021±0.0031) YCHT治疗组 10 0.253±0.1782) 3.404±0.1292) F值 119.7 1111.0 P值 <0.001 <0.001 注:与SO组相比,1)P<0.05;与SAP模型组相比,2)P<0.05。 -
[1] LANKISCH PG, APTE M, BANKS PA. Acute pancreatitis[J]. Lancet, 2015, 386(9988): 85-96. DOI: 10.1016/S0140-6736(14)60649-8 [2] XIANG H, ZHANG Q, QI B, et al. Chinese herbal medicines attenuate acute pancreatitis: Pharmacological activities and mechanisms[J]. Front Pharmacol, 2017, 8: 216. DOI: 10.3389/fphar.2017.00216 [3] SENDLER M, WEISS FU, GOLCHERT J, et al. Cathepsin B-mediated activation of trypsinogen in endocytosing macrophages increases severity of pancreatitis in mice[J]. Gastroenterology, 2018, 154(3): 704-718.e10. DOI: 10.1053/j.gastro.2017.10.018 [4] LIU Y, CHEN XD, YU J, et al. Deletion Of XIAP reduces the severity of acute pancreatitis via regulation of cell death and nuclear factor-κB activity[J]. Cell Death Dis, 2017, 8(3): e2685. DOI: 10.1038/cddis.2017.70 [5] LEVINE B. Cell biology: Autophagy and cancer[J]. Nature, 2007, 446(7137): 745-747. DOI: 10.1038/446745a [6] SU Z, YANG Z, XU Y, et al. MicroRNAs in apoptosis, autophagy and necroptosis[J]. Oncotarget, 2015, 6(11): 8474-8490. DOI: 10.18632/oncotarget.3523 [7] LI Y, JIANG J, LIU W, et al. microRNA-378 promotes autophagy and inhibits apoptosis in skeletal muscle[J]. Proc Natl Acad Sci U S A, 2018, 115(46): e10849-e10858. DOI: 10.1073/pnas.1803377115 [8] ZHOU W, XU J, WANG C, et al. miR-23b-3p regulates apoptosis and autophagy via suppressing SIRT1 in lens epithelial cells[J]. J Cell Biochem, 2019, 120(12): 19635-19646. DOI: 10.1002/jcb.29270 [9] QUINN JJ, CHANG HY. Unique features of long non-coding RNA biogenesis and function[J]. Nat Rev Genet, 2016, 17(1): 47-62. DOI: 10.1038/nrg.2015.10 [10] TIAN Q, LIU F, XU Z, et al. Evaluation of the chemical consistency of Yin-Chen-Hao-Tang prepared by combined and separated decoction methods using high-performance liquid chromatography and quadrupole time-of-flight mass spectrometry coupled with multivariate statistical analysis[J]. J Sep Sci, 2019, 42(9): 1664-1675. DOI: 10.1002/jssc.201800961 [11] XIANG H, WANG G, QU J, et al. Yin-Chen-Hao Tang attenuates severe acute pancreatitis in rat: An experimental verification of in silico network target prediction[J]. Front Pharmacol, 2016, 7: 378. DOI: 10.3389/fphar.2016.00378 [12] XIANG H, TAO X, XIA S, et al. Emodin alleviates sodium taurocholate-induced pancreatic acinar cell injury via MicroRNA-30a-5p-mediated inhibition of high-temperature requirement a/transforming growth factor beta 1 inflammatory signaling[J]. Front Immunol, 2017, 8: 1488. DOI: 10.3389/fimmu.2017.01488 [13] LEE PJ, PAPACHRISTOU GI. New insights into acute pancreatitis[J]. Nat Rev Gastroenterol Hepatol, 2019, 16(8): 479-496. DOI: 10.1038/s41575-019-0158-2 [14] HAHM KB, KIM JH, YOU BM, et al. Induction of apoptosis with an extract of Artemisia asiatica attenuates the severity of cerulein-induced pancreatitis in rats[J]. Pancreas, 1998, 17(2): 153-157. DOI: 10.1097/00006676-199808000-00007 [15] IWAHASHI K, HIKITA H, MAKINO Y, et al. Autophagy impairment in pancreatic acinar cells causes zymogen granule accumulation and pancreatitis[J]. Biochem Biophys Res Commun, 2018, 503(4): 2576-2582. DOI: 10.1016/j.bbrc.2018.07.018 [16] GUKOVSKAYA AS, PERKINS P, ZANINOVIC V, et al. Mechanisms of cell death after pancreatic duct obstruction in the opossum and the rat[J]. Gastroenterology, 1996, 110(3): 875-884. DOI: 10.1053/gast.1996.v110.pm8608898 [17] PARZYCH KR, KLIONSKY DJ. An overview of autophagy: Morphology, mechanism, and regulation[J]. Antioxid Redox Signal, 2014, 20(3): 460-473. DOI: 10.1089/ars.2013.5371 [18] WANG S, DING WX. Does autophagy promote or protect against the pathogenesis of pancreatitis?[J]. Gastroenterology, 2018, 155(4): 1273-1274. DOI: 10.1053/j.gastro.2018.02.046 [19] KONDO Y, KANZAWA T, SAWAYA R, et al. The role of autophagy in cancer development and response to therapy[J]. Nat Rev Cancer, 2005, 5(9): 726-734. DOI: 10.1038/nrc1692 [20] YANG Y, HUANG Q, LUO C, et al. MicroRNAs in acute pancreatitis: From pathogenesis to novel diagnosis and therapy[J]. J Cell Physiol, 2020, 235(3): 1948-1961. DOI: 10.1002/jcp.29212 [21] XIANG H, TAO X, XIA S, et al. Targeting MicroRNA function in acute pancreatitis[J]. Front Physiol, 2017, 8: 726. DOI: 10.3389/fphys.2017.00726 [22] ABREU FB, LIU X, TSONGALIS GJ. miRNA analysis in pancreatic cancer: The Dartmouth experience[J]. Clin Chem Lab Med, 2017, 55(5): 755-762. [23] QUAN X, LI X, YIN Z, et al. p53/miR-30a-5p/ SOX4 feedback loop mediates cellular proliferation, apoptosis, and migration of non-small-cell lung cancer[J]. J Cell Physiol, 2019, 234(12): 22884-22895. DOI: 10.1002/jcp.28851 [24] KIM JW, YOU YH, JUNG S, et al. miRNA-30a-5p-mediated silencing of Beta2/NeuroD expression is an important initial event of glucotoxicity-induced beta cell dysfunction in rodent models[J]. Diabetologia, 2013, 56(4): 847-855. DOI: 10.1007/s00125-012-2812-x [25] ZHOU L, JIA S, DING G, et al. Down-regulation of miR-30a-5p is associated with poor prognosis and promotes chemoresistance of gemcitabine in pancreatic ductal adenocarcinoma[J]. J Cancer, 2019, 10(21): 5031-5040. DOI: 10.7150/jca.31191 [26] YANG X, BAI F, XU Y, et al. Intensified Beclin-1 Mediated by low expression of Mir-30a-5p promotes chemoresistance in human small cell lung cancer[J]. Cell Physiol Biochem, 2017, 43(3): 1126-1139. DOI: 10.1159/000481754 [27] XU X, JIN S, MA Y, et al. miR-30a-5p enhances paclitaxel sensitivity in non-small cell lung cancer through targeting BCL-2 expression[J]. J Mol Med (Berl), 2017, 95(8): 861-871. DOI: 10.1007/s00109-017-1539-z [28] YAO RW, WANG Y, CHEN LL. Cellular functions of long noncoding RNAs[J]. Nat Cell Biol, 2019, 21(5): 542-551. DOI: 10.1038/s41556-019-0311-8 -



 PDF下载 ( 6634 KB)
PDF下载 ( 6634 KB)

 下载:
下载: