| [1] |
SUNG H, FERLAY J, SIEGEL RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2021, 71( 3): 209- 249. DOI: 10.3322/caac.21660. |
| [2] |
WANG Y, DENG BC. Hepatocellular carcinoma: Molecular mechanism, targeted therapy, and biomarkers[J]. Cancer Metastasis Rev, 2023, 42( 3): 629- 652. DOI: 10.1007/s10555-023-10084-4. |
| [3] |
ZHANG C, LIU XY, JIN SD, et al. Ferroptosis in cancer therapy: A novel approach to reversing drug resistance[J]. Mol Cancer, 2022, 21( 1): 47. DOI: 10.1186/s12943-022-01530-y. |
| [4] |
ZHU HY, YANG YJ, DUAN YN, et al. Nrf2/FSP1/CoQ10 axis-mediated ferroptosis is involved in sodium aescinate-induced nephrotoxicity[J]. Arch Biochem Biophys, 2024, 759: 110100. DOI: 10.1016/j.abb.2024.110100. |
| [5] |
CAO JH, CHEN X, CHEN LL, et al. DHODH-mediated mitochondrial redox homeostasis: A novel ferroptosis regulator and promising therapeutic target[J]. Redox Biol, 2025, 85: 103788. DOI: 10.1016/j.redox.2025.103788. |
| [6] |
JIANG XJ, STOCKWELL BR, CONRAD M. Ferroptosis: Mechanisms, biology and role in disease[J]. Nat Rev Mol Cell Biol, 2021, 22( 4): 266- 282. DOI: 10.1038/s41580-020-00324-8. |
| [7] |
DIXON SJ, LEMBERG KM, LAMPRECHT MR, et al. Ferroptosis: An iron-dependent form of nonapoptotic cell death[J]. Cell, 2012, 149( 5): 1060- 1072. DOI: 10.1016/j.cell.2012.03.042. |
| [8] |
STOCKWELL BR. Ferroptosis turns 10: Emerging mechanisms, physiological functions, and therapeutic applications[J]. Cell, 2022, 185( 14): 2401- 2421. DOI: 10.1016/j.cell.2022.06.003. |
| [9] |
NGHIEM TH THI, KUSUMA F, PARK J, et al. Brief guide to detecting ferroptosis[J]. Mol Cells, 2025, 48( 11): 100276. DOI: 10.1016/j.mocell.2025.100276. |
| [10] |
MARTINEZ AM, KIM A, YANG WS. Detection of ferroptosis by BODIPY TM 581/591 C11[J]. Methods Mol Biol, 2020, 2108: 125- 130. DOI: 10.1007/978-1-0716-0247-8_11. |
| [11] |
YAN HF, ZOU T, TUO QZ, et al. Ferroptosis: Mechanisms and links with diseases[J]. Signal Transduct Target Ther, 2021, 6( 1): 49. DOI: 10.1038/s41392-020-00428-9. |
| [12] |
ZILKA O, SHAH R, LI B, et al. On the mechanism of cytoprotection by ferrostatin-1 and liproxstatin-1 and the role of lipid peroxidation in ferroptotic cell death[J]. ACS Cent Sci, 2017, 3( 3): 232- 243. DOI: 10.1021/acscentsci.7b00028. |
| [13] |
LI WT, LIANG L, LIU SY, et al. FSP1: A key regulator of ferroptosis[J]. Trends Mol Med, 2023, 29( 9): 753- 764. DOI: 10.1016/j.molmed.2023.05.013. |
| [14] |
MISHIMA E, ITO J, WU ZJ, et al. A non-canonical vitamin K cycle is a potent ferroptosis suppressor[J]. Nature, 2022, 608( 7924): 778- 783. DOI: 10.1038/s41586-022-05022-3. |
| [15] |
FANG WX, XIE SY, DENG W. Ferroptosis mechanisms and regulations in cardiovascular diseases in the past, present, and future[J]. Cell Biol Toxicol, 2024, 40( 1): 17. DOI: 10.1007/s10565-024-09853-w. |
| [16] |
WEAVER K, SKOUTA R. The selenoprotein glutathione peroxidase 4: From molecular mechanisms to novel therapeutic opportunities[J]. Biomedicines, 2022, 10( 4): 891. DOI: 10.3390/biomedicines10040891. |
| [17] |
CONRAD M, KAGAN VE, BAYIR H, et al. Regulation of lipid peroxidation and ferroptosis in diverse species[J]. Genes Dev, 2018, 32( 9-10): 602- 619. DOI: 10.1101/gad.314674.118. |
| [18] |
BERSUKER K, HENDRICKS JM, LI ZP, et al. The CoQ oxidoreductase FSP1 acts parallel to GPX4 to inhibit ferroptosis[J]. Nature, 2019, 575( 7784): 688- 692. DOI: 10.1038/s41586-019-1705-2. |
| [19] |
DOLL S, FREITAS FP, SHAH R, et al. FSP1 is a glutathione-independent ferroptosis suppressor[J]. Nature, 2019, 575( 7784): 693- 698. DOI: 10.1038/s41586-019-1707-0. |
| [20] |
WANG C, CHEN CC, ZHOU JB, et al. DHODH alleviates heart failure via the modulation of CoQ-related ferroptotic inhibition[J]. Front Biosci, 2024, 29( 7): 267. DOI: 10.31083/j.fbl2907267. |
| [21] |
PENG W, LIANG J, QIAN XL, et al. IGF2BP1/AIFM2 axis regulates ferroptosis and glycolysis to drive hepatocellular carcinoma progression[J]. Cell Signal, 2025, 130: 111660. DOI: 10.1016/j.cellsig.2025.111660. |
| [22] |
PEI WC, JIANG MR, LIU HY, et al. The prognostic and antitumor roles of key genes of ferroptosis in liver hepatocellular cancer and stomach adenocarcinoma[J]. Cancer Biomark, 2024, 39( 4): 335- 347. DOI: 10.3233/CBM-230114. |
| [23] |
CHEU JW, LEE D, LI QD, et al. Ferroptosis suppressor protein 1 inhibition promotes tumor ferroptosis and anti-tumor immune responses in liver cancer[J]. Cell Mol Gastroenterol Hepatol, 2023, 16( 1): 133- 159. DOI: 10.1016/j.jcmgh.2023.03.001. |
| [24] |
KOPPULA P, LEI G, ZHANG YL, et al. A targetable CoQ-FSP1 axis drives ferroptosis- and radiation-resistance in KEAP1 inactive lung cancers[J]. Nat Commun, 2022, 13( 1): 2206. DOI: 10.1038/s41467-022-29905-1. |
| [25] |
EMMANUEL N, LI HE, CHEN J, et al. FSP1 a novel KEAP1/NRF2 target gene regulating ferroptosis and radioresistance in lung cancers[J]. Oncotarget, 2022, 13: 1136- 1139. DOI: 10.18632/oncotarget.28301. |
| [26] |
LEE J, SEO Y, ROH JL. Emerging therapeutic strategies targeting GPX4-mediated ferroptosis in head and neck cancer[J]. Int J Mol Sci, 2025, 26( 13): 6452. DOI: 10.3390/ijms26136452. |
| [27] |
CHEN YL, LEE D, KWAN KK, et al. Mevalonate pathway promotes liver cancer by suppressing ferroptosis through CoQ10 production and selenocysteine-tRNA modification[J]. J Hepatol, 2025, 83( 6): 1338- 1352. DOI: 10.1016/j.jhep.2025.06.034. |
| [28] |
GONG J, LIU YH, WANG WJ, et al. TRIM21-promoted FSP1 plasma membrane translocation confers ferroptosis resistance in human cancers[J]. Adv Sci, 2023, 10( 29): 2302318. DOI: 10.1002/advs.202302318. |
| [29] |
LIU MR, SHI C, SONG QY, et al. Sorafenib induces ferroptosis by promoting TRIM54-mediated FSP1 ubiquitination and degradation in hepatocellular carcinoma[J]. Hepatol Commun, 2023, 7( 10): e0246. DOI: 10.1097/HC9.0000000000000246. |
| [30] |
YUAN JS, LV T, YANG J, et al. HDLBP-stabilized lncFAL inhibits ferroptosis vulnerability by diminishing Trim69-dependent FSP1 degradation in hepatocellular carcinoma[J]. Redox Biol, 2022, 58: 102546. DOI: 10.1016/j.redox.2022.102546. |
| [31] |
ZHU XY, SHA XD, ZANG Y, et al. Current progress of ferroptosis study in hepatocellular carcinoma[J]. Int J Biol Sci, 2024, 20( 9): 3621- 3637. DOI: 10.7150/ijbs.96014. |
| [32] |
ISEDA N, ITOH S, TOSHIDA K, et al. Ferroptosis is induced by lenvatinib through fibroblast growth factor receptor-4 inhibition in hepatocellular carcinoma[J]. Cancer Sci, 2022, 113( 7): 2272- 2287. DOI: 10.1111/cas.15378. |
| [33] |
ZENG FR, CHEN X, DENG GT. The anti-ferroptotic role of FSP1: Current molecular mechanism and therapeutic approach[J]. Mol Biomed, 2022, 3( 1): 37. DOI: 10.1186/s43556-022-00105-z. |
| [34] |
XAVIER DA SILVA TN, SCHULTE C, ALVES AN, et al. Molecular characterization of AIFM2/FSP1 inhibition by iFSP1-like molecules[J]. Cell Death Dis, 2023, 14( 4): 281. DOI: 10.1038/s41419-023-05787-z. |
| [35] |
NAKAMURA T, MISHIMA E, YAMADA N, et al. Integrated chemical and genetic screens unveil FSP1 mechanisms of ferroptosis regulation[J]. Nat Struct Mol Biol, 2023, 30( 11): 1806- 1815. DOI: 10.1038/s41594-023-01136-y. |
| [36] |
HENDRICKS JM, DOUBRAVSKY CE, WEHRI E, et al. Identification of structurally diverse FSP1 inhibitors that sensitize cancer cells to ferroptosis[J]. Cell Chem Biol, 2023, 30( 9): 1090- 1103. DOI: 10.1016/j.chembiol.2023.04.007. |
| [37] |
NAKAMURA T, HIPP C, SANTOS DIAS MOURÃO A, et al. Phase separation of FSP1 promotes ferroptosis[J]. Nature, 2023, 619( 7969): 371- 377. DOI: 10.1038/s41586-023-06255-6. |
| [38] |
MAO C, LIU XG, ZHANG YL, et al. DHODH-mediated ferroptosis defence is a targetable vulnerability in cancer[J]. Nature, 2021, 593( 7860): 586- 590. DOI: 10.1038/s41586-021-03539-7. |
| [39] |
MA TY, DU JT, ZHANG YF, et al. GPX4-independent ferroptosis-a new strategy in disease’s therapy[J]. Cell Death Discov, 2022, 8( 1): 434. DOI: 10.1038/s41420-022-01212-0. |
| [40] |
WANG FD, MIN JX. DHODH tangoing with GPX4 on the ferroptotic stage[J]. Signal Transduct Target Ther, 2021, 6( 1): 244. DOI: 10.1038/s41392-021-00656-7. |
| [41] |
MULLEN NJ, SHUKLA SK, THAKUR R, et al. DHODH inhibition enhances the efficacy of immune checkpoint blockade by increasing cancer cell antigen presentation[J]. eLife, 2024, 12: RP87292. DOI: 10.7554/eLife.87292. |
| [42] |
YUAN YH, SUN WJ, XIE JQ, et al. RNA nanotherapeutics for hepatocellular carcinoma treatment[J]. Theranostics, 2025, 15( 3): 965- 992. DOI: 10.7150/thno.102964. |
| [43] |
ZHENG XG, JIN XD, YE F, et al. Ferroptosis: A novel regulated cell death participating in cellular stress response, radiotherapy, and immunotherapy[J]. Exp Hematol Oncol, 2023, 12( 1): 65. DOI: 10.1186/s40164-023-00427-w. |
| [44] |
SCHWARTSMANN G, DODION P, VERMORKEN JB, et al. Phase I study of Brequinar sodium(NSC 368390) in patients with solid malignancies[J]. Cancer Chemother Pharmacol, 1990, 25( 5): 345- 351. DOI: 10.1007/BF00686235. |
| [45] |
BANKA VK, SAINAS S, MARTINO E, et al. Radiosynthesis of[ 18F] brequinar for in vivo PET imaging of hDHODH for potential studies of acute myeloid leukemia and cancers[J]. RSC Med Chem, 2024, 15( 9): 3147- 3161. DOI: 10.1039/d4md00433g. |
| [46] |
KEPP O, KROEMER G. Is ferroptosis immunogenic? The devil is in the details![J]. Oncoimmunology, 2022, 11( 1): 2127273. DOI: 10.1080/2162402X.2022.2127273. |
| [47] |
GAO CL, ZHANG HR, WANG XW. Current advances on the role of ferroptosis in tumor immune evasion[J]. Discov Oncol, 2024, 15( 1): 736. DOI: 10.1007/s12672-024-01573-1. |
| [48] |
LOPES-COELHO F, MARTINS F, HIPÓLITO A, et al. The activation of endothelial cells relies on a ferroptosis-like mechanism: Novel perspectives in management of angiogenesis and cancer therapy[J]. Front Oncol, 2021, 11: 656229. DOI: 10.3389/fonc.2021.656229. |
| [49] |
ZHOU Y, TAO L, ZHOU X, et al. DHODH and cancer: Promising prospects to be explored[J]. Cancer Metab, 2021, 9( 1): 22. DOI: 10.1186/s40170-021-00250-z. |
| [50] |
CHEN HS, WANG CY, LIU ZM, et al. Ferroptosis and its multifaceted role in cancer: Mechanisms and therapeutic approach[J]. Antioxidants, 2022, 11( 8): 1504. DOI: 10.3390/antiox11081504. |
| [51] |
CHEN FQ, KANG R, LIU J, et al. The ACSL4 network regulates cell death and autophagy in diseases[J]. Biology, 2023, 12( 6): 864. DOI: 10.3390/biology12060864. |
| [52] |
DOLL S, PRONETH B, TYURINA YY, et al. ACSL4 dictates ferroptosis sensitivity by shaping cellular lipid composition[J]. Nat Chem Biol, 2017, 13( 1): 91- 98. DOI: 10.1038/nchembio.2239. |
| [53] |
EBELING MC, POLANCO JR, QU J, et al. Improving retinal mitochondrial function as a treatment for age-related macular degeneration[J]. Redox Biol, 2020, 34: 101552. DOI: 10.1016/j.redox.2020.101552. |



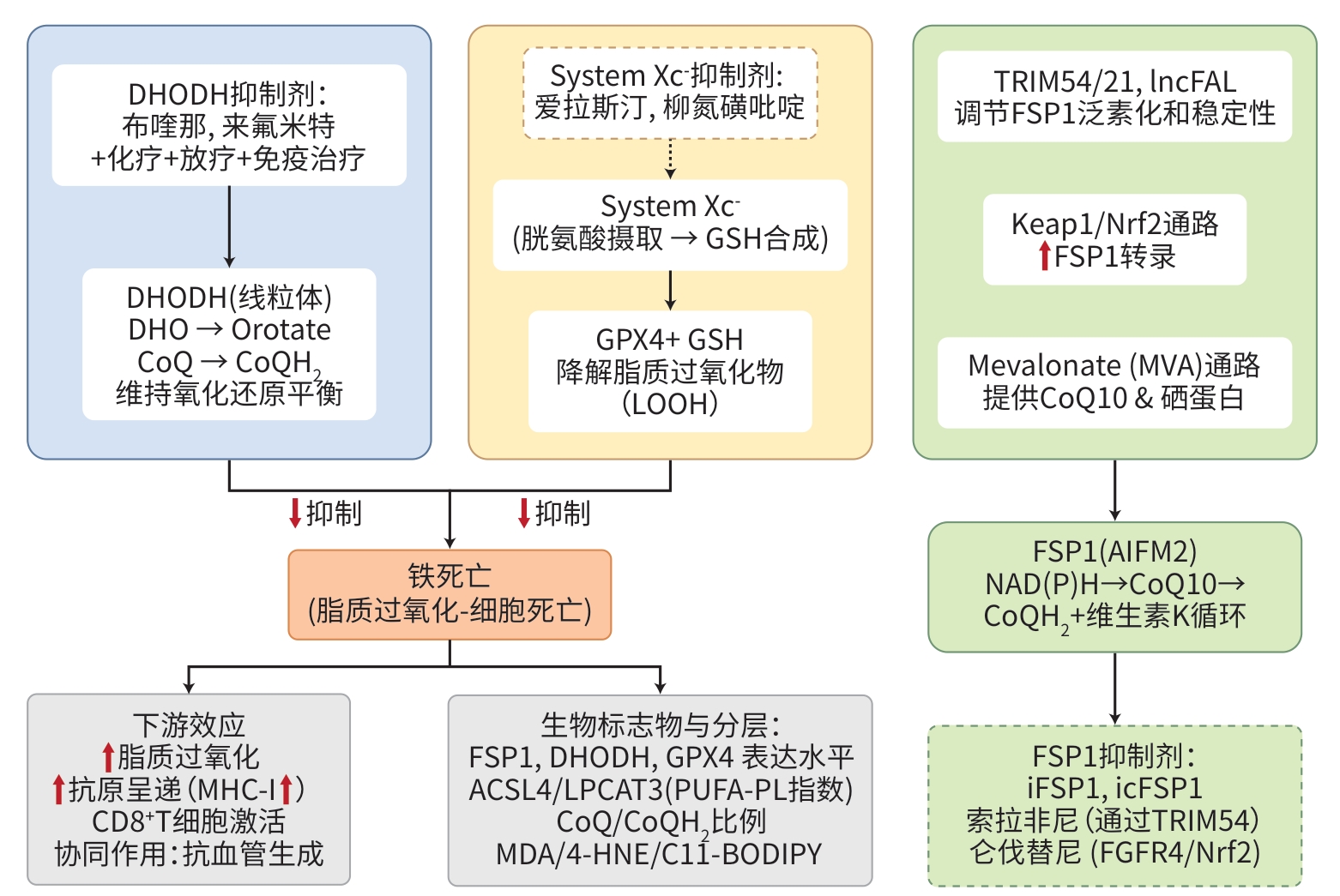




 DownLoad:
DownLoad: