| [1] |
VALM AM, COHEN S, LEGANT WR, et al. Applying systems-level spectral imaging and analysis to reveal the organelle interactome[J]. Nature, 2017, 546( 7656): 162- 167. DOI: 10.1038/nature22369. |
| [2] |
LIAO ZH, XIE ZY. Research progress in molecular mechanism of hepatic fibrosis and related therapeutic targets[J]. J Jilin Univ Med Ed, 2024, 50( 5): 1450- 1456. DOI: 10.13481/j.1671-587X.20240532. |
| [3] |
ILHA M, SEHGAL R, MATILAINEN J, et al. Indole-3-propionic acid promotes hepatic stellate cells inactivation[J]. J Transl Med, 2025, 23( 1): 253. DOI: 10.1186/s12967-025-06266-z. |
| [4] |
KIM H, CHEN Q, JU DH, et al. ER-tethered stress sensor CREBH regulates mitochondrial unfolded protein response to maintain energy homeostasis[J]. Proc Natl Acad Sci USA, 2024, 121( 49): e2410486121. DOI: 10.1073/pnas.2410486121. |
| [5] |
JIANG Y, LI LL, CHEN X, et al. Three-dimensional ATUM-SEM reconstruction and analysis of hepatic endoplasmic reticulum-organelle interactions[J]. J Mol Cell Biol, 2021, 13( 9): 636- 645. DOI: 10.1093/jmcb/mjab032. |
| [6] |
GUPTA P, SATA TN, YADAV AK, et al. TGF-β induces liver fibrosis via miRNA-181a-mediated down regulation of augmenter of liver regeneration in hepatic stellate cells[J]. PLoS One, 2019, 14( 6): e0214534. DOI: 10.1371/journal.pone.0214534. |
| [7] |
WANG XL, HE JH, XIE P, et al. Augmenter of liver regeneration crotonylation assists in mitochondria-ER contact to alleviate hepatic steatosis[J]. Cell Mol Gastroenterol Hepatol, 2025, 19( 3): 101436. DOI: 10.1016/j.jcmgh.2024.101436. |
| [8] |
WU HX, CARVALHO P, VOELTZ GK. Here, there, and everywhere: The importance of ER membrane contact sites[J]. Science, 2018, 361( 6401): eaan5835. DOI: 10.1126/science.aan5835. |
| [9] |
HERNÁNDEZ-ALVAREZ MI, SEBASTIÁN D, VIVES S, et al. Deficient endoplasmic reticulum-mitochondrial phosphatidylserine transfer causes liver disease[J]. Cell, 2019, 177( 4): 881- 895.e17. DOI: 10.1016/j.cell.2019.04.010. |
| [10] |
ILHA M, MEIRA MARTINS LA, SILVEIRA MORAES K DA, et al. Caveolin-1 influences mitochondrial plasticity and function in hepatic stellate cell activation[J]. Cell Biol Int, 2022, 46( 11): 1787- 1800. DOI: 10.1002/cbin.11876. |
| [11] |
ZIEGLER DV, VINDRIEUX D, GOEHRIG D, et al. Calcium channel ITPR2 and mitochondria-ER contacts promote cellular senescence and aging[J]. Nat Commun, 2021, 12( 1): 720. DOI: 10.1038/s41467-021-20993-z. |
| [12] |
KIM SH, KIM JY, PARK SY, et al. Activation of the EGFR-PI3K-CaM pathway by PRL-1-overexpressing placenta-derived mesenchymal stem cells ameliorates liver cirrhosis via ER stress-dependent calcium[J]. Stem Cell Res Ther, 2021, 12( 1): 551. DOI: 10.1186/s13287-021-02616-y. |
| [13] |
SUN T, LV HM, SHAO HR, et al. Chondroitin sulfate as a lysosomal enhancer attenuates lipid-driven inflammation via lipophagy and mitophagy[J]. Mar Drugs, 2025, 23( 6): 228. DOI: 10.3390/md23060228. |
| [14] |
PAN M, SHI XY. Role of mitophagy in the development and progression of liver-related diseases[J]. J Clin Hepatol, 2024, 40( 2): 413- 418. DOI: 10.12449/JCH240232. 潘萌, 史晓燕. 线粒体自噬在肝脏相关疾病发生发展中的作用[J]. 临床肝胆病杂志, 2024, 40( 2): 413- 418. DOI: 10.12449/JCH240232. |
| [15] |
LEE JH, SEO KH, YANG JH, et al. CCCP induces hepatic stellate cell activation and liver fibrogenesis via mitochondrial and lysosomal dysfunction[J]. Free Radic Biol Med, 2024, 225: 181- 192. DOI: 10.1016/j.freeradbiomed.2024.10.259. |
| [16] |
BAULIES A, RIBAS V, NÚÑEZ S, et al. Lysosomal cholesterol accumulation sensitizes to acetaminophen hepatotoxicity by impairing mitophagy[J]. Sci Rep, 2015, 5: 18017. DOI: 10.1038/srep18017. |
| [17] |
GOU YS, WANG LF, ZHAO JH, et al. PNPLA3-I148M variant promotes the progression of liver fibrosis by inducing mitochondrial dysfunction[J]. Int J Mol Sci, 2023, 24( 11): 9681. DOI: 10.3390/ijms24119681. |
| [18] |
TORRES S, SOLSONA-VILARRASA E, NUÑEZ S, et al. Acid ceramidase improves mitochondrial function and oxidative stress in Niemann-Pick type C disease by repressing STARD1 expression and mitochondrial cholesterol accumulation[J]. Redox Biol, 2021, 45: 102052. DOI: 10.1016/j.redox.2021.102052. |
| [19] |
SANTRA A, BISHNU D, SANTRA S, et al. Arsenic-induced injury of mouse hepatocytes through lysosome and mitochondria: An in vitro study[J]. Int J Hepatol, 2022, 2022: 1546297. DOI: 10.1155/2022/1546297. |
| [20] |
WEI HR, XIAO F, WEI HS. Research progress on the mechanism of endoplasmic reticulum stress in metabolic dysfunction-associated fatty liver disease[J/OL]. Chin J Liver Dis Electron Version, 2024, 16( 1): 13- 17. DOI: 10.3969/j.issn.1674-7380.2024.01.003. |
| [21] |
OKISHIO S, YAMAGUCHI K, ISHIBA H, et al. PPARα agonist and metformin co-treatment ameliorates NASH in mice induced by a choline-deficient, amino acid-defined diet with 45% fat[J]. Sci Rep, 2020, 10( 1): 19578. DOI: 10.1038/s41598-020-75805-z. |
| [22] |
ANTELO-CEA DA, MARTÍNEZ-ROJAS L, CABRERIZO-IBÁÑEZ I, et al. Regulation of mitochondrial and peroxisomal metabolism in female obesity and type 2 diabetes[J]. Int J Mol Sci, 2024, 25( 20): 11237. DOI: 10.3390/ijms252011237. |
| [23] |
HERNÁNDEZ-GEA V, HILSCHER M, ROZENFELD R, et al. Endoplasmic reticulum stress induces fibrogenic activity in hepatic stellate cells through autophagy[J]. J Hepatol, 2013, 59( 1): 98- 104. DOI: 10.1016/j.jhep.2013.02.016. |
| [24] |
HETZ C, ZHANG KZ, KAUFMAN RJ. Mechanisms, regulation and functions of the unfolded protein response[J]. Nat Rev Mol Cell Biol, 2020, 21( 8): 421- 438. DOI: 10.1038/s41580-020-0250-z. |
| [25] |
XUE F, LU JW, BUCHL SC, et al. Coordinated signaling of activating transcription factor 6α and inositol-requiring enzyme 1α regulates hepatic stellate cell-mediated fibrogenesis in mice[J]. Am J Physiol Gastrointest Liver Physiol, 2021, 320( 5): G864- G879. DOI: 10.1152/ajpgi.00453.2020. |
| [26] |
YU HJ, WANG CQ, QIAN BL, et al. GRINA alleviates hepatic ischemia-eperfusion injury-induced apoptosis and ER-phagy by enhancing HRD1-mediated ATF6 ubiquitination[J]. J Hepatol, 2025, 83( 1): 131- 145. DOI: 10.1016/j.jhep.2025.01.012. |
| [27] |
MOHAMMADPOUR-ASL S, ROSHAN-MILANI B, ROSHAN-MILANI S, et al. Endoplasmic reticulum stress PERK-ATF4-CHOP pathway is involved in non-alcoholic fatty liver disease in type 1 diabetic rats: The rescue effect of treatment exercise and insulin-like growth factor I[J]. Heliyon, 2024, 10( 5): e27225. DOI: 10.1016/j.heliyon.2024.e27225. |
| [28] |
KIM HJ, JOE Y, KIM SK, et al. Carbon monoxide protects against hepatic steatosis in mice by inducing sestrin-2 via the PERK-eIF2α-ATF4 pathway[J]. Free Radic Biol Med, 2017, 110: 81- 91. DOI: 10.1016/j.freeradbiomed.2017.05.026. |
| [29] |
UZHYTCHAK M, LUNOVA M, SMOLKOVÁ B, et al. Iron oxide nanoparticles trigger endoplasmic reticulum damage in steatotic hepatic cells[J]. Nanoscale Adv, 2023, 5( 16): 4250- 4268. DOI: 10.1039/d3na00071k. |
| [30] |
MAEDA M, ARAKAWA M, SAITO K. Disease-associated factors at the endoplasmic reticulum-Golgi interface[J]. Traffic, 2025, 26( 1-3): e70001. DOI: 10.1111/tra.70001. |
| [31] |
JI C. Dissecting the role of disturbed ER-Golgi trafficking in antivirals and alcohol abuse-induced pathogenesis of liver disorders[J]. J Drug Abuse, 2017, 3( 3): 14. DOI: 10.21767/2471-853X.100054. |
| [32] |
KHALATBARI A, MISHRA P, HAN H, et al. Ritonavir and lopinavir suppress RCE1 and CAAX rab proteins sensitizing the liver to organelle stress and injury[J]. Hepatol Commun, 2020, 4( 6): 932- 944. DOI: 10.1002/hep4.1515. |
| [33] |
HAN H, HE YX, HU J, et al. Disrupted ER-to-Golgi trafficking underlies anti-HIV drugs and alcohol-induced cellular stress and hepatic injury[J]. Hepatol Commun, 2017, 1( 2): 122- 139. DOI: 10.1002/hep4.1030. |
| [34] |
KE CL, XIAO CC, LI JM, et al. FMO2 ameliorates nonalcoholic fatty liver disease by suppressing ER-to-Golgi transport of SREBP1[J]. Hepatology, 2025, 81( 1): 181- 197. DOI: 10.1097/HEP.0000000000000643. |
| [35] |
MAST FD, RACHUBINSKI RA, AITCHISON JD. Peroxisome prognostications: Exploring the birth, life, and death of an organelle[J]. J Cell Biol, 2020, 219( 3): e201912100. DOI: 10.1083/jcb.201912100. |
| [36] |
ILACQUA N, ANASTASIA I, RAIMONDI A, et al. A three-organelle complex made by wrappER contacts with peroxisomes and mitochondria responds to liver lipid flux changes[J]. J Cell Sci, 2022, 135( 5): jcs259091. DOI: 10.1242/jcs.259091. |
| [37] |
CHU BB, LIAO YC, QI W, et al. Cholesterol transport through lysosome-peroxisome membrane contacts[J]. Cell, 2015, 161( 2): 291- 306. DOI: 10.1016/j.cell.2015.02.019. |
| [38] |
WANG YH, TWU YC, WANG CK, et al. Niemann-pick type C2 protein regulates free cholesterol accumulation and influences hepatic stellate cell proliferation and mitochondrial respiration function[J]. Int J Mol Sci, 2018, 19( 6): 1678. DOI: 10.3390/ijms19061678. |
| [39] |
HE AY, CHEN XW, TAN M, et al. Acetyl-CoA derived from hepatic peroxisomal β-oxidation inhibits autophagy and promotes steatosis via mTORC1 activation[J]. Mol Cell, 2020, 79( 1): 30- 42.e4. DOI: 10.1016/j.molcel.2020.05.007. |
| [40] |
HOU XX, LI YW, SONG JL, et al. Cryptotanshinone induces apoptosis of activated hepatic stellate cells via modulating endoplasmic reticulum stress[J]. World J Gastroenterol, 2023, 29( 17): 2616- 2627. DOI: 10.3748/wjg.v29.i17.2616. |
| [41] |
MEI GB. Quercetin ameliorates metabolic-associated fatty liver disease in type 2 diabetes mellitus and the mediated mechanisms of MAMs-NLRP3[D]. Wuhan: Huazhong University of Science and Technology, 2021. DOI: 10.27157/d.cnki. ghzku.2021.004842.
梅桂斌. 槲皮素缓解T2DM代谢相关脂肪性肝损伤及MAMs-NLRP3的介导机制[D]. 武汉: 华中科技大学, 2021. DOI: 10.27157/d.cnki. ghzku.2021.004842.
|
| [42] |
GAO ZS. Study on the protective effects and mechanisms of salidroside in drug-induced liver injury[D]. Chongqing: Southwest University, 2024. DOI: 10.27684/d.cnki.gxndx.2024.000062. 高正山. 红景天苷对药物性肝损伤的保护作用及机制研究[D]. 重庆: 西南大学, 2024. DOI: 10.27684/d.cnki. gxndx.2024.000062.
|
| [43] |
MEIRA MARTINS LA, VIEIRA MQ, ILHA M, et al. The interplay between apoptosis, mitophagy and mitochondrial biogenesis induced by resveratrol can determine activated hepatic stellate cells death or survival[J]. Cell Biochem Biophys, 2015, 71( 2): 657- 672. DOI: 10.1007/s12013-014-0245-5. |
| [44] |
LEE AY, JANG Y, HONG SH, et al. Ephedrine-induced mitophagy via oxidative stress in human hepatic stellate cells[J]. J Toxicol Sci, 2017, 42( 4): 461- 473. DOI: 10.2131/jts.42.461. |
| [45] |
MENG ZQ. Study on the mechanism of apigenin alleviates liver injury induced by high fat diet through activating[D]. Changchun: Jilin University, 2023. DOI: 10.27162/d.cnki.gjlin.2023.007517. 孟卓群. 芹菜素激活自噬缓解高脂饮食所致肝损伤的作用机制研究[D]. 长春: 吉林大学, 2023. DOI: 10.27162/ d.cnki.gjlin.2023.007517.
|
| [46] |
DUSABIMANA T, KIM SR, KIM HJ, et al. Nobiletin ameliorates hepatic ischemia and reperfusion injury through the activation of SIRT-1/FOXO3a-mediated autophagy and mitochondrial biogenesis[J]. Exp Mol Med, 2019, 51( 4): 51. DOI: 10.1038/s12276-019-0245-z. |
| [47] |
XIAO HY, WANG L, WANG JH, et al. Exploring the role of curcumol on mitochondrial autophagy in hepatic stellate cells based on the PINK1/Parkin signalling pathway[J]. Acta Univ Med Anhui, 2025, 60( 5): 919- 928. DOI: 10.19405/j.cnki.issn1000-1492.2025.05.020. |
| [48] |
DENG ZW, REN CX, TANG CL, et al. Syringin alleviates hepatic fibrosis by enhancing autophagic flux and attenuating ER stress-TRIB3/SMAD3 in diabetic mice[J]. Tissue Cell, 2023, 83: 102159. DOI: 10.1016/j.tice.2023.102159. |
| [49] |
SUN YF, PAN HH, XIA ZN, et al. Alisma Shugan Decoction attenuates hepatic fibrosis and endoplasmic reticulum stress in mice with carbon tetrachloride-induced fibrosis[J]. Afr Health Sci, 2023, 23( 2): 422- 434. DOI: 10.4314/ahs.v23i2.49. |
| [50] |
GAO XB, GUO S, ZHANG S, et al. Matrine attenuates endoplasmic reticulum stress and mitochondrion dysfunction in nonalcoholic fatty liver disease by regulating SERCA pathway[J]. J Transl Med, 2018, 16( 1): 319. DOI: 10.1186/s12967-018-1685-2. |



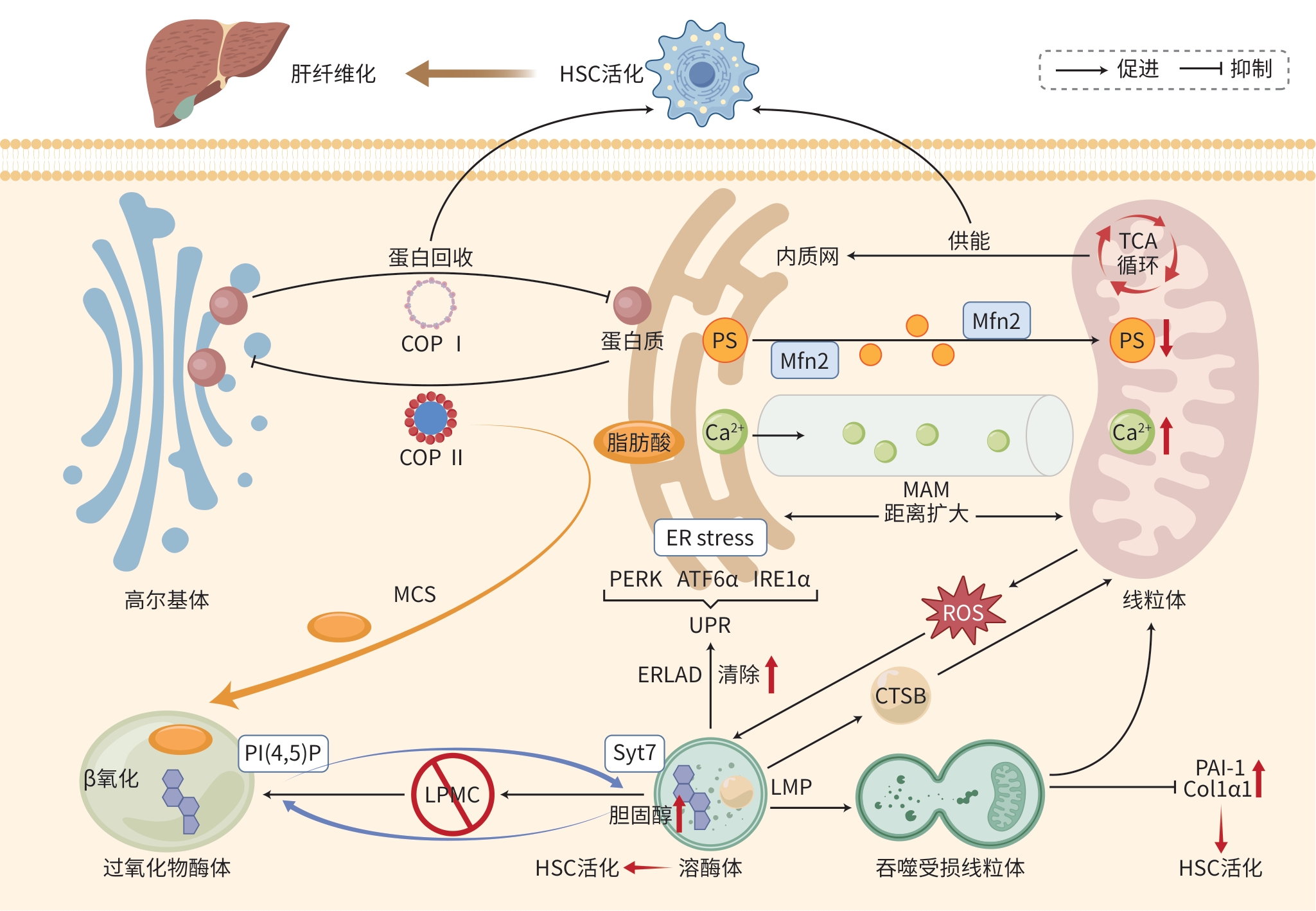




 DownLoad:
DownLoad: