| [1] |
YANG X, LI Q, LIU WT, et al. Mesenchymal stromal cells in hepatic fibrosis/cirrhosis: From pathogenesis to treatment[J]. Cell Mol Immunol, 2023, 20( 6): 583- 599. DOI: 10.1038/s41423-023-00983-5. |
| [2] |
LIAO ZH, XIE ZY. Research progress in molecular mechanism of hepatic fibrosis and related therapeutic targets[J]. J Jilin Univ Med Ed, 2024, 50( 5): 1450- 1456. DOI: 10.13481/j.1671-587X.20240532. |
| [3] |
ZHONG L, ZHAO JQ, HUANG L, et al. Runx2 activates hepatic stellate cells to promote liver fibrosis via transcriptionally regulating Itgav expression[J]. Clin Transl Med, 2023, 13( 7): e1316. DOI: 10.1002/ctm2.1316. |
| [4] |
TSUCHIDA T, FRIEDMAN SL. Mechanisms of hepatic stellate cell activation[J]. Nat Rev Gastroenterol Hepatol, 2017, 14( 7): 397- 411. DOI: 10.1038/nrgastro.2017.38. |
| [5] |
LV XY, LV YY, DAI XF. Lactate, histone lactylation and cancer hallmarks[J]. Expert Rev Mol Med, 2023, 25: e7. DOI: 10.1017/erm.2022.42. |
| [6] |
KOPPENOL WH, BOUNDS PL, DANG CV. Otto Warburg’s contributions to current concepts of cancer metabolism[J]. Nat Rev Cancer, 2011, 11( 5): 325- 337. DOI: 10.1038/nrc3038. |
| [7] |
GAO X, PANG CY, FAN ZY, et al. Regulation of newly identified lysine lactylation in cancer[J]. Cancer Lett, 2024, 587: 216680. DOI: 10.1016/j.canlet.2024.216680. |
| [8] |
ZHU RX, YE XL, LU XT, et al. ACSS2 acts as a lactyl-CoA synthetase and couples KAT2A to function as a lactyltransferase for histone lactylation and tumor immune evasion[J]. Cell Metab, 2025, 37( 2): 361- 376. DOI: 10.1016/j.cmet.2024.10.015. |
| [9] |
LEGUBE G, TROUCHE D. Regulating histone acetyltransferases and deacetylases[J]. EMBO Rep, 2003, 4( 10): 944- 947. DOI: 10.1038/sj.embor.embor941. |
| [10] |
MORENO-YRUELA C, ZHANG D, WEI W, et al. Class I histone deacetylases(HDAC1-3) are histone lysine delactylases[J]. Sci Adv, 2022, 8( 3): eabi6696. DOI: 10.1126/sciadv.abi6696. |
| [11] |
KADOCH C, CRABTREE GR. Mammalian SWI/SNF chromatin remodeling complexes and cancer: Mechanistic insights gained from human genomics[J]. Sci Adv, 2015, 1( 5): e1500447. DOI: 10.1126/sciadv.1500447. |
| [12] |
HU XL, HUANG XW, YANG Y, et al. Dux activates metabolism-lactylation-MET network during early iPSC reprogramming with Brg1 as the histone lactylation reader[J]. Nucleic Acids Res, 2024, 52( 10): 5529- 5548. DOI: 10.1093/nar/gkae183. |
| [13] |
ZHAI GJ, NIU ZP, JIANG ZX, et al. DPF2 reads histone lactylation to drive transcription and tumorigenesis[J]. Proc Natl Acad Sci USA, 2024, 121( 50): e2421496121. DOI: 10.1073/pnas.2421496121. |
| [14] |
NUÑEZ R, SIDLOWSKI PFW, STEEN EA, et al. The TRIM33 bromodomain recognizes histone lysine lactylation[J]. ACS Chem Biol, 2024, 19( 12): 2418- 2428. DOI: 10.1021/acschembio.4c00248. |
| [15] |
LIU RL, REN XL, PARK YE, et al. Nuclear GTPSCS functions as a lactyl-CoA synthetase to promote histone lactylation and gliomagenesis[J]. Cell Metab, 2025, 37( 2): 377- 394. DOI: 10.1016/j.cmet.2024.11.005. |
| [16] |
GALLE E, WONG CW, GHOSH A, et al. H3K18 lactylation marks tissue-specific active enhancers[J]. Genome Biol, 2022, 23( 1): 207. DOI: 10.1186/s13059-022-02775-y. |
| [17] |
CERTO M, TSAI CH, PUCINO V, et al. Lactate modulation of immune responses in inflammatory versus tumour microenvironments[J]. Nat Rev Immunol, 2021, 21( 3): 151- 161. DOI: 10.1038/s41577-020-0406-2. |
| [18] |
HANAHAN D, WEINBERG RA. Hallmarks of cancer: The next generation[J]. Cell, 2011, 144( 5): 646- 674. DOI: 10.1016/j.cell.2011.02.013. |
| [19] |
MURRAY PJ, WYNN TA. Protective and pathogenic functions of macrophage subsets[J]. Nat Rev Immunol, 2011, 11( 11): 723- 737. DOI: 10.1038/nri3073. |
| [20] |
WYNN TA, CHAWLA A, POLLARD JW. Macrophage biology in development, homeostasis and disease[J]. Nature, 2013, 496( 7446): 445- 455. DOI: 10.1038/nature12034. |
| [21] |
IRIZARRY-CARO RA, MCDANIEL MM, OVERCAST GR, et al. TLR signaling adapter BCAP regulates inflammatory to reparatory macrophage transition by promoting histone lactylation[J]. Proc Natl Acad Sci USA, 2020, 117( 48): 30628- 30638. DOI: 10.1073/pnas.2009778117. |
| [22] |
DICHTL S, LINDENTHAL L, ZEITLER L, et al. Lactate and IL6 define separable paths of inflammatory metabolic adaptation[J]. Sci Adv, 2021, 7( 26): eabg3505. DOI: 10.1126/sciadv.abg3505. |
| [23] |
ZHANG D, TANG ZY, HUANG H, et al. Metabolic regulation of gene expression by histone lactylation[J]. Nature, 2019, 574( 7779): 575- 580. DOI: 10.1038/s41586-019-1678-1. |
| [24] |
MA W, AO SX, ZHOU JP, et al. Methylsulfonylmethane protects against lethal dose MRSA-induced sepsis through promoting M2 macrophage polarization[J]. Mol Immunol, 2022, 146: 69- 77. DOI: 10.1016/j.molimm.2022.04.001. |
| [25] |
GU J, ZHOU JR, CHEN QY, et al. Tumor metabolite lactate promotes tumorigenesis by modulating MOESIN lactylation and enhancing TGF-β signaling in regulatory T cells[J]. Cell Rep, 2022, 39( 12): 110986. DOI: 10.1016/j.celrep.2022.110986. |
| [26] |
LI LN, LI WW, XIAO LS, et al. Lactylation signature identifies liver fibrosis phenotypes and traces fibrotic progression to hepatocellular carcinoma[J]. Front Immunol, 2024, 15: 1433393. DOI: 10.3389/fimmu.2024.1433393. |
| [27] |
ZHAO TT, LI JF, ZHANG LT. Progress in the potential therapeutic mechanism of mesenchymal stem cell-derived exosomes for liver fibrosis[J]. Chin J Clin Pharmacol Ther, 2024, 29( 4): 475- 480. DOI: 10.12092/j.issn.1009-2501.2024.04.017. |
| [28] |
TRIVEDI P, WANG S, FRIEDMAN SL. The power of plasticity-metabolic regulation of hepatic stellate cells[J]. Cell Metab, 2021, 33( 2): 242- 257. DOI: 10.1016/j.cmet.2020.10.026. |
| [29] |
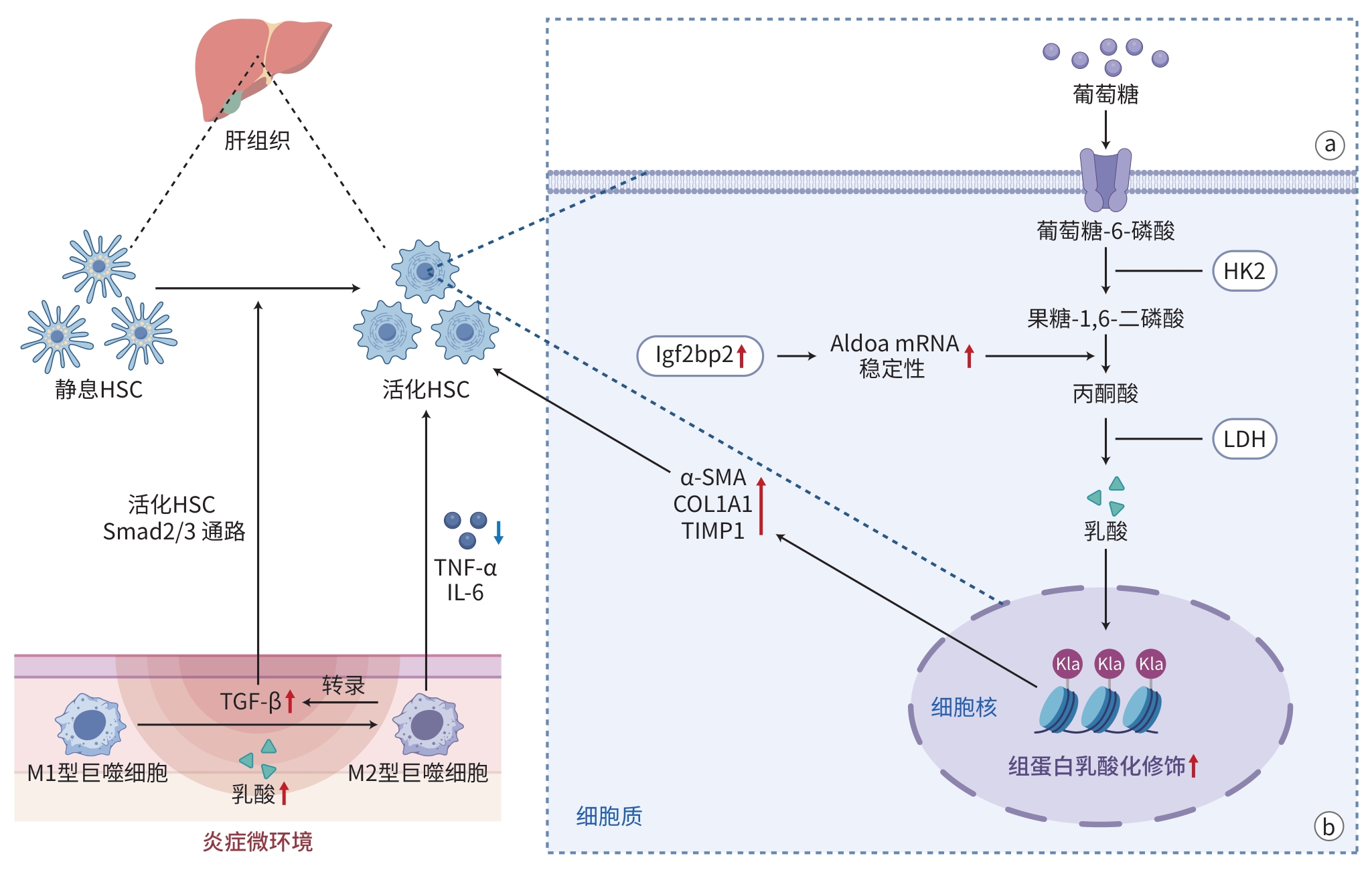
RHO H, TERRY AR, CHRONIS C, et al. Hexokinase 2-mediated gene expression via histone lactylation is required for hepatic stellate cell activation and liver fibrosis[J]. Cell Metab, 2023, 35( 8): 1406- 1423. DOI: 10.1016/j.cmet.2023.06.013. |
| [30] |
ZHOU YQ, YAN JX, HUANG H, et al. The m 6A reader IGF2BP2 regulates glycolytic metabolism and mediates histone lactylation to enhance hepatic stellate cell activation and liver fibrosis[J]. Cell Death Dis, 2024, 15( 3): 189. DOI: 10.1038/s41419-024-06509-9. |
| [31] |
WU SJ, LI JH, ZHAN YF. H3K18 lactylation accelerates liver fibrosis progression through facilitating SOX9 transcription[J]. Exp Cell Res, 2024, 440( 2): 114135. DOI: 10.1016/j.yexcr.2024.114135. |
| [32] |
TROGISCH FA, ABOUISSA A, KELES M, et al. Endothelial cells drive organ fibrosis in mice by inducing expression of the transcription factor SOX9[J]. Sci Transl Med, 2024, 16( 736): eabq4581. DOI: 10.1126/scitranslmed.abq4581. |
| [33] |
YU YF, LI YN, ZHOU L, et al. Hepatic stellate cells promote hepatocellular carcinoma development by regulating histone lactylation: Novel insights from single-cell RNA sequencing and spatial transcriptomics analyses[J]. Cancer Lett, 2024, 604: 217243. DOI: 10.1016/j.canlet.2024.217243. |
| [34] |
XIE N, TAN Z, BANERJEE S, et al. Glycolytic reprogramming in myofibroblast differentiation and lung fibrosis[J]. Am J Respir Crit Care Med, 2015, 192( 12): 1462- 1474. DOI: 10.1164/rccm.201504-0780OC. |
| [35] |
DEWIDAR B, MEYER C, DOOLEY S, et al. TGF-β in hepatic stellate cell activation and liver fibrogenesis-updated 2019[J]. Cells, 2019, 8( 11): 1419. DOI: 10.3390/cells8111419. |
| [36] |
GAO R, LI Y, XU Z, et al. Mitochondrial pyruvate carrier 1 regulates fatty acid synthase lactylation and mediates treatment of nonalcoholic fatty liver disease[J]. Hepatology, 2023, 78( 6): 1800- 1815. DOI: 10.1097/HEP.0000000000000279. |
| [37] |
PAN RY, HE L, ZHANG J, et al. Positive feedback regulation of microglial glucose metabolism by histone H4 lysine 12 lactylation in Alzheimer’s disease[J]. Cell Metab, 2022, 34( 4): 634- 648 DOI: 10.1016/j.cmet.2022.02.013. |
| [38] |
WANG PW, LIN TY, YANG PM, et al. Hepatic stellate cell modulates the immune microenvironment in the progression of hepatocellular carcinoma[J]. Int J Mol Sci, 2022, 23( 18): 10777. DOI: 10.3390/ijms231810777. |
| [39] |
HUANG JL, FU YP, GAN W, et al. Hepatic stellate cells promote the progression of hepatocellular carcinoma through microRNA-1246-RORα-Wnt/β-Catenin axis[J]. Cancer Lett, 2020, 476: 140- 151. DOI: 10.1016/j.canlet.2020.02.012. |
| [40] |
SUN C, XU WW, XIA YH, et al. PRDM16 from hepatic stellate cells-derived extracellular vesicles promotes hepatocellular carcinoma progression[J]. Am J Cancer Res, 2023, 13( 11): 5254- 5270.
|
| [41] |
YANG ZJ, YAN C, MA JQ, et al. Lactylome analysis suggests lactylation-dependent mechanisms of metabolic adaptation in hepatocellular carcinoma[J]. Nat Metab, 2023, 5( 1): 61- 79. DOI: 10.1038/s42255-022-00710-w. |
| [42] |
WU QJ, LI X, LONG MH, et al. Integrated analysis of histone lysine lactylation(Kla)-specific genes suggests that NR6A1 OSBP2 and UNC119B are novel therapeutic targets for hepatocellular carcinoma[J]. Sci Rep, 2023, 13( 1): 18642. DOI: 10.1038/s41598-023-46057-4. |
| [43] |
PAN LH, FENG F, WU JQ, et al. Demethylzeylasteral targets lactate by inhibiting histone lactylation to suppress the tumorigenicity of liver cancer stem cells[J]. Pharmacol Res, 2022, 181: 106270. DOI: 10.1016/j.phrs.2022.106270. |
| [44] |
JIN J, BAI L, WANG DY, et al. SIRT3-dependent delactylation of cyclin E2 prevents hepatocellular carcinoma growth[J]. EMBO Rep, 2023, 24( 5): e56052. DOI: 10.15252/embr.202256052. |







 DownLoad:
DownLoad: