| [1] |
RINELLA ME, LAZARUS JV, RATZIU V, et al. A multisociety Delphi consensus statement on new fatty liver disease nomenclature[J]. Hepatology, 2023, 78( 6): 1966- 1986. DOI: 10.1097/HEP.0000000000000520. |
| [2] |
FAN JG, XU XY, YANG RX, et al. Guideline for the prevention and treatment of metabolic dysfunction-associated fatty liver disease(version 2024)[J]. J Clin Transl Hepatol, 2024, 12( 11): 955- 974. DOI: 10.14218/jcth.2024.00311 |
| [3] |
MOHAMED ABDELGAWWAD EL-SEHRAWY A ALI, MOHAMMED AN, GUPTA J, et al. Combating oxidative stress in non-alcoholic fatty liver disease: From mechanisms to therapeutic strategies[J]. Pathol Res Pract, 2025, 272: 156053. DOI: 10.1016/j.prp.2025.156053. |
| [4] |
MARTÍN-MATEOS R, ALBILLOS A. The role of the gut-liver axis in metabolic dysfunction-associated fatty liver disease[J]. Front Immunol, 2021, 12: 660179. DOI: 10.3389/fimmu.2021.660179. |
| [5] |
VALLIANOU N, CHRISTODOULATOS GS, KARAMPELA I, et al. Understanding the role of the gut microbiome and microbial metabolites in non-alcoholic fatty liver disease: Current evidence and perspectives[J]. Biomolecules, 2021, 12( 1): 56. DOI: 10.3390/biom-12010056. |
| [6] |
RAMOS-TOVAR E, MURIEL P. NLRP3 inflammasome in hepatic diseases: A pharmacological target[J]. Biochem Pharmacol, 2023, 217: 115861. DOI: 10.1016/j.bcp.2023.115861. |
| [7] |
QIN WW, WENG JP. Hepatocyte NLRP3 interacts with PKCε to drive hepatic insulin resistance and steatosis[J]. Sci Bull, 2023, 68( 13): 1413- 1429. DOI: 10.1016/j.scib.2023.06.003. |
| [8] |
YUAN XX, BHAT OM, ZOU Y, et al. Contribution of hepatic steatosis-intensified extracellular vesicle release to aggravated inflammatory endothelial injury in liver-specific Asah1 gene knockout mice[J]. Am J Pathol, 2023, 193( 4): 493- 508. DOI: 10.1016/j.ajpath.2022.12.007. |
| [9] |
de DEUS IJ, MARTINS-SILVA AF, de ANDRADE FAGUNDES MM, et al. Role of NLRP3 inflammasome and oxidative stress in hepatic insulin resistance and the ameliorative effect of phytochemical intervention[J]. Front Pharmacol, 2023, 14: 1188829. DOI: 10.3389/fphar.2023.1188829. |
| [10] |
YU TT, LUO L, XUE J, et al. Gut microbiota-NLRP3 inflammasome crosstalk in metabolic dysfunction-associated steatotic liver disease[J]. Clin Res Hepatol Gastroenterol, 2024, 48( 8): 102458. DOI: 10.1016/j.clinre.2024.102458. |
| [11] |
INIA JA, STOKMAN G, PIETERMAN EJ, et al. Atorvastatin attenuates diet-induced non-alcoholic steatohepatitis in APOE*3-leiden mice by reducing hepatic inflammation[J]. Int J Mol Sci, 2023, 24( 9): 7818. DOI: 10.3390/ijms24097818. |
| [12] |
ZHANG X, COKER OO, CHU ES, et al. Dietary cholesterol drives fatty liver-associated liver cancer by modulating gut microbiota and metabolites[J]. Gut, 2021, 70( 4): 761- 774. DOI: 10.1136/gutjnl-2019-319664. |
| [13] |
TAKAGURI A. Elucidation of a new mechanism of onset of insulin resistance: Effects of statins and tumor necrosis factor-α on insulin signal transduction[J]. Yakugaku Zasshi, 2018, 138( 11): 1329- 1334. DOI: 10.1248/yakushi.18-00116. |
| [14] |
CLIMENT E, BENAIGES D, PEDRO-BOTET J. Hydrophilic or lipophilic statins[J]. Front Cardiovasc Med, 2021, 8: 687585. DOI: 10.3389/fcvm.2021.687585. |
| [15] |
SIZAR O, KHARE S, PATEL P, TALATI R. Statin Medications[M]. StatPearls. Treasure Island(FL): StatPearls Publishing, 2024.
|
| [16] |
FOSTER T, BUDOFF MJ, SAAB S, et al. Atorvastatin and antioxidants for the treatment of nonalcoholic fatty liver disease: The St Francis Heart Study randomized clinical trial[J]. Am J Gastroenterol, 2011, 106( 1): 71- 77. DOI: 10.1038/ajg.2010.299. |
| [17] |
HADZI-PETRUSHEV N, DIMOVSKA K, JANKULOVSKI N, et al. Supplementation with alpha-tocopherol and ascorbic acid to nonalcoholic fatty liver disease’s statin therapy in men[J]. Adv Pharmacol Sci, 2018, 2018: 4673061. DOI: 10.1155/2018/4673061. |
| [18] |
WANG X, LYU L, LI W, et al. Impact of rosuvastatin on metabolic syndrome patients with moderate to severe metabolic associated fatty liver disease without overt diabetes: A randomized clinical trial[J]. Diabetes Metab Syndr, 2024, 18( 9): 103126. DOI: 10.1016/j.dsx.2024.103126. |
| [19] |
SFIKAS G, PSALLAS M, KOUMARAS C, et al. Prevalence, diagnosis, and treatment with 3 different statins of non-alcoholic fatty liver disease/non-alcoholic steatohepatitis in military personnel. Do genetics play a role?[J]. Curr Vasc Pharmacol, 2021, 19( 5): 572- 581. DOI: 10.2174/1570161118666201015152921. |
| [20] |
FATIMA K, MOEED A, WAQAR E, et al. Efficacy of statins in treatment and development of non-alcoholic fatty liver disease and steatohepatitis: A systematic review and meta-analysis[J]. Clin Res Hepatol Gastroenterol, 2022, 46( 4): 101816. DOI: 10.1016/j.clinre.2021.101816. |
| [21] |
ABDALLAH M, BROWN L, PROVENZA J, et al. Safety and efficacy of dyslipidemia treatment in NAFLD patients: A meta-analysis of randomized controlled trials[J]. Ann Hepatol, 2022, 27( 6): 100738. DOI: 10.1016/j.aohep.2022.100738. |
| [22] |
DENG YF, XU QQ, CHEN TQ, et al. Kinsenoside alleviates inflammation and fibrosis in experimental NASH mice by suppressing the NF-κB/NLRP3 signaling pathway[J]. Phytomedicine, 2022, 104: 154241. DOI: 10.1016/j.phymed.2022.154241. |
| [23] |
LIN HZ, QI JW, LIN SW, et al. Inhibition of cholesterol synthesis in T cells protects the liver against steatosis, inflammation, and oxidative stress in metabolic dysfunction-associated steatohepatitis[J]. FASEB J, 2025, 39( 11): e70665. DOI: 10.1096/fj.202500115R. |
| [24] |
MCGLONE ER, ANSELL TB, DUNSTERVILLE C, et al. Hepatocyte cholesterol content modulates glucagon receptor signalling[J]. Mol Metab, 2022, 63: 101530. DOI: 10.1016/j.molmet.2022.101530. |
| [25] |
da SILVA PEREIRA ENG, de ARAUJO BP, RODRIGUES KL, et al. Simvastatin improves microcirculatory function in nonalcoholic fatty liver disease and downregulates oxidative and ALE-RAGE stress[J]. Nutrients, 2022, 14( 3): 716. DOI: 10.3390/nu14030716. |
| [26] |
LASTUVKOVA H, FARADONBEH FA, SCHREIBEROVA J, et al. Atorvastatin modulates bile acid homeostasis in mice with diet-induced nonalcoholic steatohepatitis[J]. Int J Mol Sci, 2021, 22( 12): 6468. DOI: 10.3390/ijms22126468. |
| [27] |
ESLAMI Z, MOGHANLOU AE, KANDI YMNP, et al. Atorvastatin and flaxseed effects on biochemical indices and hepatic fat of NAFLD model in rats[J]. Adv Biomed Res, 2023, 12: 98. DOI: 10.4103/abr.abr_21_22. |
| [28] |
ESLAMI Z, JOSHAGHANI H, EGHBAL MOGHANLOU A, et al. Atorvastatin and flaxseed dietary treatments improve dyslipidemia and liver injuries in a diet-induced rat model of non-alcoholic fatty liver disease[J]. Avicenna J Phytomed, 2025, 15( 3): 1102- 1112. DOI: 10.22038/ajp.2024.25220. |
| [29] |
EGHDAMI S, AFRASHTEH F, SHOJAII A, et al. The therapeutic effect of alcoholic extract of Fumaria parviflora on high-fat diet-induced nonalcoholic fatty liver in rats: An animal experiment[J]. Ann Med Surg, 2024, 86( 5): 2657- 2664. DOI: 10.1097/MS9.0000000000001890. |
| [30] |
AYADA I, VAN KLEEF LA, ZHANG H, et al. Dissecting the multifaceted impact of statin use on fatty liver disease: A multidimensional study[J]. EBioMedicine, 2023, 87: 104392. DOI: 10.1016/j.ebiom.2022.104392. |
| [31] |
HO A, KIENER T, NGUYEN QN, et al. Effect of statin use on liver enzymes and lipid profile in patients with non-alcoholic fatty liver disease(NAFLD)[J]. J Clin Lipidol, 2024, 18( 4): e501- e508. DOI: 10.1016/j.jacl.2024.03.003. |
| [32] |
THOMSON MJ, SERPER M, KHUNGAR V, et al. Prevalence and factors associated with statin use among patients with nonalcoholic fatty liver disease in the TARGET-NASH study[J]. Clin Gastroenterol Hepatol, 2022, 20( 2): 458- 460. e 4. DOI: 10.1016/j.cgh.2021.03.031. |
| [33] |
NAKAGAWA C, YOKOYAMA S, HOSOMI K. Association of statin adherence with the development of nonalcoholic fatty liver disease: A nested case-control study using a Japanese claims database[J]. Ann Pharmacother, 2023, 57( 6): 637- 645. DOI: 10.1177/10600280221126971. |
| [34] |
SCHREINER AD, ZHANG JW, PETZ CA, et al. Statin prescriptions and progression of advanced fibrosis risk in primary care patients with MAFLD[J]. BMJ Open Gastroenterol, 2024, 11( 1): e001404. DOI: 10.1136/bmjgast-2024-001404. |
| [35] |
ZHOU XD, KIM SU, YIP TC, et al. Long-term liver-related outcomes and liver stiffness progression of statin usage in steatotic liver disease[J]. Gut, 2024, 73( 11): 1883- 1892. DOI: 10.1136/gutjnl-2024-333074. |
| [36] |
TARAR ZI, FAROOQ U, INAYAT F, et al. Statins decrease the risk of hepatocellular carcinoma in metabolic dysfunction-associated steatotic liver disease: A systematic review and meta-analysis[J]. World J Exp Med, 2024, 14( 4): 98543. DOI: 10.5493/wjem.v14.i4.98543. |
| [37] |
ISLAM MM, POLY TN, WALTHER BA, et al. Statin use and the risk of hepatocellular carcinoma: A meta-analysis of observational studies[J]. Cancers, 2020, 12( 3): 671. DOI: 10.3390/cancers12030671. |
| [38] |
ZHANG J, FU SF, LIU DL, et al. Statin can reduce the risk of hepatocellular carcinoma among patients with nonalcoholic fatty liver disease: A systematic review and meta-analysis[J]. Eur J Gastroenterol Hepatol, 2023, 35( 4): 353- 358. DOI: 10.1097/MEG.0000000000002517. |
| [39] |
TOMAH S, ALKHOURI N, HAMDY O. Nonalcoholic fatty liver disease and type 2 diabetes: Where do Diabetologists stand?[J]. Clin Diabetes Endocrinol, 2020, 6: 9. DOI: 10.1186/s40842-020-00097-1. |
| [40] |
NASCIMBENI F, ARON-WISNEWSKY J, PAIS R, et al. Statins, antidiabetic medications and liver histology in patients with diabetes with non-alcoholic fatty liver disease[J]. BMJ Open Gastroenterol, 2016, 3( 1): e000075. DOI: 10.1136/bmjgast-2015-000075. |
| [41] |
BRIL F, PORTILLO SANCHEZ P, LOMONACO R, et al. Liver safety of statins in prediabetes or T2DM and nonalcoholic steatohepatitis: Post hoc analysis of a randomized trial[J]. J Clin Endocrinol Metab, 2017, 102( 8): 2950- 2961. DOI: 10.1210/jc.2017-00867. |
| [42] |
YU HZ, ZHANG SD, LI JC, et al. Effects of statins on progressive liver fibrosis,liver cirrhosis and metabolic associated fatty liver disease in patients with type 2 diabetes[J/CD]. Chin J Liver Dis(Electronic Version), 2025, 17( 1): 35- 43. DOI: 10.3969/j.issn.1674-7380.2025.01.006. |
| [43] |
WU SY, CHEN WM, CHIANG MF, et al. Protective effects of statins on the incidence of NAFLD-related decompensated cirrhosis in T2DM[J]. Liver Int, 2023, 43( 10): 2232- 2244. DOI: 10.1111/liv.15656. |
| [44] |
ATHYROS VG, POLYZOS SA, KOUNTOURAS J, et al. Non-alcoholic fatty liver disease treatment in patients with type 2 diabetes mellitus; new kids on the block[J]. Curr Vasc Pharmacol, 2020, 18( 2): 172- 181. DOI: 10.2174/1570161117666190405164313. |
| [45] |
CIARDULLO S, PERSEGHIN G. Statin use is associated with lower prevalence of advanced liver fibrosis in patients with type 2 diabetes[J]. Metabolism, 2021, 121: 154752. DOI: 10.1016/j.metabol.2021.154752. |
| [46] |
ZISIS M, CHONDROGIANNI ME, ANDROUTSAKOS T, et al. Linking cardiovascular disease and metabolic dysfunction-associated steatotic liver disease(MAFLD): The role of cardiometabolic drugs in MAFLD treatment[J]. Biomolecules, 2025, 15( 3): 324. DOI: 10.3390/biom15030324. |
| [47] |
SAFAROVA MS, WEINTRAUB S, SADANIANTZ K, et al. Statin use in special populations for the prevention of cardiovascular disease in adults[J]. Curr Atheroscler Rep, 2025, 27( 1): 54. DOI: 10.1007/s11883-025-01298-8. |
| [48] |
GRUNDY SM, STONE NJ, BAILEY AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/ APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: A report of the American college of cardiology/American heart association task force on clinical practice guidelines[J]. J Am Coll Cardiol, 2019, 73( 24): e285- e350. DOI: 10.1016/j.jacc.2018.11.003. |
| [49] |
YOO J, JEON J, BAIK M, et al. Effect of statins for primary prevention of cardiovascular disease according to the fatty liver index[J]. J Epidemiol Glob Health, 2024, 14( 3): 710- 719. DOI: 10.1007/s44197-024-00205-9. |
| [50] |
SÁNCHEZ-POLO MT, CASTELLS MT, GARCÍA-PÉREZ B, et al. Effect of diet/atorvastatin on atherosclerotic lesions associated to nonalcoholic fatty liver disease in chickens[J]. Histol Histopathol, 2015, 30( 12): 1439- 1446. DOI: 10.14670/HH-11-639. |
| [51] |
CAI T, ABEL L, LANGFORD O, et al. Associations between statins and adverse events in primary prevention of cardiovascular disease: Systematic review with pairwise, network, and dose-response meta-analyses[J]. BMJ, 2021, 374: n1537. DOI: 10.1136/bmj.n1537. |
| [52] |
ZHANG HB, PLUTZKY J, SHUBINA M, et al. Continued statin prescriptions after adverse reactions and patient outcomes: A cohort study[J]. Ann Intern Med, 2017, 167( 4): 221- 227. DOI: 10.7326/M16-0838. |
| [53] |
SARKAR M, KUSHNER T. Metabolic dysfunction-associated steatotic liver disease and pregnancy[J]. J Clin Invest, 2025, 135( 10): e186426. DOI: 10.1172/JCI186426. |
| [54] |
LINS SERAFIM J, LUCAS SANTOS DE MENEZES TELES P, SOUZA LIMA AK, et al. Clinical repercussions of statin use during pregnancy: A review of the literature[J]. Rev Bras Ginecol Obstet, 2025, 47: e-rbgo 2. DOI: 10.61622/rbgo/2025rbgo2. |
| [55] |
CHANG JC, CHEN YJ, CHEN IC, et al. Perinatal outcomes after statin exposure during pregnancy[J]. JAMA Netw Open, 2021, 4( 12): e214‑ 1321. DOI: 10.1001/jamanetworkopen.2021.41321. |
| [56] |
ZHANG Y, BU YF, ZHAO R, et al. Metabolic-associated fatty liver disease and pregnancy complications: New challenges and clinical perspectives[J]. Ther Adv Endocrinol Metab, 2024, 15: 204201882412743‑ 50. DOI: 10.1177/20420188241274350. |
| [57] |
HUANG SY, HU WT, FANG TY. The global impact of non-alcoholic fatty liver disease(including cirrhosis) in the elderly from 1990 to 2021 and future projections of disease burden[J]. PLoS One, 2025, 20( 6): e0325961. DOI: 10.1371/journal.pone.0325961. |
| [58] |
GUO MZ, ZHAO JL, ZHAI YJ, et al. A prospective study of hepatic safety of statins used in very elderly patients[J]. BMC Geriatr, 2019, 19( 1): 352. DOI: 10.1186/s12877-019-1361-2. |
| [59] |
XU WC, YAU YK, PAN YY, et al. Effectiveness and safety of using statin therapy for the primary prevention of cardiovascular diseases in older patients with chronic kidney disease who are hypercholesterolemic: A target trial emulation study[J]. Lancet Healthy Longev, 2025, 6( 3): 100683. DOI: 10.1016/j.lanhl.2025.100683. |
| [60] |
PASTORI D, PANI A, DI ROCCO A, et al. Statin liver safety in non-alcoholic fatty liver disease: A systematic review and metanalysis[J]. Br J Clin Pharmacol, 2022, 88( 2): 441- 451. DOI: 10.1111/bcp.14943. |
| [61] |
|
| [62] |
NEWMAN CB. Safety of statins and nonstatins for treatment of dyslipidemia[J]. Endocrinol Metab Clin North Am, 2022, 51( 3): 655- 679. DOI: 10.1016/j.ecl.2022.01.004. |
| [63] |
|
| [64] |
MORRIS R, BU K, HAN WR, et al. The association between statin drugs and rhabdomyolysis: An analysis of FDA adverse event reporting system(FAERS) data and transcriptomic profiles[J]. Genes, 2025, 16( 3): 248. DOI: 10.3390/genes16030248. |
| [65] |
XIAO M, LI L, ZHU WW, et al. Statin-related neurocognitive disorder: A real-world pharmacovigilance study based on the FDA adverse event reporting system[J]. Expert Rev Clin Pharmacol, 2024, 17( 3): 255- 261. DOI: 10.1080/17512433.2024.2311875. |
| [66] |
KIM H, KIM N, LEE DH, et al. Analysis of national pharmacovigilance data associated with statin use in Korea[J]. Basic Clin Pharmacol Toxicol, 2017, 121( 5): 409- 413. DOI: 10.1111/bcpt.12808. |
| [67] |
KARAHALIL B, HARE E, KOÇ G, et al. Hepatotoxicity associated with statins[J]. Arh Hig Rada Toksikol, 2017, 68( 4): 254- 260. DOI: 10.1515/aiht-2017-68-2994. |
| [68] |
AVERBUKH LD, TURSHUDZHYAN A, WU DC, et al. Statin-induced liver injury patterns: A clinical review[J]. J Clin Transl Hepatol, 2022, 10( 3): 543- 552. DOI: 10.14218/JCTH.2021.00271. |
| [69] |
WANG HJ, LIU SY, ZHOU CJ, et al. Fatal hepatic failure following atorvastatin treatment: A case report[J]. Medicine, 2023, 102( 19): e33743. DOI: 10.1097/MD.0000000000033743. |



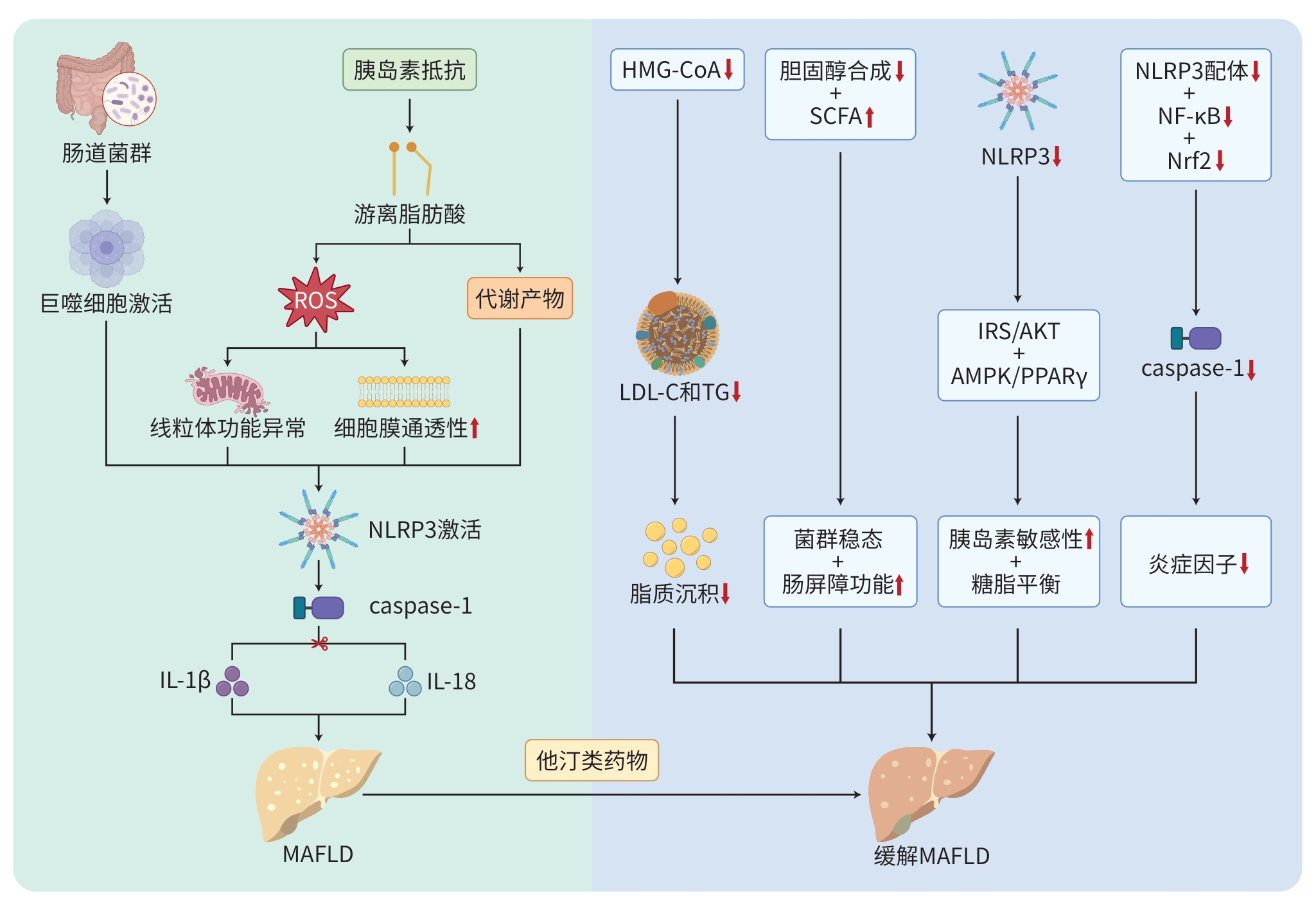




 DownLoad:
DownLoad: