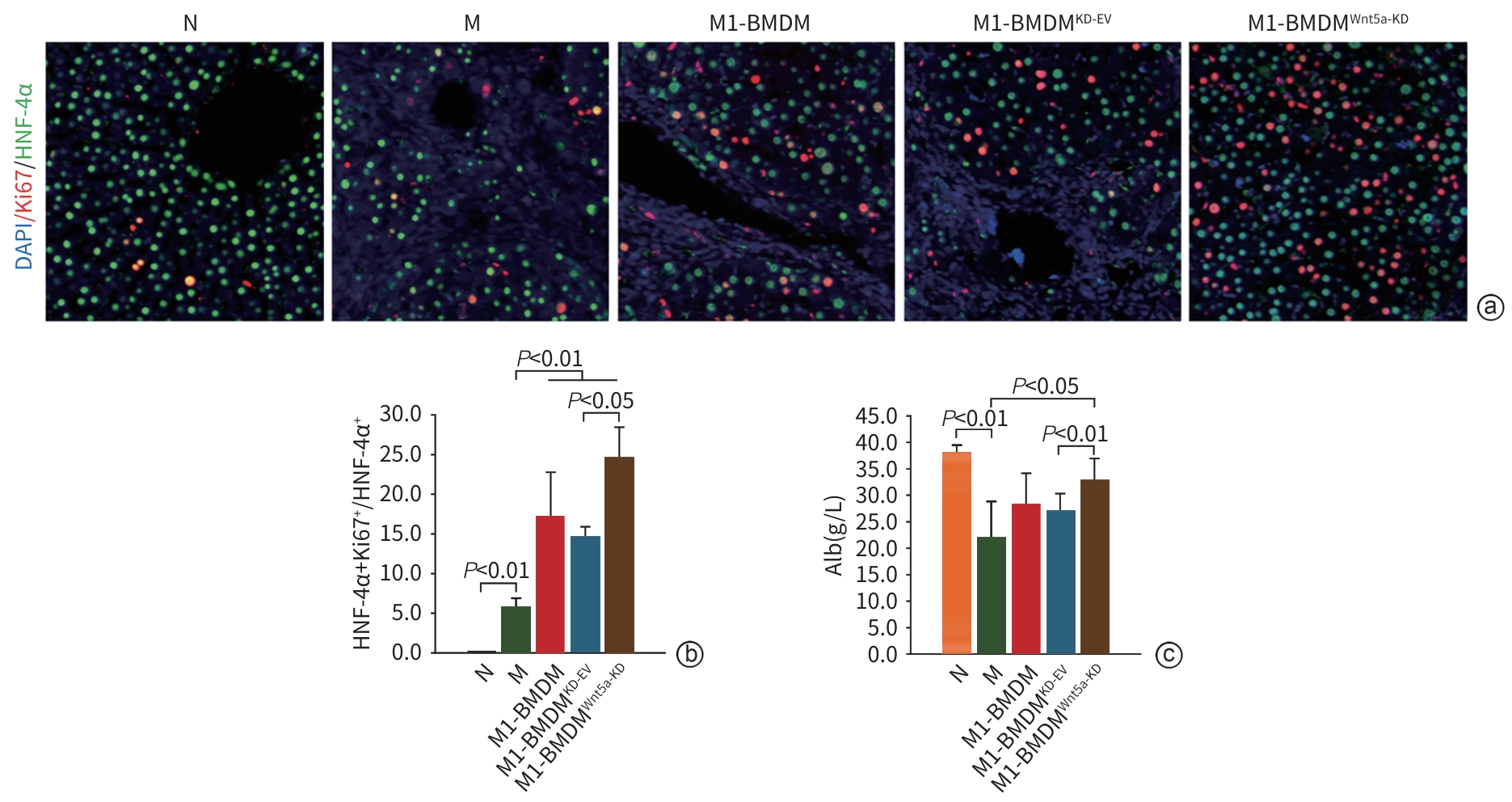
| [1] |
OAKLEY F. Interrogating mechanisms of liver fibrosis with omics[J]. Nat Rev Gastroenterol Hepatol, 2022, 19( 2): 89- 90. DOI: 10.1038/s41575-021-00567-6. |
| [2] |
LIAO ZH, XIE ZY. Research progress in molecular mechanism of hepatic fibrosis and related therapeutic targets[J]. J Jilin Univ: Med Edit, 2024, 50( 5): 1450- 1456. DOI: 10.13481/j.1671-587X.20240532. |
| [3] |
LI L, CUI L, LIN P, et al. Kupffer-cell-derived IL-6 is repurposed for hepatocyte dedifferentiation via activating progenitor genes from injury-specific enhancers[J]. Cell Stem Cell, 2023, 30( 3): 283- 299.e9. DOI: 10.1016/j.stem.2023.01.009. |
| [4] |
THOMAS JA, POPE C, WOJTACHA D, et al. Macrophage therapy for murine liver fibrosis recruits host effector cells improving fibrosis, regeneration, and function[J]. Hepatology, 2011, 53( 6): 2003- 2015. DOI: 10.1002/hep.24315. |
| [5] |
MA PF, GAO CC, YI J, et al. Cytotherapy with M1-polarized macrophages ameliorates liver fibrosis by modulating immune microenvironment in mice[J]. J Hepatol, 2017, 67( 4): 770- 779. DOI: 10.1016/j.jhep.2017.05.022. |
| [6] |
ZONG MY, JIAN X, WANG DY, et al. Effect of Yiguan Decoction on the efficacy of M1 bone marrow-derived macrophages in treatment of liver cirrhosis rats and its mechanism[J]. J Clin Hepatol, 2024, 40( 8): 1612- 1619. DOI: 10.12449/JCH240817. 宗梦瑶, 简迅, 王丹阳, 等. 一贯煎对M1型骨髓巨噬细胞治疗肝硬化大鼠模型效果的影响及其机制分析[J]. 临床肝胆病杂志, 2024, 40( 8): 1612- 1619. DOI: 10.12449/JCH240817. |
| [7] |
CAO MZ, CHAN RWS, CHENG FHC, et al. Myometrial cells stimulate self-renewal of endometrial mesenchymal stem-like cells through WNT5A/β-catenin signaling[J]. Stem Cells, 2019, 37( 11): 1455- 1466. DOI: 10.1002/stem.3070. |
| [8] |
LIN QQ, WU LM, CHATLA S, et al. Hematopoietic stem cell regeneration through paracrine regulation of the Wnt5a/Prox1 signaling axis[J]. J Clin Invest, 2022, 132( 12): e155914. DOI: 10.1172/JCI155914. |
| [9] |
ZHAO CG, QIN J, LI J, et al. LINGO-1 regulates Wnt5a signaling during neural stem and progenitor cell differentiation by modulating miR-15b-3p levels[J]. Stem Cell Res Ther, 2021, 12( 1): 372. DOI: 10.1186/s13287-021-02452-0. |
| [10] |
TIAN Y, CHEN JC, HUANG WS, et al. Myeloid-derived Wnts play an indispensible role in macrophage and fibroblast activation and kidney fibrosis[J]. Int J Biol Sci, 2024, 20( 6): 2310- 2322. DOI: 10.7150/ijbs.94166. |
| [11] |
JIAN X, WANG DY, XU YN, et al. Effect of polarized bone marrow-derived macrophage transplantation on the progression of CCl 4-induced liver fibrosis in rats[J]. J Clin Hepatol, 2021, 37( 12): 2830- 2837. DOI: 10.3969/j.issn.1001-5256.2021.12.020. |
| [12] |
HAAG SM, MURTHY A. Murine monocyte and macrophage culture[J]. Bio Protoc, 2021, 11( 6): e3928. DOI: 10.21769/BioProtoc.3928. |
| [13] |
JAMALL IS, FINELLI VN, QUE HEE SS. A simple method to determine nanogram levels of 4-hydroxyproline in biological tissues[J]. Anal Biochem, 1981, 112( 1): 70- 75. DOI: 10.1016/0003-2697(81)90261-x. |
| [14] |
GUILLOT A, TACKE F. Spatial dimension of macrophage heterogeneity in liver diseases[J]. eGastroenterology, 2023, 1( 1): e000003. DOI: 10.1136/egastro-2023-000003. |
| [15] |
FLORES MOLINA M, ABDELNABI MN, MAZOUZ S, et al. Distinct spatial distribution and roles of Kupffer cells and monocyte-derived macrophages in mouse acute liver injury[J]. Front Immunol, 2022, 13: 994480. DOI: 10.3389/fimmu.2022.994480. |
| [16] |
NA YR, KIM SW, SEOK SH. A new era of macrophage-based cell therapy[J]. Exp Mol Med, 2023, 55( 9): 1945- 1954. DOI: 10.1038/s12276-023-01068-z. |
| [17] |
CHEN T, CHEN Y. Research progress in mechanism of fibrosis regulated by macrophage polarization[J]. J Jilin Univ: Med Edit, 2024, 50( 5): 1465- 1473. DOI: 10.13481/j.1671-587X.20240534. |
| [18] |
SHAO Y, ZHENG QQ, WANG W, et al. Biological functions of macrophage-derived Wnt5a, and its roles in human diseases[J]. Oncotarget, 2016, 7( 41): 67674- 67684. DOI: 10.18632/oncotarget.11874. |
| [19] |
MENCK K, KLEMM F, GROSS JC, et al. Induction and transport of Wnt 5a during macrophage-induced malignant invasion is mediated by two types of extracellular vesicles[J]. Oncotarget, 2013, 4( 11): 2057- 2066. DOI: 10.18632/oncotarget.1336. |
| [20] |
LIU Q, SONG JL, PAN Y, et al. Wnt5a/CaMKII/ERK/CCL2 axis is required for tumor-associated macrophages to promote colorectal cancer progression[J]. Int J Biol Sci, 2020, 16( 6): 1023- 1034. DOI: 10.7150/ijbs.40535. |
| [21] |
MA X, HUANG TD, CHEN XZ, et al. Molecular mechanisms in liver repair and regeneration: From physiology to therapeutics[J]. Signal Transduct Target Ther, 2025, 10( 1): 63. DOI: 10.1038/s41392-024-02104-8. |
| [22] |
HUCK I, GUNEWARDENA S, ESPANOL-SUNER R, et al. Hepatocyte nuclear factor 4 alpha activation is essential for termination of liver regeneration in mice[J]. Hepatology, 2019, 70( 2): 666- 681. DOI: 10.1002/hep.30405. |
| [23] |
SONG ZM, LIN SR, WU XW, et al. Hepatitis B virus-related intrahepatic cholangiocarcinoma originates from hepatocytes[J]. Hepatol Int, 2023, 17( 5): 1300- 1317. DOI: 10.1007/s12072-023-10556-3. |
| [24] |
SCHOLZEN T, GERDES J. The Ki-67 protein: From the known and the unknown[J]. J Cell Physiol, 2000, 182( 3): 311- 322. DOI: 3.0.CO;2-9">10.1002/(SICI)1097-4652(200003)182: 3<311:: AID-JCP1>3.0.CO;2-9.
|
| [25] |
SUN DQ, ZHOU XL, WU T, et al. Quercetin promotes the M1-to-M2 macrophage phenotypic switch during liver fibrosis treatment by modulating the JAK2/STAT3 signaling pathway[J]. Recent Pat Anticancer Drug Discov, 2026, 21( 1): 31- 46. DOI: 10.2174/0115748928318948240920044716. |
| [26] |
MORONI F, DWYER BJ, GRAHAM C, et al. Safety profile of autologous macrophage therapy for liver cirrhosis[J]. Nat Med, 2019, 25( 10): 1560- 1565. DOI: 10.1038/s41591-019-0599-8. |
| [27] |
BRENNAN PN, MACMILLAN M, MANSHIP T, et al. Autologous macrophage therapy for liver cirrhosis: A phase 2 open-label randomized controlled trial[J]. Nat Med, 2025, 31( 3): 979- 987. DOI: 10.1038/s41591-024-03406-8. |



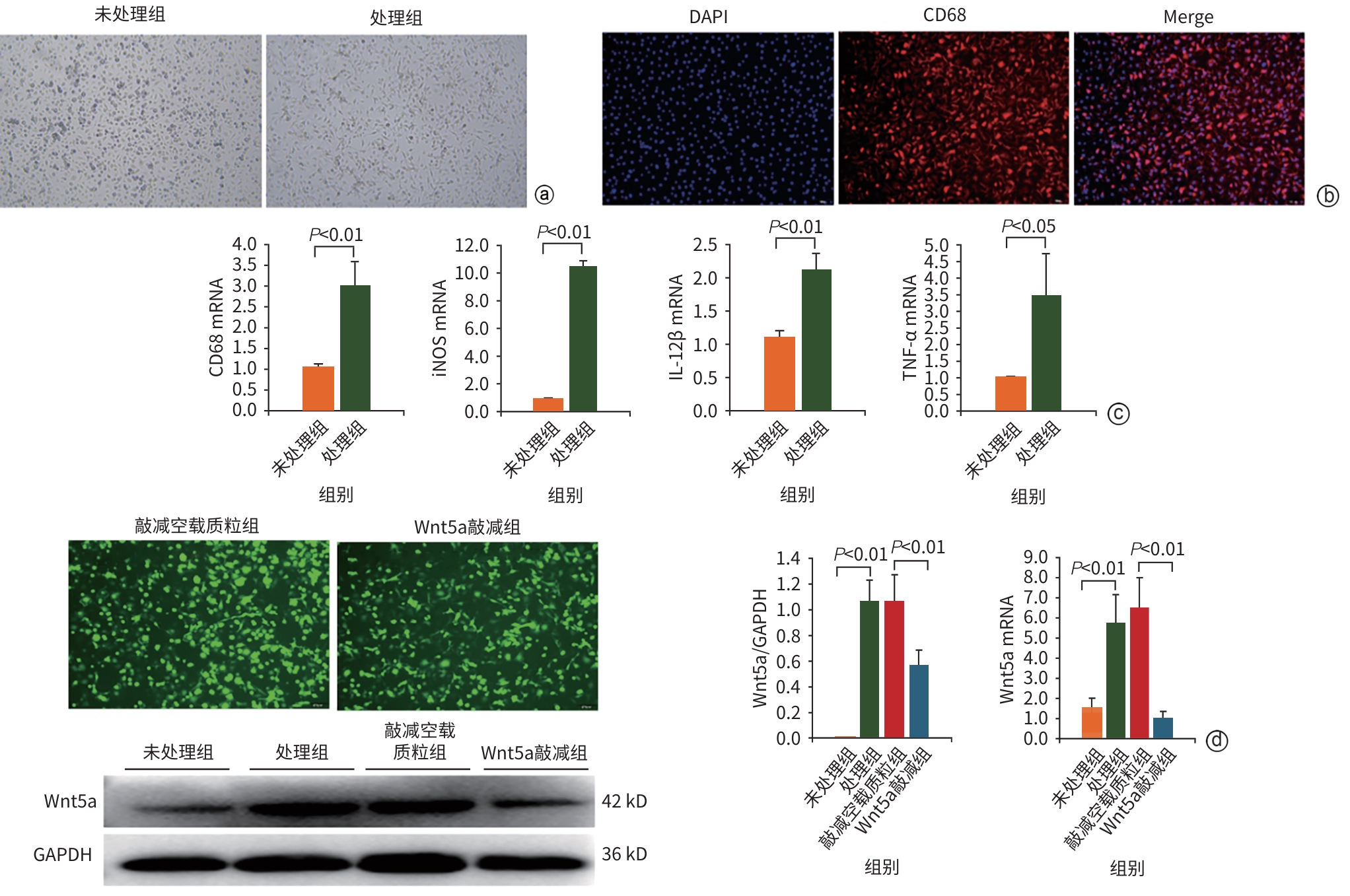




 DownLoad:
DownLoad: