| [1] |
HUANG DQ, EL-SERAG HB, LOOMBA R. Global epidemiology of NAFLD-related HCC: Trends, predictions, risk factors and prevention[J]. Nat Rev Gastroenterol Hepatol, 2021, 18( 4): 223- 238. DOI: 10.1038/s41575-020-00381-6. |
| [2] |
GRANDER C, GRABHERR F, TILG H. Non-alcoholic fatty liver disease: Pathophysiological concepts and treatment options[J]. Cardiovasc Res, 2023, 119( 9): 1787- 1798. DOI: 10.1093/cvr/cvad095. |
| [3] |
ZOU HM, GE Y, LEI Q, et al. Epidemiology and disease burden of non-alcoholic steatohepatitis in greater China: A systematic review[J]. Hepatol Int, 2022, 16( 1): 27- 37. DOI: 10.1007/s12072-021-10286-4. |
| [4] |
National Workshop on Fatty Liver and Alcoholic Liver Disease, Chinese Society of Hepatology, Chinese Medical Association; Fatty Liver Expert Committee, Chinese Medical Doctor Association. Guidelines of prevention and treatment for nonalcoholic fatty liver disease: a 2018 update[J]. J Clin Hepatol, 2018, 34( 5): 947- 957. DOI: 10.3969/j.issn.1001-5256.2018.05.007. |
| [5] |
DUFOUR JF, ANSTEE QM, BUGIANESI E, et al. Current therapies and new developments in NASH[J]. Gut, 2022, 71( 10): 2123- 2134. DOI: 10.1136/gutjnl-2021-326874. |
| [6] |
GREENWELL BJ, TROTT AJ, BEYTEBIERE JR, et al. Rhythmic food intake drives rhythmic gene expression more potently than the hepatic circadian clock in mice[J]. Cell Rep, 2019, 27( 3): 649- 657. DOI: 10.1016/j.celrep.2019.03.064. |
| [7] |
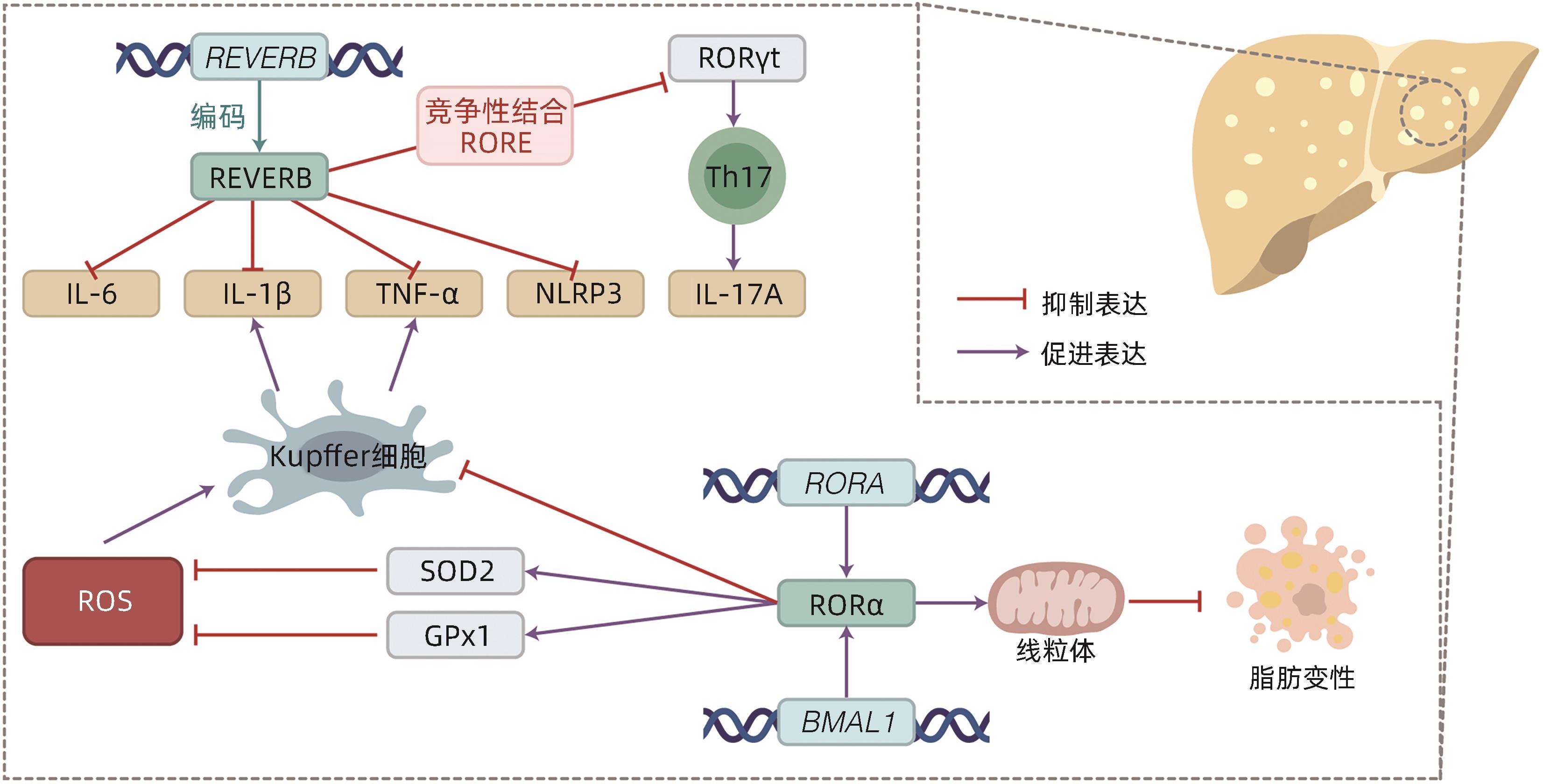
KIM HJ, HAN YH, NA H, et al. Liver-specific deletion of RORα aggravates diet-induced nonalcoholic steatohepatitis by inducing mitochondrial dysfunction[J]. Sci Rep, 2017, 7( 1): 16041. DOI: 10.1038/s41598-017-16077-y. |
| [8] |
FAGIANI F, MARINO DD, ROMAGNOLI A, et al. Molecular regulations of circadian rhythm and implications for physiology and diseases[J]. Signal Transduct Target Ther, 2022, 7( 1): 41. DOI: 10.1038/s41392-022-00899-y. |
| [9] |
VANDENBERGHE A, LEFRANC M, FURLAN A. An overview of the circadian clock in the frame of chronotherapy: From bench to bedside[J]. Pharmaceutics, 2022, 14( 7): 1424. DOI: 10.3390/pharmaceutics14071424. |
| [10] |
MUKHERJI A, BAILEY SM, STAELS B, et al. The circadian clock and liver function in health and disease[J]. J Hepatol, 2019, 71( 1): 200- 211. DOI: 10.1016/j.jhep.2019.03.020. |
| [11] |
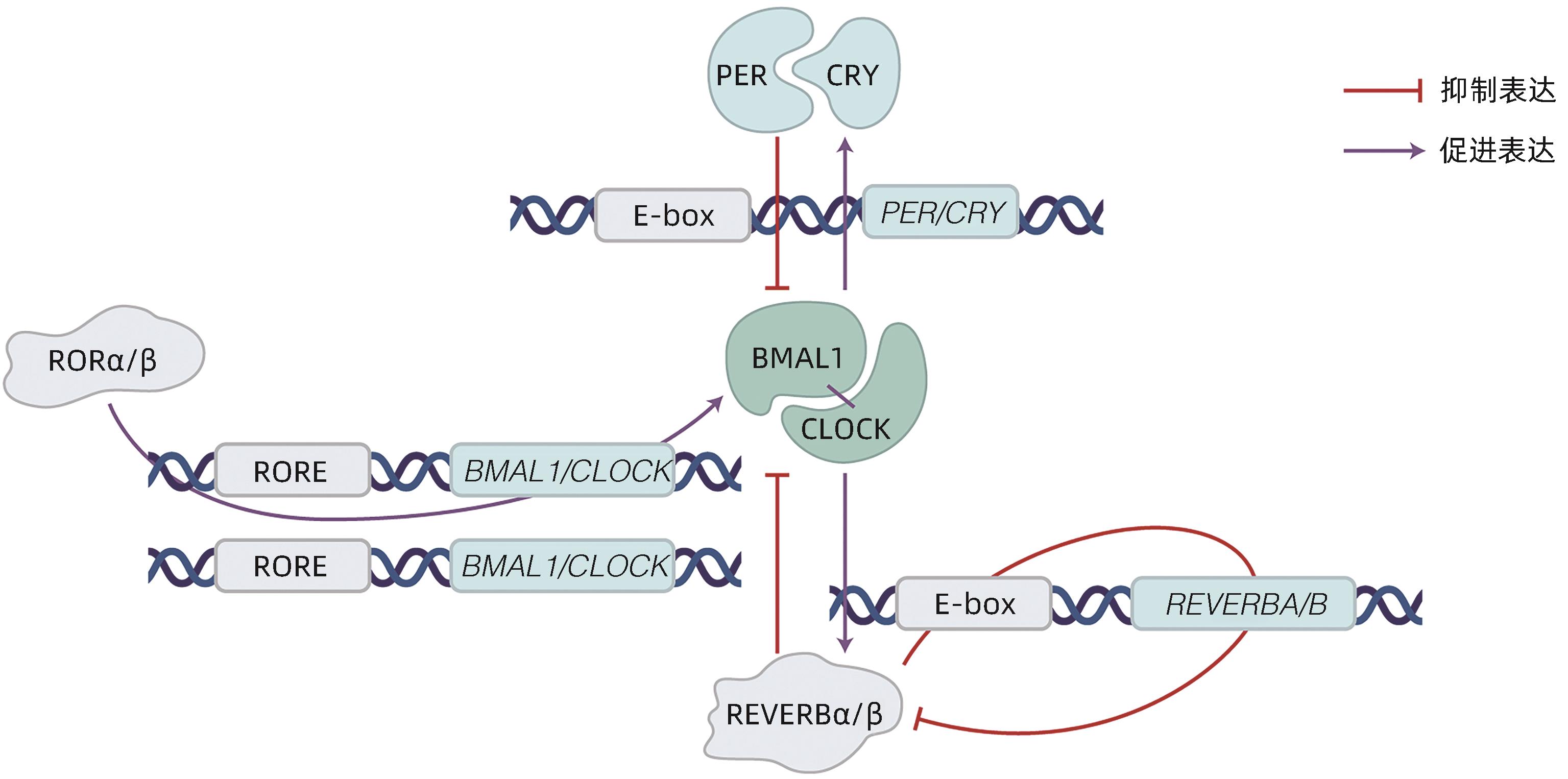
ABE YO, YOSHITANE H, KIM DW, et al. Rhythmic transcription of Bmal1 stabilizes the circadian timekeeping system in mammals[J]. Nat Commun, 2022, 13( 1): 4652. DOI: 10.1038/s41467-022-32326-9. |
| [12] |
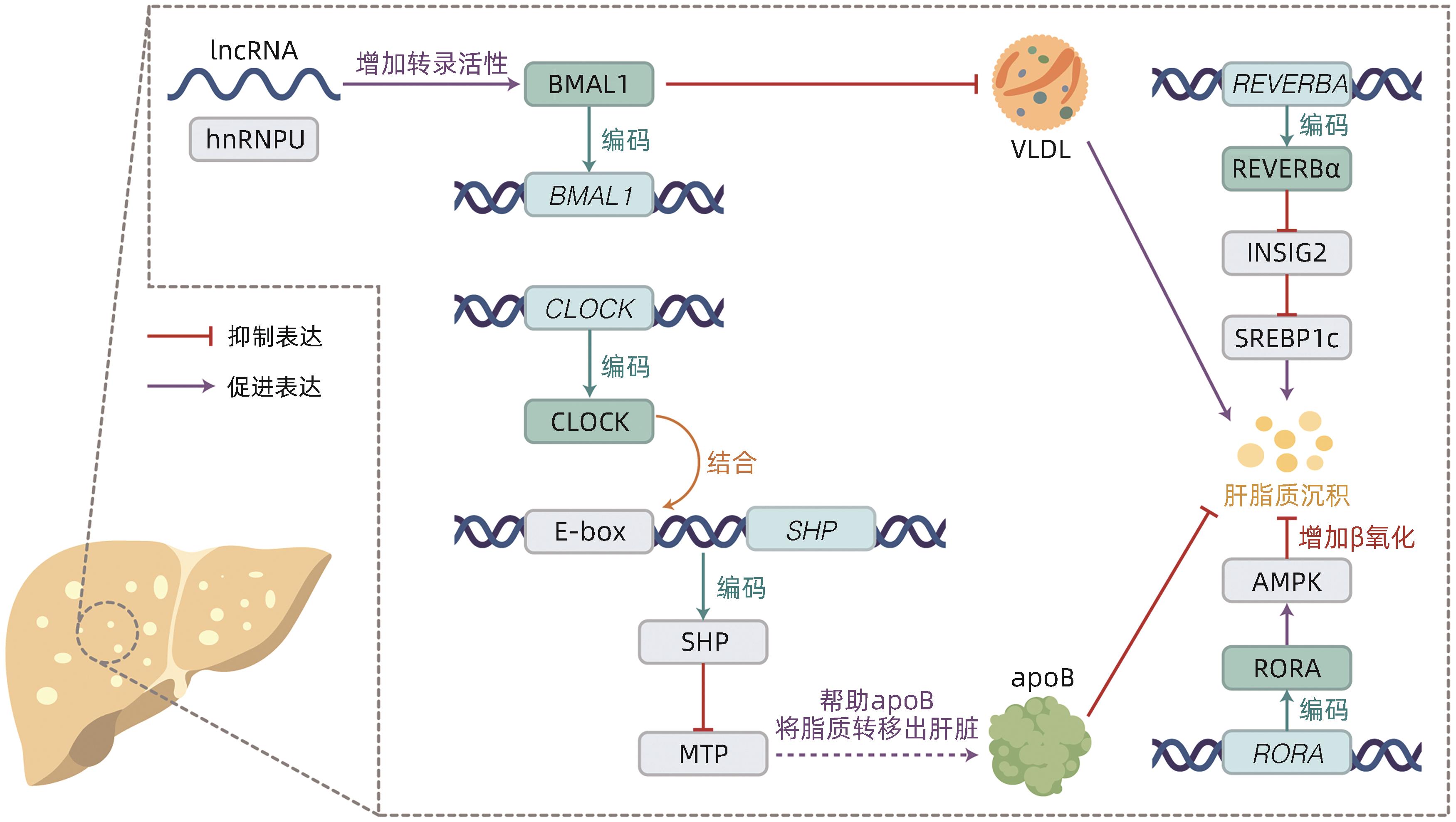
SHEN X, ZHANG Y, JI X, et al. Long noncoding RNA lncRHL regulates hepatic VLDL secretion by modulating hnRNPU/BMAL1/MTTP axis[J]. Diabetes, 2022, 71( 9): 1915- 1928. DOI: 10.2337/db21-1145. |
| [13] |
YE CS, ZHANG YJ, LIN SM, et al. Berberine ameliorates metabolic-associated fatty liver disease mediated metabolism disorder and redox homeostasis by upregulating clock genes: Clock and Bmal1 expressions[J]. Molecules, 2023, 28( 4): 1874. DOI: 10.3390/molecules28041874. |
| [14] |
LIN HG, WANG L, LIU ZH, et al. Hepatic MDM2 causes metabolic associated fatty liver disease by blocking triglyceride-VLDL secretion via ApoB degradation[J]. Adv Sci(Weinh), 2022, 9( 20): e2200742. DOI: 10.1002/advs.202200742. |
| [15] |
PAN XY, ZHANG YX, WANG L, et al. Diurnal regulation of MTP and plasma triglyceride by CLOCK is mediated by SHP[J]. Cell Metab, 2010, 12( 2): 174- 186. DOI: 10.1016/j.cmet.2010.05.014. |
| [16] |
BOLSHETTE N, IBRAHIM H, REINKE H, et al. Circadian regulation of liver function: From molecular mechanisms to disease pathophysiology[J]. Nat Rev Gastroenterol Hepatol, 2023, 20( 11): 695- 707. DOI: 10.1038/s41575-023-00792-1. |
| [17] |
REINKE H, ASHER G. Crosstalk between metabolism and circadian clocks[J]. Nat Rev Mol Cell Biol, 2019, 20( 4): 227- 241. DOI: 10.1038/s41580-018-0096-9. |
| [18] |
GRIFFETT K, HAYES ME, BOECKMAN MP, et al. The role of REV-ERB in NASH[J]. Acta Pharmacol Sin, 2022, 43( 5): 1133- 1140. DOI: 10.1038/s41401-022-00883-w. |
| [19] |
LEE KC, WU PS, LIN HC. Pathogenesis and treatment of non-alcoholic steatohepatitis and its fibrosis[J]. Clin Mol Hepatol, 2023, 29( 1): 77- 98. DOI: 10.3350/cmh.2022.0237. |
| [20] |
CHHUNCHHA B, KUBO ER, SINGH DP. Clock protein Bmal1 and Nrf2 cooperatively control aging or oxidative response and redox homeostasis by regulating rhythmic expression of Prdx6[J]. Cells, 2020, 9( 8): 1861. DOI: 10.3390/cells9081861. |
| [21] |
LENNICKE C, COCHEMÉ HM. Redox regulation of the insulin signalling pathway[J]. Redox Biol, 2021, 42: 101964. DOI: 10.1016/j.redox.2021.101964. |
| [22] |
WANG S, LIN YK, YUAN X, et al. REV-ERBα integrates colon clock with experimental colitis through regulation of NF-κB/NLRP3 axis[J]. Nat Commun, 2018, 9( 1): 4246. DOI: 10.1038/s41467-018-06568-5. |
| [23] |
HAN YH, KIM HJ, NA H, et al. RORα induces KLF4-mediated M2 polarization in the liver macrophages that protect against nonalcoholic steatohepatitis[J]. Cell Rep, 2017, 20( 1): 124- 135. DOI: 10.1016/j.celrep.2017.06.017. |
| [24] |
KIM HJ, HAN YH, KIM JY, et al. RORα enhances lysosomal acidification and autophagic flux in the hepatocytes[J]. Hepatol Commun, 2021, 5( 12): 2121- 2138. DOI: 10.1002/hep4.1785. |
| [25] |
GUAN DY, BAE H, ZHOU DS, et al. Hepatocyte SREBP signaling mediates clock communication within the liver[J]. J Clin Invest, 2023, 133( 8): e163018. DOI: 10.1172/JCI163018. |
| [26] |
NI YH, ZHAO YF, MA LY, et al. Pharmacological activation of REV-ERBα improves nonalcoholic steatohepatitis by regulating intestinal permeability[J]. Metabolism, 2021, 114: 154409. DOI: 10.1016/j.metabol.2020.154409. |
| [27] |
LEE YS, CHA BY, SAITO K, et al. Nobiletin improves hyperglycemia and insulin resistance in obese diabetic ob/ob mice[J]. Biochem Pharmacol, 2010, 79( 11): 1674- 1683. DOI: 10.1016/j.bcp.2010.01.034. |
| [28] |
KHAMBU B, YAN SM, HUDA N, et al. Autophagy in non-alcoholic fatty liver disease and alcoholic liver disease[J]. Liver Res, 2018, 2( 3): 112- 119. DOI: 10.1016/j.livres.2018.09.004. |
| [29] |
SARAN AR, DAVE S, ZARRINPAR A. Circadian rhythms in the pathogenesis and treatment of fatty liver disease[J]. Gastroenterology, 2020, 158( 7): 1948- 1966. DOI: 10.1053/j.gastro.2020.01.050. |
| [30] |
FANG CQ, PAN JH, QU N, et al. The AMPK pathway in fatty liver disease[J]. Front Physiol, 2022, 13: 970292. DOI: 10.3389/fphys.2022.970292. |
| [31] |
SHI DM, CHEN J, WANG JF, et al. Circadian clock genes in the metabolism of non-alcoholic fatty liver disease[J]. Front Physiol, 2019, 10: 423. DOI: 10.3389/fphys.2019.00423. |
| [32] |
ZHAO HK, WU L, YAN GF, et al. Inflammation and tumor progression: Signaling pathways and targeted intervention[J]. Signal Transduct Target Ther, 2021, 6( 1): 263. DOI: 10.1038/s41392-021-00658-5. |
| [33] |
SINGH V, UBAID S. Role of silent information regulator 1(SIRT1) in regulating oxidative stress and inflammation[J]. Inflammation, 2020, 43( 5): 1589- 1598. DOI: 10.1007/s10753-020-01242-9. |
| [34] |
HAN SC, LI ZZ, HAN F, et al. ROR alpha protects against LPS-induced inflammation by down-regulating SIRT1/NF-kappa B pathway[J]. Arch Biochem Biophys, 2019, 668: 1- 8. DOI: 10.1016/j.abb.2019.05.003. |
| [35] |
CHYAU CC, WANG HF, ZHANG WJ, et al. Antrodan alleviates high-fat and high-fructose diet-induced fatty liver disease in C57BL/6 mice model via AMPK/Sirt1/SREBP-1c/PPARγ pathway[J]. Int J Mol Sci, 2020, 21( 1): 360. DOI: 10.3390/ijms21010360. |
| [36] |
ZHANG CY, TAN XH, YANG HH, et al. COX-2/sEH dual inhibitor alleviates hepatocyte senescence in NAFLD mice by restoring autophagy through Sirt1/PI3K/AKT/mTOR[J]. Int J Mol Sci, 2022, 23( 15): 8267. DOI: 10.3390/ijms23158267. |
| [37] |
ZHOU B, ZHANG Y, ZHANG F, et al. CLOCK/BMAL1 regulates circadian change of mouse hepatic insulin sensitivity by SIRT1[J]. Hepatology, 2014, 59( 6): 2196- 2206. DOI: 10.1002/hep.26992. |
| [38] |
ZHONG DD, CAI J, HU C, et al. Inhibition of mPGES-2 ameliorates NASH by activating NR1D1 via heme[J]. Hepatology, 2023, 78( 2): 547- 561. DOI: 10.1002/hep.32671. |
| [39] |
SUN Y, JIA ZJ, YANG GR, et al. mPGES-2 deletion remarkably enhances liver injury in streptozotocin-treated mice via induction of GLUT2[J]. J Hepatol, 2014, 61( 6): 1328- 1336. DOI: 10.1016/j.jhep.2014.07.018. |
| [40] |
LI RB, XIN T, LI DD, et al. Therapeutic effect of Sirtuin 3 on ameliorating nonalcoholic fatty liver disease: The role of the ERK-CREB pathway and Bnip3-mediated mitophagy[J]. Redox Biol, 2018, 18: 229- 243. DOI: 10.1016/j.redox.2018.07.011. |
| [41] |
WU SN, LU QL, WANG QL, et al. Binding of FUN14 domain containing 1 with inositol 1, 4, 5-trisphosphate receptor in mitochondria-associated endoplasmic reticulum membranes maintains mitochondrial dynamics and function in hearts in vivo[J]. Circulation, 2017, 136( 23): 2248- 2266. DOI: 10.1161/CIRCULATIONAHA.117.030235. |
| [42] |
CUI AY, DING D, LI Y. Regulation of hepatic metabolism and cell growth by the ATF/CREB family of transcription factors[J]. Diabetes, 2021, 70( 3): 653- 664. DOI: 10.2337/dbi20-0006. |
| [43] |
SHIMIZU-ALBERGINE M, BASU D, KANTER JE, et al. CREBH normalizes dyslipidemia and halts atherosclerosis in diabetes by decreasing circulating remnant lipoproteins[J]. J Clin Invest, 2021, 131( 22): e153285. DOI: 10.1172/JCI153285. |
| [44] |
YANG Z, KIM H, ALI A, et al. Interaction between stress responses and circadian metabolism in metabolic disease[J]. Liver Res, 2017, 1( 3): 156- 162. DOI: 10.1016/j.livres.2017.11.002. |







 DownLoad:
DownLoad: