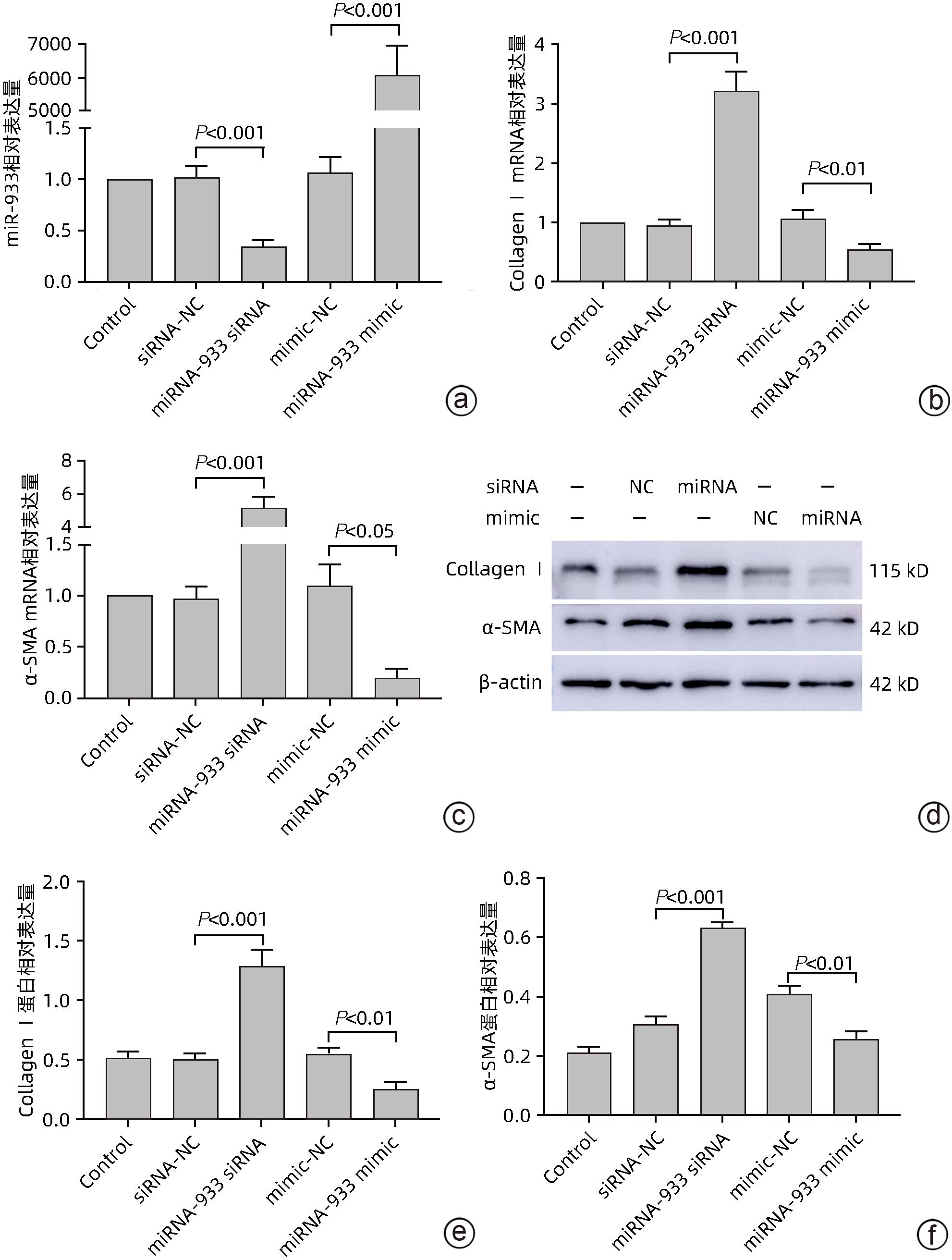
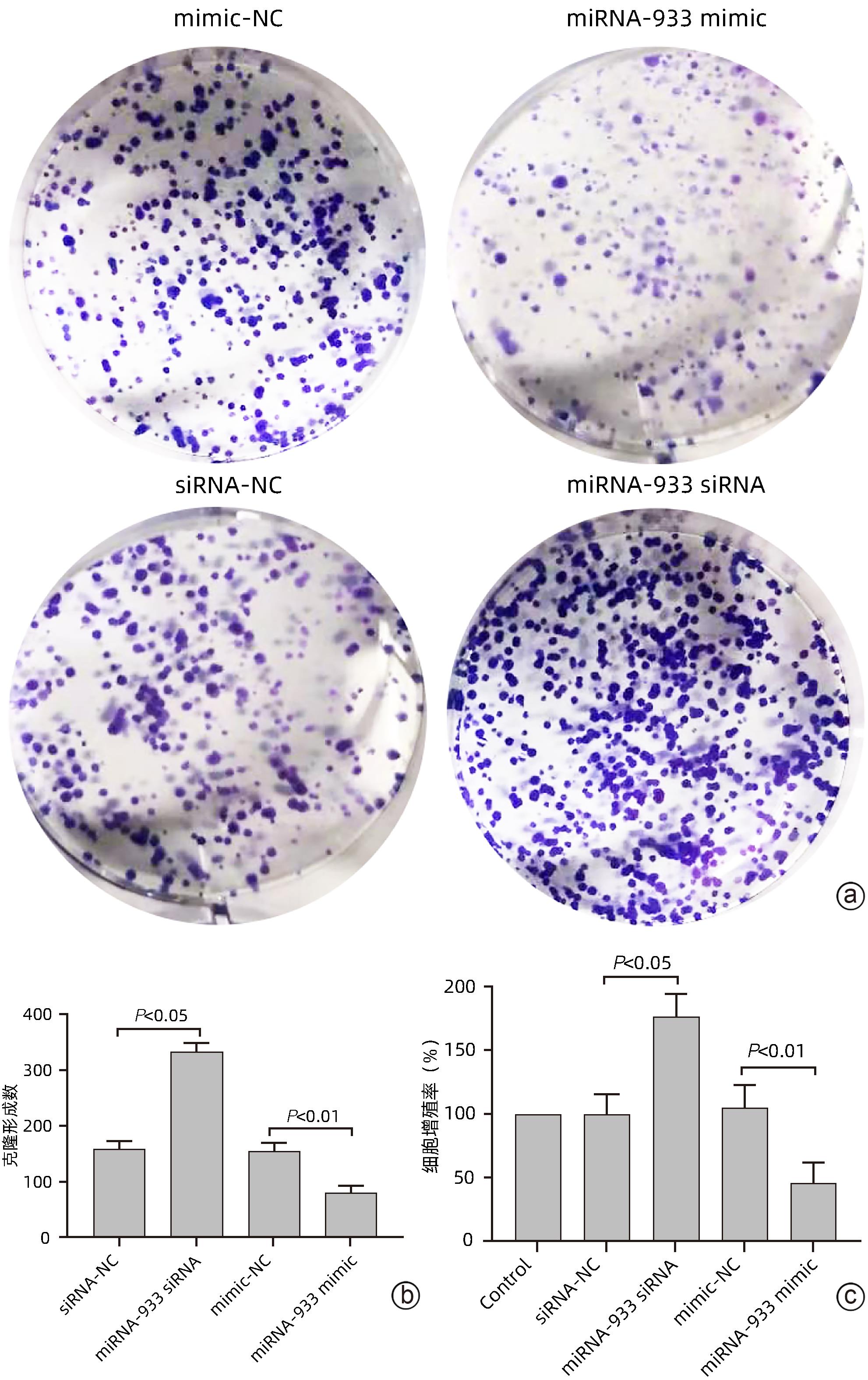
| [1] |
TSUCHIDA T, FRIEDMAN SL. Mechanisms of hepatic stellate cell activation[J]. Nat Rev Gastroenterol Hepatol, 2017, 14( 7): 397- 411. DOI: 10.1038/nrgastro.2017.38. |
| [2] |
ZHANG CY, YUAN WG, HE P, et al. Liver fibrosis and hepatic stellate cells: Etiology, pathological hallmarks and therapeutic targets[J]. World J Gastroenterol, 2016, 22( 48): 10512- 10522. DOI: 10.3748/wjg.v22.i48.10512. |
| [3] |
HIGASHI T, FRIEDMAN SL, HOSHIDA Y. Hepatic stellate cells as key target in liver fibrosis[J]. Adv Drug Deliv Rev, 2017, 121: 27- 42. DOI: 10.1016/j.addr.2017.05.007. |
| [4] |
MARTÍ-RODRIGO A, ALEGRE F, ÁB MORAGREGA, et al. Rilpivirine attenuates liver fibrosis through selective STAT1-mediated apoptosis in hepatic stellate cells[J]. Gut, 2020, 69( 5): 920- 932. DOI: 10.1136/gutjnl-2019-318372. |
| [5] |
RUPAIMOOLE R, SLACK FJ. MicroRNA therapeutics: Towards a new era for the management of cancer and other diseases[J]. Nat Rev Drug Discov, 2017, 16( 3): 203- 222. DOI: 10.1038/nrd.2016.246. |
| [6] |
JIANG XP, AI WB, WAN LY, et al. The roles of microRNA families in hepatic fibrosis[J]. Cell Biosci, 2017, 7: 34. DOI: 10.1186/s13578-017-0161-7. |
| [7] |
LIN HY, WANG FS, YANG YL, et al. MicroRNA-29a suppresses CD36 to ameliorate high fat diet-induced steatohepatitis and liver fibrosis in mice[J]. Cells, 2019, 8( 10): 1298. DOI: 10.3390/cells8101298. |
| [8] |
SONG L, CHEN TY, ZHAO XJ, et al. Pterostilbene prevents hepatocyte epithelial-mesenchymal transition in fructose-induced liver fibrosis through suppressing miR-34a/Sirt1/p53 and TGF-β1/Smads signalling[J]. Br J Pharmacol, 2019, 176( 11): 1619- 1634. DOI: 10.1111/bph.14573. |
| [9] |
|
| [10] |
LOU WY, LIU JX, GAO YJ, et al. MicroRNA regulation of liver cancer stem cells[J]. Am J Cancer Res, 2018, 8( 7): 1126- 1141.
|
| [11] |
ROEHLEN N, CROUCHET E, BAUMERT TF. Liver fibrosis: Mechanistic concepts and therapeutic perspectives[J]. Cells, 2020, 9( 4): 875. DOI: 10.3390/cells9040875. |
| [12] |
EZHILARASAN D, SOKAL E, NAJIMI M. Hepatic fibrosis: It is time to go with hepatic stellate cell-specific therapeutic targets[J]. Hepatobiliary Pancreat Dis Int, 2018, 17( 3): 192- 197. DOI: 10.1016/j.hbpd.2018.04.003. |
| [13] |
LI J, CHAN MC, YU Y, et al. MiR-29b contributes to multiple types of muscle atrophy[J]. Nat Commun, 2017, 8: 15201. DOI: 10.1038/ncomms15201. |
| [14] |
CAI FF, BIAN YQ, WU R, et al. Yinchenhao decoction suppresses rat liver fibrosis involved in an apoptosis regulation mechanism based on network pharmacology and transcriptomic analysis[J]. Biomed Pharmacother, 2019, 114: 108863. DOI: 10.1016/j.biopha.2019.108863. |
| [15] |
HUANG YH, TIAO MM, HUANG LT, et al. Activation of miR-29a in activated hepatic stellate cells modulates its profibrogenic phenotype through inhibition of histone deacetylases 4[J]. PLoS One, 2015, 10( 8): e0136453. DOI: 10.1371/journal.pone.0136453. |
| [16] |
GUO CJ, PAN Q, LI DG, et al. MiR-15b and miR-16 are implicated in activation of the rat hepatic stellate cell: An essential role for apoptosis[J]. J Hepatol, 2009, 50( 4): 766- 778. DOI: 10.1016/j.jhep.2008.11.025. |
| [17] |
ZHU XQ, SHAO P, TANG YC, et al. hsa_circRNA_100533 regulates GNAS by sponging hsa_miR_933 to prevent oral squamous cell carcinoma[J]. J Cell Biochem, 2019, 120( 11): 19159- 19171. DOI: 10.1002/jcb.29245. |
| [18] |
LI HZ, ZHANG YW, XU YJ, et al. MiR-933 inhibits proliferation, migration and invasion of lung cancer cell lines by regulation of KLF6 gene[J]. China Oncol, 2021, 31( 7): 581- 588. DOI: 10.19401/j.cnki.1007-3639.2021.07.004. |
| [19] |
ZHANG YT, MA YY, XU WH, et al. Association of microRNA-933 variant with the susceptibility to gastric cancer[J]. J BUON, 2017, 22( 2): 390- 395.
|
| [20] |
ZHOU B, GUO H, TANG J. Long non-coding RNA TFAP2A-AS1 inhibits cell proliferation and invasion in breast cancer via miR-933/SMAD2[J]. Med Sci Monit, 2019, 25: 1242- 1253. DOI: 10.12659/MSM.912421. |
| [21] |
CHENG Y, SHI WW, CUI XD, et al. Long noncoding RNA TFAP2A-AS1 suppressed hepatitis B virus replication by modulating miR-933/HDAC11[J]. Dis Markers, 2022, 2022: 7733390. DOI: 10.1155/2022/7733390. |
| [22] |
ISLAM ABMMK, MOHAMMAD E, KHAN MA. Aberration of the modulatory functions of intronic microRNA hsa-miR-933 on its host gene ATF2 results in type II diabetes mellitus and neurodegenerative disease development[J]. Hum Genomics, 2020, 14( 1): 34. DOI: 10.1186/s40246-020-00285-1. |
| [23] |
RANE MJ, ZHAO YG, CAI L. Krϋppel-like factors(KLFs) in renal physiology and disease[J]. EBioMedicine, 2019, 40: 743- 750. DOI: 10.1016/j.ebiom.2019.01.021. |
| [24] |
SYAFRUDDIN SE, RODRIGUES P, VOJTASOVA E, et al. A KLF6-driven transcriptional network links lipid homeostasis and tumour growth in renal carcinoma[J]. Nat Commun, 2019, 10( 1): 1152. DOI: 10.1038/s41467-019-09116-x. |
| [25] |
LANG UE, KOCABAYOGLU P, CHENG GZ, et al. GSK3β phosphorylation of the KLF6 tumor suppressor promotes its transactivation of p21[J]. Oncogene, 2013, 32( 38): 4557- 4564. DOI: 10.1038/onc.2012.457. |
| [26] |
CAI MG, SHAO W, YU HJ, et al. Paeonol inhibits cell proliferation, migration and invasion and induces apoptosis in hepatocellular carcinoma by regulating miR-21-5p/KLF6 axis[J]. Cancer Manag Res, 2020, 12: 5931- 5943. DOI: 10.2147/CMAR.S254485. |
| [27] |
ZHANG N, YAN QQ, LU L, et al. The KLF6 splice variant KLF6-SV1 promotes proliferation and invasion of non-small cell lung cancer by up-regultating PI3K-AKT signaling pathway[J]. J Cancer, 2019, 10( 22): 5324- 5331. DOI: 10.7150/jca.34212. |
| [28] |
CHIAM K, RYAN NK, RICCIARDELLI C, et al. Characterization of the prostate cancer susceptibility gene KLF6 in human and mouse prostate cancers[J]. Prostate, 2013, 73( 2): 182- 193. DOI: 10.1002/pros.22554. |
| [29] |
GHIASSI-NEJAD Z, HERNANDEZ-GEA V, WOODRELL C, et al. Reduced hepatic stellate cell expression of Kruppel-like factor 6 tumor suppressor isoforms amplifies fibrosis during acute and chronic rodent liver injury[J]. Hepatology, 2013, 57( 2): 786- 796. DOI: 10.1002/hep.26056. |
| [30] |
TIAN F, ZHAO JZ, BU SC, et al. KLF6 induces apoptosis in human lens epithelial cells through the ATF4-ATF3-CHOP axis[J]. Drug Des Devel Ther, 2020, 14: 1041- 1055. DOI: 10.2147/DDDT.S218467. |



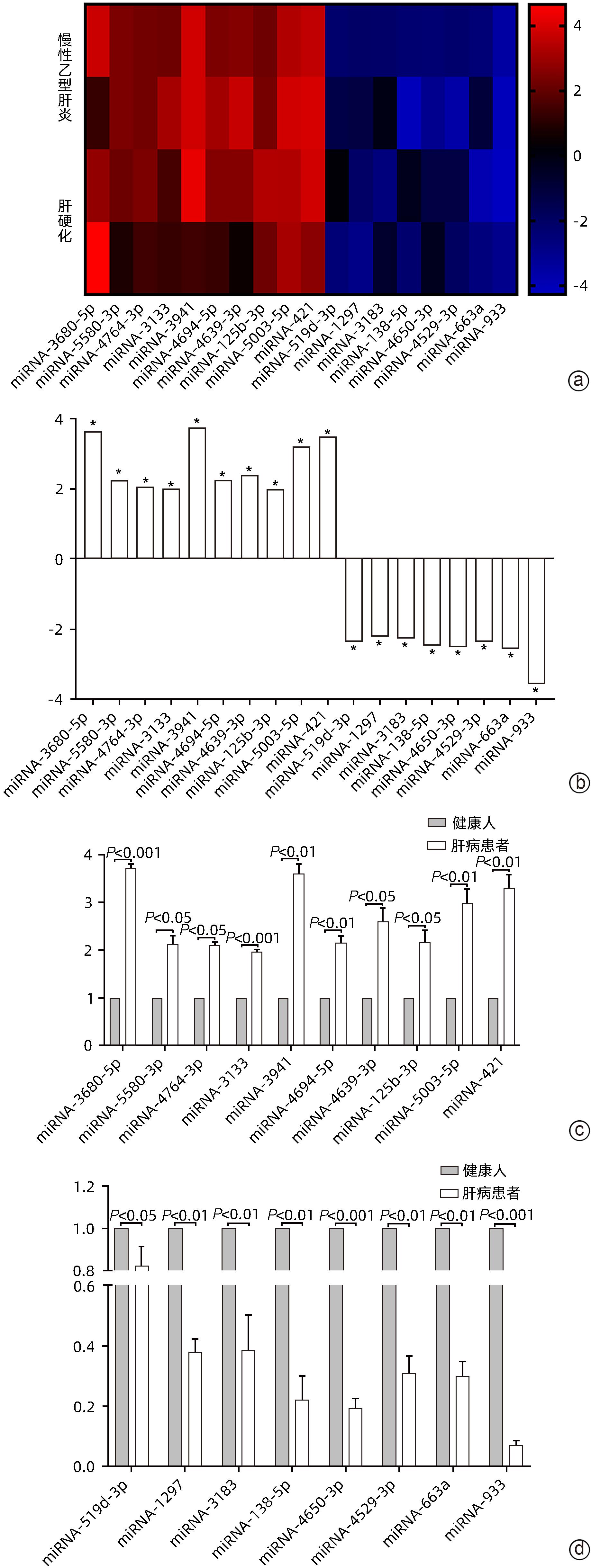




 DownLoad:
DownLoad: