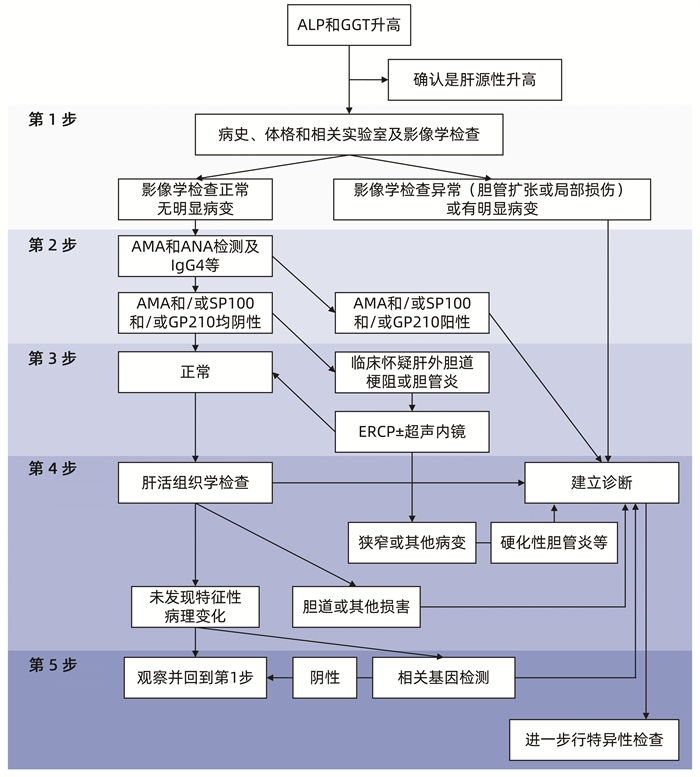
| [1] |
KUNTZ K, KUNTZ HD. Cholestasi hepatology: Principles and practice (2nd edition)[M]. Heidelberg, Germany: Springer Medizin Verlag Heidelberg, 2006: 227-242.
|
| [2] |
WU H, CHEN C, ZIANI S, et al. Fibrotic events in the progression of cholestatic liver disease[J]. Cells, 2021, 10(5): 1107. DOI: 10.3390/cells10051107. |
| [3] |
Chinese Society of Hepatology, Chinese Medical Association; Chinese Society of Gastroenterology, Chinese Medical Association; Chinese Society of Infectious Diseases, Chinese Medical Association. Consensus on the diagnosis and treatment of cholestasis liver diseases (2015) [J]. J Clin Hepatol, 2015, 31(12): 1989-1999. DOI: 10.3969/j.issn.1001-5256.2015.12.005. |
| [4] |
SHERLOCK S, DOOLEY J. Cholestasis[M]//SHERLOCK S, DOOLEY J. Disease of the liver and biliary system. 11th ed. Oxford, England: Blackwell Publishing, 2002: 219-240.
|
| [5] |
European Association for the Study of the Liver. EASL clinical practice guidelines: Management of cholestatic liver diseases[J]. J Hepatol, 2009, 51(2): 237-267. DOI: 10.1016/j.jhep.2009.04.009. |
| [6] |
BURT A, PORTMANN B, FERRELL L. MacSween's pathology of the liver (6th edition)[M]. London, England: Churchill Livingstone Elsevier, 2012: 503-562.
|
| [7] |
LEFKOWITCH JH. Scheuer's liver biopsy interpretation (9th edition)[M]. New York: Elsevier, 2016: 53.
|
| [8] |
HILSCHER MB, KAMATH PS, EATON JE. Cholestatic liver diseases: A primer for generalists and subspecialists[J]. Mayo Clin Proc, 2020, 95(10): 2263-2279. DOI: 10.1016/j.mayocp.2020.01.015. |
| [9] |
BORTOLINI M, ALMASIO P, BRAY G, et al. Multicentre survey of the prevalence of intrahepatic cholestasis in 2520 consecutive patients with newly diagnosed chronic liver disease[J]. Drug Investigation, 1992, 4(Suppl 4): 83-89.
|
| [10] |
XIE W, CAO Y, XU M, et al. Prognostic significance of elevated cholestatic enzymes for fibrosis and hepatocellular carcinoma in hospital discharged chronic viral hepatitis patients[J]. Sci Rep, 2017, 7(1): 10289. DOI: 10.1038/s41598-017-11111-5. |
| [11] |
CAO XX, GAO YQ, ZHANG WH, et al. Cholestasis morbidity rate in first-hospitalized patients with chronic liver disease in Shanghai[J]. Chin J Hepatol, 2015, 23(8): 569-573. DOI: 10.3760/cma.j.issn.1007-3418.2015.08.003. |
| [12] |
SIDDIQUE A, KOWDLEY KV. Approach to a patient with elevated serum alkaline phosphatase[J]. Clin Liver Dis, 2012, 16(2): 199-229. DOI: 10.1016/j.cld.2012.03.012. |
| [13] |
REICHERT MC, HALL RA, KRAWCZYK M, et al. Genetic determinants of cholangiopathies: Molecular and systems genetics[J]. Biochim Biophys Acta Mol Basis Dis, 2018, 1864(4 Pt B): 1484-1490. DOI: 10.1016/j.bbadis.2017.07.029. |
| [14] |
VITALE G, GITTO S, VUKOTIC R, et al. Familial intrahepatic cholestasis: New and wide perspectives[J]. Dig Liver Dis, 2019, 51(7): 922-933. DOI: 10.1016/j.dld.2019.04.013. |
| [15] |
PIETERS A, GIJBELS E, COGLIATI B, et al. Biomarkers of cholestasis[J]. Biomark Med, 2021, 15(6): 437-454. DOI: 10.2217/bmm-2020-0691. |
| [16] |
GELLER S, PETOVIC LM. Evaluation of cholestasis//Biopsy interpretation of the liver (2nd edition)[M]. Philadelphia, USA: Lippincott Williams & Wilkins, 2009: 404-416.
|
| [17] |
PAUMGARTNER G, BEUERS U. Ursodeoxycholic acid in cholestatic liver disease: Mechanisms of action and therapeutic use revisited[J]. Hepatology, 2002, 36(3): 525-531. DOI: 10.1053/jhep.2002.36088. |
| [18] |
APPANNA G, KALLIS Y. An update on the management of cholestatic liver diseases[J]. Clin Med (Lond), 2020, 20(5): 513-516. DOI: 10.7861/clinmed.2020-0697. |
| [19] |
|
| [20] |
FABRIS L, FIOROTTO R, SPIRLI C, et al. Pathobiology of inherited biliary diseases: A roadmap to understand acquired liver diseases[J]. Nat Rev Gastroenterol Hepatol, 2019, 16(8): 497-511. DOI: 10.1038/s41575-019-0156-4. |
| [21] |
STAUFER K. Current treatment options for cystic fibrosis-related liver disease[J]. Int J Mol Sci, 2020, 21(22): 8586. DOI: 10.3390/ijms21228586. |
| [22] |
BOЁLLE PY, DEBRAY D, GUILLOT L, et al. Cystic fibrosis liver disease: Outcomes and risk factors in a large cohort of French patients[J]. Hepatology, 2019, 69(4): 1648-1656. DOI: 10.1002/hep.30148. |
| [23] |
LI LT, LI ZD, YANG Y, et al. ABCB11 deficiency presenting as transient neonatal cholestasis: Correlation with genotypes and BSEP expression[J]. Liver Int, 2020, 40(11): 2788-2796. DOI: 10.1111/liv.14642. |
| [24] |
LI L, DEHERAGODA M, LU Y, et al. Hypothyroidism associated with ATP8B1 deficiency[J]. J Pediatr, 2015, 167(6): 1334-1339. e1. DOI: 10.1016/j.jpeds.2015.08.037. |
| [25] |
LI LT, LI ZD, YANG Y, et al. ABCB11 deficiency presenting as transient neonatal cholestasis: Correlation with genotypes and BSEP expression[J]. Liver Int, 2020, 40(11): 2788-2796. DOI: 10.1111/liv.14642. |
| [26] |
van WESSEL D, THOMPSON RJ, GONZALES E, et al. Genotype correlates with the natural history of severe bile salt export pump deficiency[J]. J Hepatol, 2020, 73(1): 84-93. DOI: 10.1016/j.jhep.2020.02.007. |
| [27] |
ZHANG J, LIU LL, GONG JY, et al. TJP2 hepatobiliary disorders: Novel variants and clinical diversity[J]. Hum Mutat, 2020, 41(2): 502-511. DOI: 10.1002/humu.23947. |
| [28] |
QIU YL, GONG JY, FENG JY, et al. Defects in myosin VB are associated with a spectrum of previously undiagnosed low γ-glutamyltransferase cholestasis[J]. Hepatology, 2017, 65(5): 1655-1669. DOI: 10.1002/hep.29020. |
| [29] |
ZHANG J, YANG Y, GONG JY, et al. Low-GGT intrahepatic cholestasis associated with biallelic USP53 variants: Clinical, histological and ultrastructural characterization[J]. Liver Int, 2020, 40(5): 1142-1150. DOI: 10.1111/liv.14422. |
| [30] |
PAN Q, LUO G, QU J, et al. A homozygous R148W mutation in Semaphorin 7A causes progressive familial intrahepatic cholestasis[J]. EMBO Mol Med, 2021, 13(11): e14563. DOI: 10.15252/emmm.202114563. |
| [31] |
GONZALES E, HARDIKAR W, STORMON M, et al. Efficacy and safety of maralixibat treatment in patients with Alagille syndrome and cholestatic pruritus (ICONIC): A randomised phase 2 study[J]. Lancet, 2021, 398(10311): 1581-1592. DOI: 10.1016/S0140-6736(21)01256-3. |
| [32] |
EMERICK KM, RAND EB, GOLDMUNTZ E, et al. Features of Alagille syndrome in 92 patients: Frequency and relation to prognosis[J]. Hepatology, 1999, 29(3): 822-829. DOI: 10.1002/hep.510290331. |
| [33] |
SHNEIDER BL, SPINO C, KAMATH BM, et al. Placebo-controlled randomized trial of an intestinal bile salt transport inhibitor for pruritus in Alagille syndrome[J]. Hepatol Commun, 2018, 2(10): 1184-1198. DOI: 10.1002/hep4.1244. |
| [34] |
FLOREANI A, GERVASI MT. New insights on intrahepatic cholestasis of pregnancy[J]. Clin Liver Dis, 2016, 20(1): 177-189. DOI: 10.1016/j.cld.2015.08.010. |
| [35] |
WOOD AM, LIVINGSTON EG, HUGHES BL, et al. Intrahepatic cholestasis of pregnancy: A review of diagnosis and management[J]. Obstet Gynecol Surv, 2018, 73(2): 103-109. DOI: 10.1097/OGX.0000000000000524. |
| [36] |
|
| [37] |
LANGEDIJK J, BEUERS UH, OUDE ELFERINK R. Cholestasis-associated pruritus and its pruritogens[J]. Front Med (Lausanne), 2021, 8: 639674. DOI: 10.3389/fmed.2021.639674. |
| [38] |
PATEL SP, VASAVDA C, HO B, et al. Cholestatic pruritus: Emerging mechanisms and therapeutics[J]. J Am Acad Dermatol, 2019, 81(6): 1371-1378. DOI: 10.1016/j.jaad.2019.04.035. |
| [39] |
BERGASA NV, JONES EA. Assessment of the visual analogue score in the evaluation of the pruritus of cholestasis[J]. J Clin Transl Hepatol, 2017, 5(3): 203-207. DOI: 10.14218/JCTH.2017.00001. |
| [40] |
THÉBAUT A, HABES D, GOTTRAND F, et al. Sertraline as an additional treatment for cholestatic pruritus in children[J]. J Pediatr Gastroenterol Nutr, 2017, 64(3): 431-435. DOI: 10.1097/MPG.0000000000001385. |
| [41] |
WEBB GJ, RAHMAN SR, LEVY C, et al. Low risk of hepatotoxicity from rifampicin when used for cholestatic pruritus: A cross-disease cohort study[J]. Aliment Pharmacol Ther, 2018, 47(8): 1213-1219. DOI: 10.1111/apt.14579. |
| [42] |
PHAW NA, LEIGHTON J, DYSON JK, et al. Managing cognitive symptoms and fatigue in cholestatic liver disease[J]. Expert Rev Gastroenterol Hepatol, 2021, 15(3): 235-241. DOI: 10.1080/17474124.2021.1844565. |
| [43] |
SWAIN MG, JONES D. Fatigue in chronic liver disease: New insights and therapeutic approaches[J]. Liver Int, 2019, 39(1): 6-19. DOI: 10.1111/liv.13919. |
| [44] |
DUBROVSKY A, BOWLUS CL. Statins, fibrates, and other peroxisome proliferator-activated receptor agonists for the treatment of cholestatic liver diseases[J]. Gastroenterol Hepatol (N Y), 2020, 16(1): 31-38.
|
| [45] |
European Association for the Study of the Liver. EASL clinical practice guidelines on nutrition in chronic liver disease[J]. J Hepatol, 2019, 70(1): 172-193. DOI: 10.1016/j.jhep.2018.06.024. |



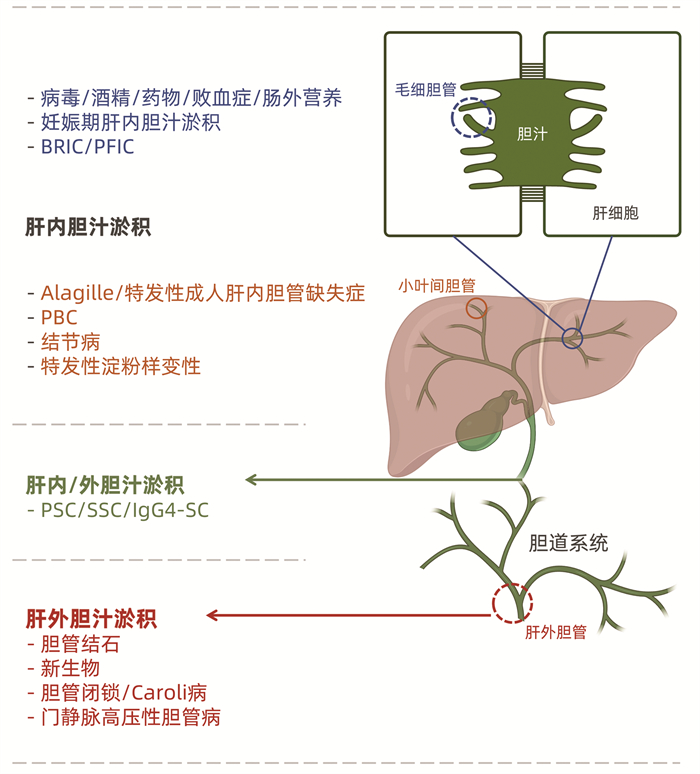




 DownLoad:
DownLoad: