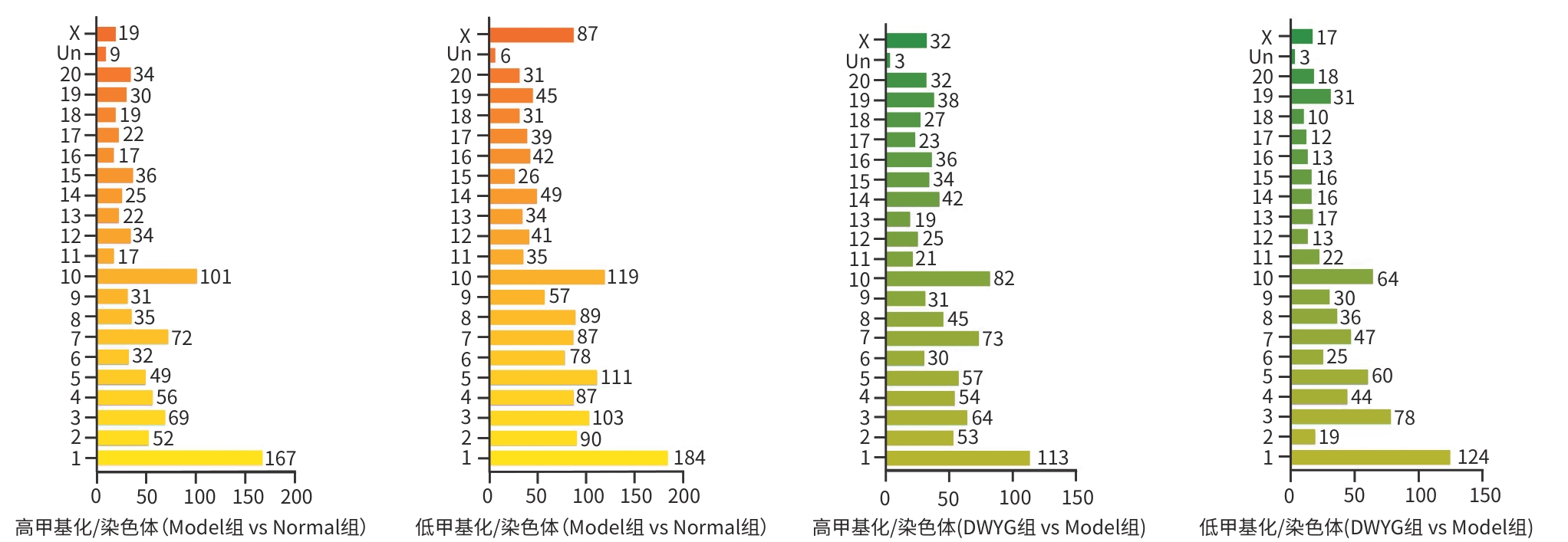
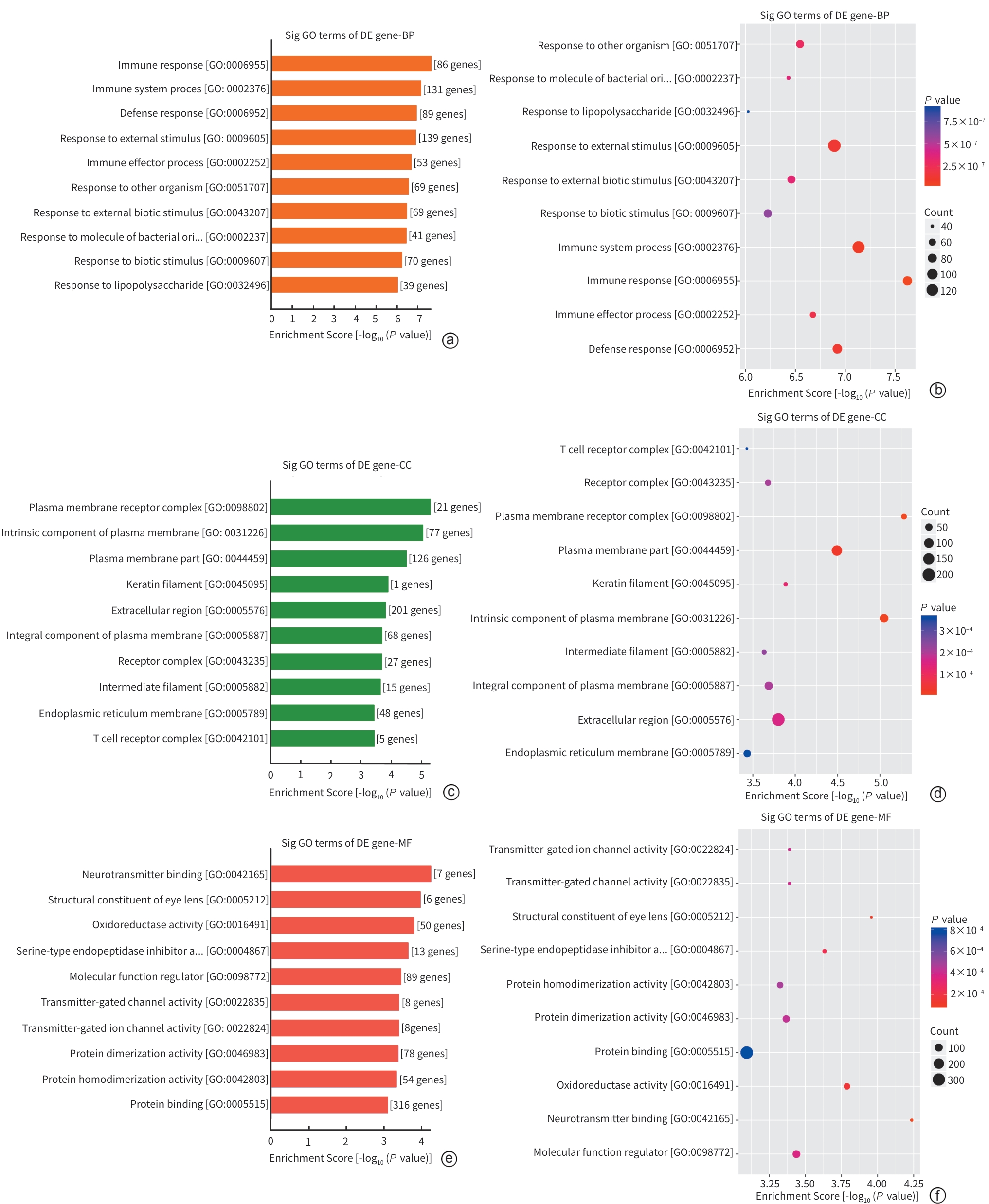
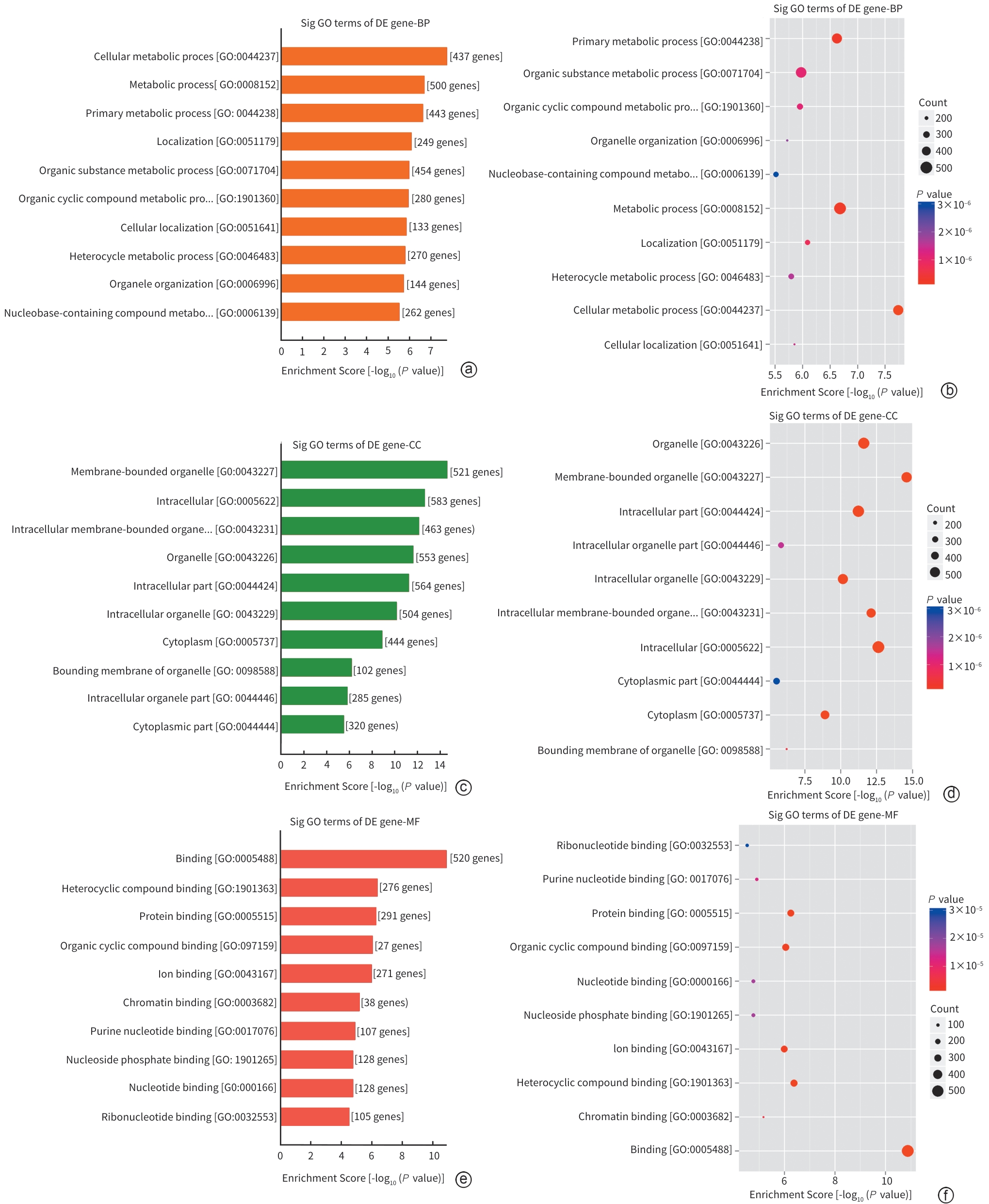
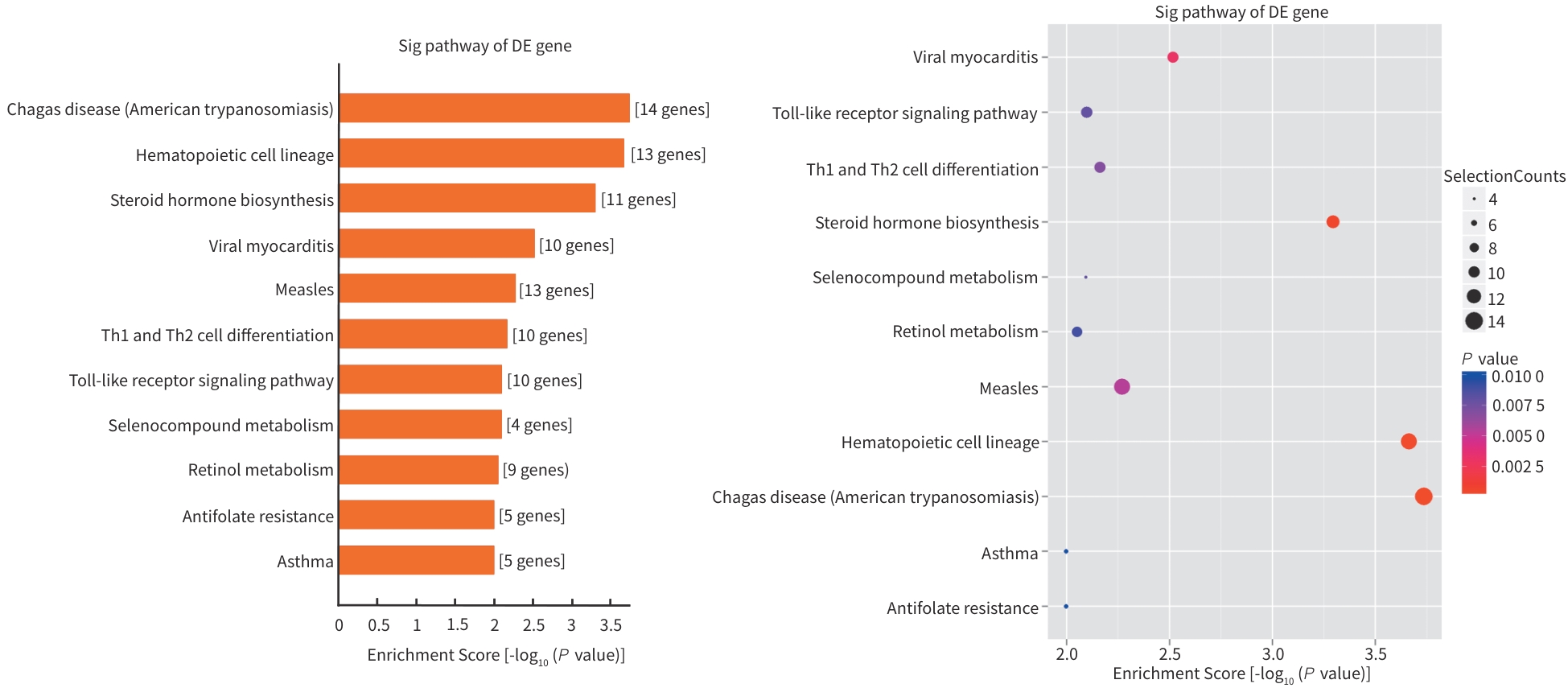
| [1] |
BROWN ZJ, TSILIMIGRAS DI, RUFF SM, et al. Management of hepatocellular carcinoma: A review[J]. JAMA Surg, 2023, 158( 4): 410- 420. DOI: 10.1001/jamasurg.2022.7989. |
| [2] |
SUNG H, FERLAY J, SIEGEL RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2021, 71( 3): 209- 249. DOI: 10.3322/caac.21660. |
| [3] |
ZHOU J, SUN HC, WANG Z, et al. Guidelines for the diagnosis and treatment of primary liver cancer(2022 edition)[J]. Liver Cancer, 2023, 12( 5): 405- 444. DOI: 10.1159/000530495. |
| [4] |
RICH NE, YOPP AC, SINGAL AG. Medical management of hepatocellular carcinoma[J]. J Oncol Pract, 2017, 13( 6): 356- 364. DOI: 10.1200/JOP.2017.022996. |
| [5] |
|
| [6] |
TAHMASEBI BIRGANI M, CARLONI V. Tumor microenvironment, a paradigm in hepatocellular carcinoma progression and therapy[J]. Int J Mol Sci, 2017, 18( 2): 405. DOI: 10.3390/ijms18020405. |
| [7] |
SAS Z, CENDROWICZ E, WEINHÄUSER I, et al. Tumor microenvironment of hepatocellular carcinoma: Challenges and opportunities for new treatment options[J]. Int J Mol Sci, 2022, 23( 7): 3778. DOI: 10.3390/ijms23073778. |
| [8] |
CLEVERS H. The cancer stem cell: Premises, promises and challenges[J]. Nat Med, 2011, 17( 3): 313- 319. DOI: 10.1038/nm.2304. |
| [9] |
WU K, DING J, CHEN C, et al. Hepatic transforming growth factor beta gives rise to tumor-initiating cells and promotes liver cancer development[J]. Hepatology, 2012, 56( 6): 2255- 2267. DOI: 10.1002/hep.26007. |
| [10] |
LI HM, ZHANG LS. Liver regeneration microenvironment of hepatocellular carcinoma for prevention and therapy[J]. Oncotarget, 2017, 8( 1): 1805- 1813. DOI: 10.18632/oncotarget.12101. |
| [11] |
LI HM. Microcirculation of liver cancer, microenvironment of liver regeneration, and the strategy of Chinese medicine[J]. Chin J Integr Med, 2016, 22( 3): 163- 167. DOI: 10.1007/s11655-016-2460-y. |
| [12] |
LI HM, YE ZH. Microenvironment of liver regeneration in liver cancer[J]. Chin J Integr Med, 2017, 23( 7): 555- 560. DOI: 10.1007/s11655-017-2806-0. |
| [13] |
WANG MG, YE QL, MAO DW, et al. Research progress in liver-regenerating microenvironment and DNA methylation in hepatocellular carcinoma: The role of traditional Chinese medicine[J]. Med Sci Monit, 2020, 26: e920310. DOI: 10.12659/MSM.920310. |
| [14] |
LI Y, LI HM, LI ZC, et al. Ingredients, anti-liver cancer effects and the possible mechanism of DWYG formula based on network prediction[J]. Onco Targets Ther, 2020, 13: 4213- 4227. DOI: 10.2147/OTT.S238901. |
| [15] |
LI HM, YE ZH, GAO X, et al. Diwu Yanggan capsule improving liver histological response for patients with HBeAg-negative chronic hepatitis B: A randomized controlled clinical trial[J]. Am J Transl Res, 2018, 10( 5): 1511- 1521.
|
| [16] |
ZHAO BB, YE ZH, GAO X, et al. Diwu Yanggan modulates the Wnt/β-catenin pathway and inhibits liver carcinogenesis signaling in 2-AAF/PH model rats[J]. Curr Med Sci, 2019, 39( 6): 913- 919. DOI: 10.1007/s11596-019-2123-2. |
| [17] |
YE ZH, GAO X, ZHAO BB, et al. Diwu Yanggan capsule inhibits the occurrence and development of liver cancer in the Solt-Farber rat model by regulating the Ras/Raf/Mek/Erk signaling pathway[J]. Am J Transl Res, 2018, 10( 11): 3797- 3805.
|
| [18] |
ZHAO BB, LI HM, GAO X, et al. The herbal compound“Diwu Yanggan” modulates liver regeneration by affecting the hepatic stem cell microenvironment in 2-acetylaminofluorene/partial hepatectomy rats[J]. Evid Based Complement Alternat Med, 2015, 2015: 468303. DOI: 10.1155/2015/468303. |
| [19] |
VIERTHALER M, SUN Q, WANG YM, et al. ADCK2 knockdown affects the migration of melanoma cells via MYL6[J]. Cancers, 2022, 14( 4): 1071. DOI: 10.3390/cancers14041071. |
| [20] |
VÁZQUEZ-FONSECA L, SCHAEFER J, NAVAS-ENAMORADO I, et al. ADCK2 haploinsufficiency reduces mitochondrial lipid oxidation and causes myopathy associated with CoQ deficiency[J]. J Clin Med, 2019, 8( 9): 1374. DOI: 10.3390/jcm8091374. |
| [21] |
ZHANG JZ, LIU J, XU YX, et al. Identification of the mitochondrial protein ADCK2 as a therapeutic oncotarget of NSCLC[J]. Int J Biol Sci, 2022, 18( 16): 6163- 6175. DOI: 10.7150/ijbs.78354. |
| [22] |
HERNÁNDEZ-CAMACHO JD, FERNÁNDEZ-AYALA DJM, VICENTE-GARCÍA C, et al. Calorie restriction rescues mitochondrial dysfunction in Adck2-deficient skeletal muscle[J]. Front Physiol, 2022, 13: 898792. DOI: 10.3389/fphys.2022.898792. |
| [23] |
XU Y, WANG L, ZHANG H, et al. ADCK2 regulates protein phosphorylation and membrane composition in mammalian cells[J]. Biochem Biophys Res Commun, 2020, 529( 4): 987- 993. DOI: 10.1016/j.bbrc.2020.07.054. |
| [24] |
SCHOOLMEESTERS A, BROWN DD, FEDOROV Y. Kinome-wide functional genomics screen reveals a novel mechanism of TNFα-induced nuclear accumulation of the HIF-1α transcription factor in cancer cells[J]. PLoS One, 2012, 7( 2): e31270. DOI: 10.1371/journal.pone.0031270. |







 DownLoad:
DownLoad: