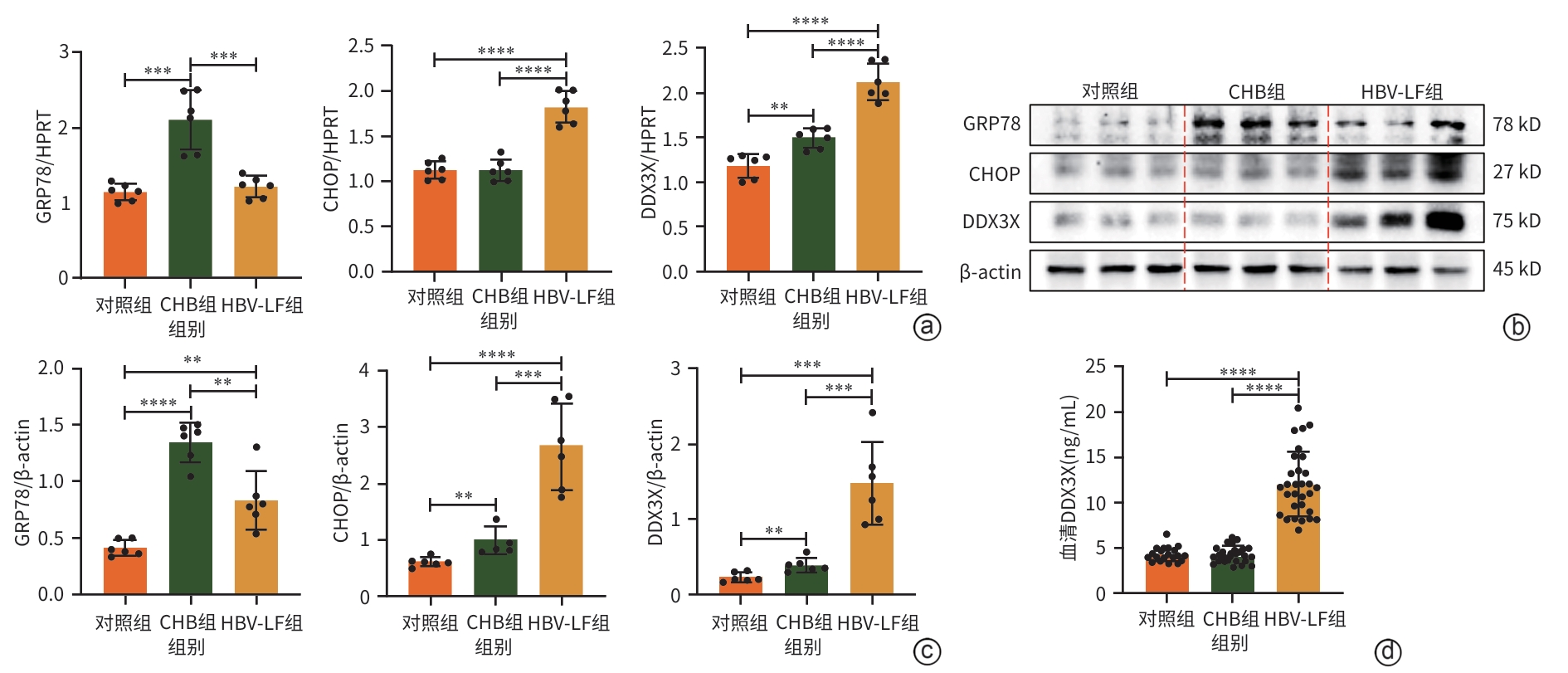
| [1] |
ROBINSON MW, HARMON C, O’FARRELLY C. Liver immunology and its role in inflammation and homeostasis[J]. Cell Mol Immunol, 2016, 13( 3): 267- 276. DOI: 10.1038/cmi.2016.3. |
| [2] |
HOOFNAGLE JH, BJÖRNSSON ES. Drug-induced liver injury-types and phenotypes[J]. N Engl J Med, 2019, 381( 3): 264- 273. DOI: 10.1056/NEJMra1816149. |
| [3] |
LONGHI MS, MA Y, MIELI-VERGANI G, et al. Aetiopathogenesis of autoimmune hepatitis[J]. J Autoimmun, 2010, 34( 1): 7- 14. DOI: 10.1016/j.jaut.2009.08.010. |
| [4] |
ADAMS DH, JU C, RAMAIAH SK, et al. Mechanisms of immune-mediated liver injury[J]. Toxicol Sci, 2010, 115( 2): 307- 321. DOI: 10.1093/toxsci/kfq009. |
| [5] |
NABEKURA T, MATSUO S, SHIBUYA A. Concanavalin-A-induced acute liver injury in mice[J]. Curr Protoc, 2024, 4( 8): e1117. DOI: 10.1002/cpz1.1117. |
| [6] |
TIEGS G, HENTSCHEL J, WENDEL A. A T cell-dependent experimental liver injury in mice inducible by concanavalin A[J]. J Clin Invest, 1992, 90( 1): 196- 203. DOI: 10.1172/JCI115836. |
| [7] |
CUI Y, QIAO FJ, QIU JH, et al. Effect of ganoderic acid A on a mouse model of concanavalin A-induced acute immune liver injury and its mechanism[J]. J Clin Hepatol, 2024, 40( 12): 2415- 2423. DOI: 10.12449/JCH241211. 崔怡, 乔凤杰, 邱嘉昊, 等. 灵芝酸A对刀豆球蛋白A诱导的急性免疫性肝损伤小鼠模型的影响及其作用机制[J]. 临床肝胆病杂志, 2024, 40( 12): 2415- 2423. DOI: 10.12449/JCH241211. |
| [8] |
HETZ C. The unfolded protein response: Controlling cell fate decisions under ER stress and beyond[J]. Nat Rev Mol Cell Biol, 2012, 13( 2): 89- 102. DOI: 10.1038/nrm3270. |
| [9] |
|
| [10] |
LEBEAUPIN C, VALLÉE D, HAZARI Y, et al. Endoplasmic reticulum stress signalling and the pathogenesis of non-alcoholic fatty liver disease[J]. J Hepatol, 2018, 69( 4): 927- 947. DOI: 10.1016/j.jhep.2018.06.008. |
| [11] |
YAN CY, HU WT, TU JQ, et al. Pathogenic mechanisms and regulatory factors involved in alcoholic liver disease[J]. J Transl Med, 2023, 21( 1): 300. DOI: 10.1186/s12967-023-04166-8. |
| [12] |
HU TY, WANG JY, LI WX, et al. Endoplasmic reticulum stress in hepatitis B virus and hepatitis C virus infection[J]. Viruses, 2022, 14( 12): 2630. DOI: 10.3390/v14122630. |
| [13] |
PU SS, PAN YY, ZHANG Q, et al. Endoplasmic reticulum stress and mitochondrial stress in drug-induced liver injury[J]. Molecules, 2023, 28( 7): 3160. DOI: 10.3390/molecules28073160. |
| [14] |
MALHI H, KAUFMAN RJ. Endoplasmic reticulum stress in liver disease[J]. J Hepatol, 2011, 54( 4): 795- 809. DOI: 10.1016/j.jhep.2010.11.005. |
| [15] |
JIANG JL, ZHOU YY, ZHONG WW, et al. Uridine diphosphate glucuronosyltransferase 1A1 prevents the progression of liver injury[J]. World J Gastroenterol, 2024, 30( 9): 1189- 1212. DOI: 10.3748/wjg.v30.i9.1189. |
| [16] |
de BISSCHOP G, AMEUR M, ULRYCK N, et al. HIV-1 gRNA, a biological substrate, uncovers the potency of DDX3X biochemical activity[J]. Biochimie, 2019, 164: 83- 94. DOI: 10.1016/j.biochi.2019.03.008. |
| [17] |
AJAMIAN L, ABEL K, RAO S, et al. HIV-1 recruits UPF1 but excludes UPF2 to promote nucleocytoplasmic export of the genomic RNA[J]. Biomolecules, 2015, 5( 4): 2808- 2839. DOI: 10.3390/biom5042808. |
| [18] |
SAIKRUANG W, YAN PING L ANG, ABE H, et al. The RNA helicase DDX3 promotes IFNB transcription via enhancing IRF-3/p300 holo complex binding to the IFNB promoter[J]. Sci Rep, 2022, 12( 1): 3967. DOI: 10.1038/s41598-022-07876-z. |
| [19] |
LI QS, PÈNE V, KRISHNAMURTHY S, et al. Hepatitis C virus infection activates an innate pathway involving IKK-α in lipogenesis and viral assembly[J]. Nat Med, 2013, 19( 6): 722- 729. DOI: 10.1038/nm.3190. |
| [20] |
RYAN CS, SCHRÖDER M. The human DEAD-box helicase DDX3X as a regulator of mRNA translation[J]. Front Cell Dev Biol, 2022, 10: 1033684. DOI: 10.3389/fcell.2022.1033684. |
| [21] |
HEATON SM, GORRY PR, BORG NA. DExD/H-box helicases in HIV-1 replication and their inhibition[J]. Trends Microbiol, 2023, 31( 4): 393- 404. DOI: 10.1016/j.tim.2022.11.001. |
| [22] |
FOX D, MAN SM. DDX3X: Stressing the NLRP3 inflammasome[J]. Cell Res, 2019, 29( 12): 969- 970. DOI: 10.1038/s41422-019-0250-8. |
| [23] |
LUO TT, YANG SZ, ZHAO TM, et al. Hepatocyte DDX3X protects against drug-induced acute liver injury via controlling stress granule formation and oxidative stress[J]. Cell Death Dis, 2023, 14( 7): 400. DOI: 10.1038/s41419-023-05913-x. |
| [24] |
PAN ZZ, XU L, ZHANG XY, et al. Construction and identification of Ddx3x conditional liver knockout mouse model based on CRISPR/Cas9 combined with Cre-LoxP method[J]. Chin J Gastroenterol Hepatol, 2024, 33( 10): 1275- 1280. DOI: 10.3969/j.issn.1006-5709.2024.10.002. |
| [25] |
SAMIR P, KESAVARDHANA S, PATMORE DM, et al. DDX3X acts as a live-or-die checkpoint in stressed cells by regulating NLRP3 inflammasome[J]. Nature, 2019, 573( 7775): 590- 594. DOI: 10.1038/s41586-019-1551-2. |
| [26] |
KIM SY, KYAW YY, CHEONG J. Functional interaction of endoplasmic reticulum stress and hepatitis B virus in the pathogenesis of liver diseases[J]. World J Gastroenterol, 2017, 23( 43): 7657- 7665. DOI: 10.3748/wjg.v23.i43.7657. |
| [27] |
SUBRAMANIAN K, PAUL S, LIBBY A, et al. HERV1-env induces unfolded protein response activation in autoimmune liver disease: A potential mechanism for regulatory T cell dysfunction[J]. J Immunol, 2023, 210( 6): 732- 744. DOI: 10.4049/jimmunol.2100186. |
| [28] |
OYADOMARI S, MORI M. Roles of CHOP/GADD153 in endoplasmic reticulum stress[J]. Cell Death Differ, 2004, 11( 4): 381- 389. DOI: 10.1038/sj.cdd.4401373. |
| [29] |
ADJIBADE P, GRENIER ST-SAUVEUR V, BERGEMAN J, et al. DDX3 regulates endoplasmic reticulum stress-induced ATF4 expression[J]. Sci Rep, 2017, 7( 1): 13832. DOI: 10.1038/s41598-017-14262-7. |
| [30] |
HAN J, BACK SH, HUR J, et al. ER-stress-induced transcriptional regulation increases protein synthesis leading to cell death[J]. Nat Cell Biol, 2013, 15( 5): 481- 490. DOI: 10.1038/ncb2738. |



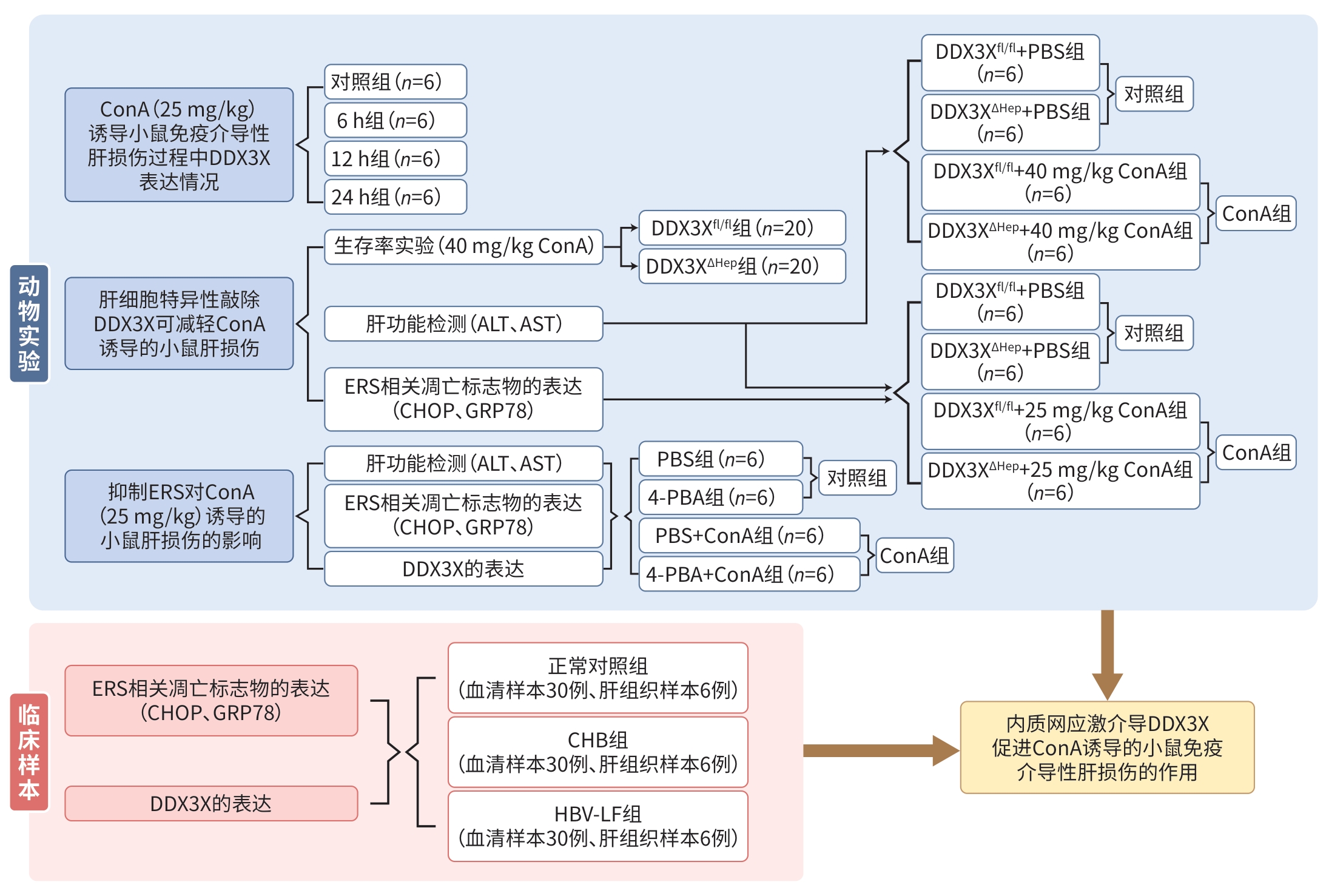




 DownLoad:
DownLoad: