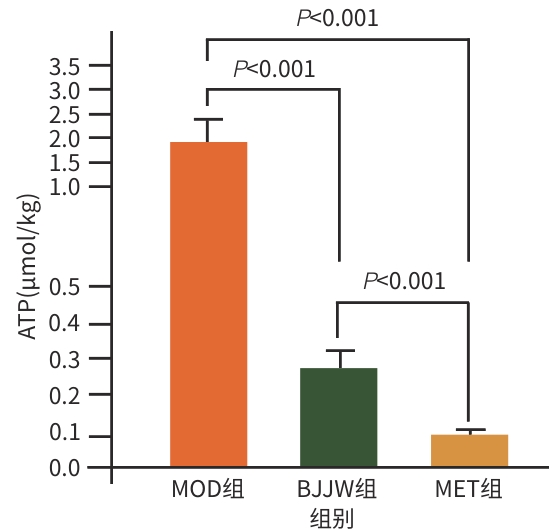
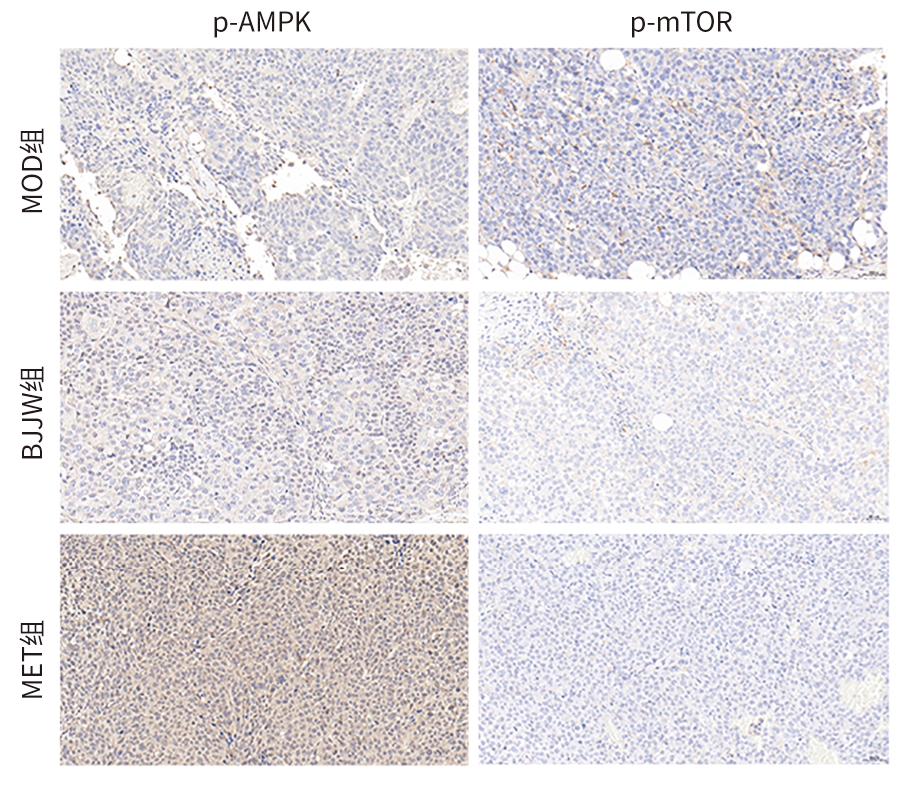
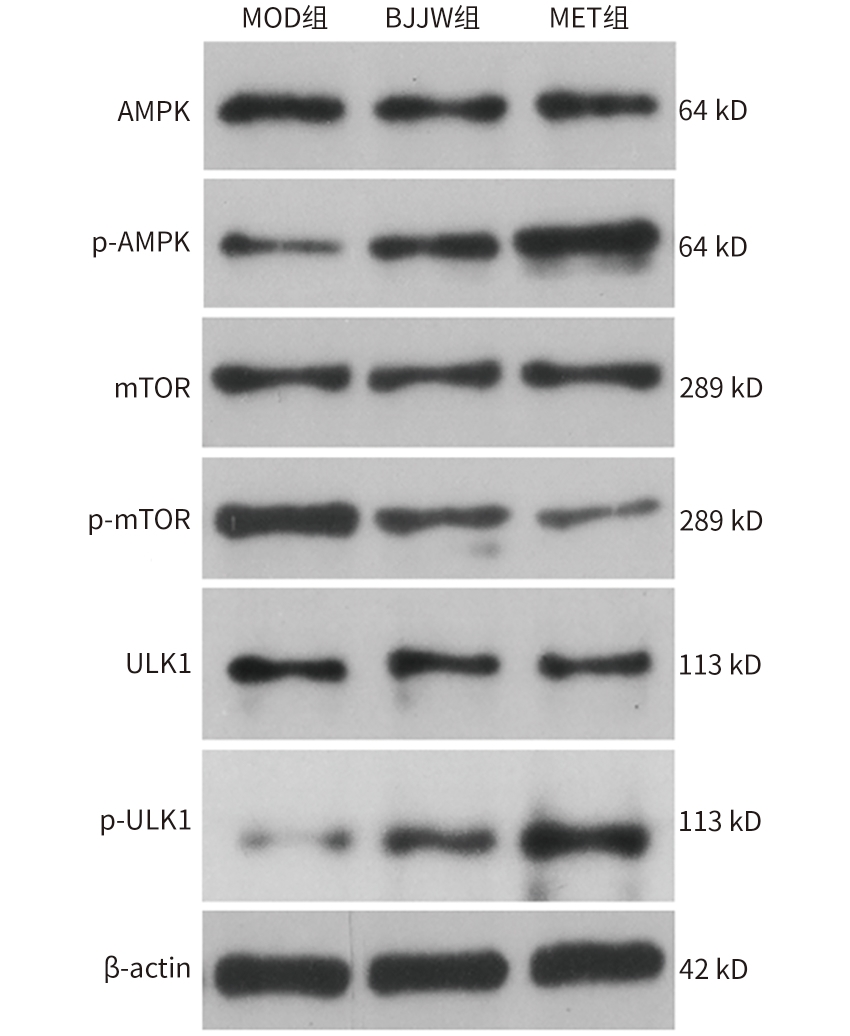
| [1] |
DENG XX, LI H, ZHONG YR, et al. Burden of liver cancer attributable to hepatitis B and alcohol globally, in China, and for five sociodemographic index regions from 1990 to 2021: A population-based study[J]. J Clin Transl Hepatol, 2025, 13( 1): 1- 14. DOI: 10.14218/JCTH.2024.00351. |
| [2] |
ZHOU Y, DING JM, QIN ZY, et al. Predicting the survival rate of patients with hepatocellular carcinoma after thermal ablation by nomograms[J]. Ann Transl Med, 2020, 8( 18): 1159. DOI: 10.21037/atm-20-6116. |
| [3] |
CAO MD, XIA CF, CAO MM, et al. Attributable liver cancer deaths and disability-adjusted life years in China and worldwide: Profiles and changing trends[J]. Cancer Biol Med, 2024, 21( 8): 679- 691. DOI: 10.20892/j.issn.2095-3941.2024.0149. |
| [4] |
LIU Y, ZHENG JX, HAO JL, et al. Global burden of primary liver cancer by five etiologies and global prediction by 2035 based on global burden of disease study 2019[J]. Cancer Med, 2022, 11( 5): 1310- 1323. DOI: 10.1002/cam4.4551. |
| [5] |
YU Z, MAN XW, JIANG Y, et al. Analysis of hospitalization expenses and influencing factors of 5 487 patients with liver cancer in Beijing[J]. Chin J Health Stat, 2020, 37( 5): 748- 751. DOI: 10.3969/j.issn.1002-3674.2020.05.029. |
| [6] |
RANKOVIĆ B, HAUPTMAN N. Circulating microRNA panels for detection of liver cancers and liver-metastasizing primary cancers[J]. Int J Mol Sci, 2023, 24( 20): 15451. DOI: 10.3390/ijms242015451. |
| [7] |
ZENG LL, ZHU LT, FU SS, et al. Mitochondrial dysfunction-molecular mechanisms and potential treatment approaches of hepatocellular carcinoma[J]. Mol Cell Biochem, 2025, 480( 4): 2131- 2142. DOI: 10.1007/s11010-024-05144-4. |
| [8] |
FAUBERT B, SOLMONSON A, DEBERARDINIS RJ. Metabolic reprogramming and cancer progression[J]. Science, 2020, 368( 6487): eaaw5473. DOI: 10.1126/science.aaw5473. |
| [9] |
LI YF, XU JJ, LAN T, et al. Biejiajian Pill inhibits the malignant biological behavior of Hep3B cells through CMTM6[J]. J Xi’an Jiaotong Univ Med Sci, 2025, 46( 3): 522- 531. DOI: 10.7652/jdyxb202503021. 李尹凡, 徐隽婕, 兰涛, 等. 鳖甲煎丸通过CMTM6抑制Hep3B细胞的恶性生物学行为[J]. 西安交通大学学报(医学版), 2025, 46( 3): 522- 531. DOI: 10.7652/jdyxb202503021. |
| [10] |
SHAO FL, CHEN QP, BI Q, et al. Intervention mechanism of Biejiajian Wan on primary liver cancer by regulating lncRNA SNHG5/miRNA-26a-5p/GSK-3β signal axis[J]. Chin J Exp Tradit Med Formulae, 2024, 30( 4): 107- 113. DOI: 10.13422/j.cnki.syfjx.20230730. |
| [11] |
FENG MH, HE SQ, HUANG SZ, et al. Inhibitory effect of Biejiajian pills against diethylnitrosamine-induced hepatocarcinogenesis in rats[J]. J South Med Univ, 2020, 40( 8): 1148- 1154. DOI: 10.12122/j.issn.1673-4254.2020.08.12. |
| [12] |
YU XP, WANG SY, MENG QG, et al. Exploring regulatory mechanism of Biejiajian pill on hepatocellular carcinoma based on bioinformatics[J]. Chin Arch Tradit Chin Med, 2025, 43( 4): 82- 89. DOI: 10.13193/j.issn.1673-7717.2025.04.015. |
| [13] |
|
| [14] |
KUAI Z, CHAO X, HE YT, et al. Metformin attenuates inflammation and boosts autophagy in the liver and intestine of chronologically aged rats[J]. Exp Gerontol, 2023, 184: 112331. DOI: 10.1016/j.exger.2023.112331. |
| [15] |
WANG GF, GAO H, DAI SN, et al. Metformin inhibits neutrophil extracellular traps-promoted pancreatic carcinogenesis in obese mice[J]. Cancer Lett, 2023, 562: 216155. DOI: 10.1016/j.canlet.2023.216155. |
| [16] |
YENMIŞ G, BEŞLI N, YAPRAK SARAÇ E, et al. Metformin promotes apoptosis in primary breast cancer cells by downregulation of cyclin D1 and upregulation of P53 through an AMPK-alpha independent mechanism[J]. Turk J Med Sci, 2021, 51( 2): 826- 834. DOI: 10.3906/sag-1908-112. |
| [17] |
DAS BK, KNOTT RM, GADAD PC. Metformin and asarone inhibit HepG2 cell proliferation in a high glucose environment by regulating AMPK and Akt signaling pathway[J]. Future J Pharm Sci, 2021, 7( 1): 43. DOI: 10.1186/s43094-021-00193-8. |
| [18] |
LUO S, SCHOOLING CM, WONG ICK, et al. Evaluating the impact of AMPK activation, a target of metformin, on risk of cardiovascular diseases and cancer in the UK Biobank: A Mendelian randomisation study[J]. Diabetologia, 2020, 63( 11): 2349- 2358. DOI: 10.1007/s00125-020-05243-z. |
| [19] |
TAN QW, XU J, ZHU RH, et al. Effects of Biejia Jianwan on the tumorigenicity and dryness of liver cancer stem cells by regulating miR-140[J]. Jilin J Chin Med, 2024, 44( 1): 90- 95. DOI: 10.13463/j.cnki.jlzyy.2024.01.021. |
| [20] |
TAN QW, HUANG JJ, ZHONG RX, et al. Effect of Biejia Decoction Pill on aerobic glycolysis in hepatocellular carcinoma by regulating the protein kinase B/mammalian target of rapamycin signaling pathway[J]. J Clin Hepatol, 2025, 41( 2): 300- 306. DOI: 10.12449/JCH250216. 谭钦文, 黄晶晶, 钟瑞熙, 等. 鳖甲煎丸调控AKT/mTOR信号通路在肝癌细胞有氧糖酵解中的作用[J]. 临床肝胆病杂志, 2025, 41( 2): 300- 306. DOI: 10.12449/JCH250216. |
| [21] |
LIN HS, TAN JN, FANG QL, et al. Analysis of the circRNA expression profile in hepatocellular carcinoma cells inhibited by Biejia Jianwan[J]. Genom Appl Biol, 2025, 44( 5): 511- 519. DOI: 10.13417/j.gab.044.000511. |
| [22] |
CHEN WG, HE CY, WEN B, et al. Biejiajian pill regulates ferroptosis in hepatocellular carcinoma cells via p62/Keap1/NRF2 signaling pathway: A mechanism study[J]. J Sichuan Univ Med Sci, 2025, 56( 1): 51- 58. DOI: 10.12182/20250160502. 陈伟光, 何春雨, 文彬, 等. 鳖甲煎丸通过p62/Keap1/NRF2信号通路调控肝癌细胞铁死亡的作用机制研究[J]. 四川大学学报(医学版), 2025, 56( 1): 51- 58. DOI: 10.12182/20250160502. |
| [23] |
SUN JL, WEN B, YANG XM, et al. Mechanism research of regulation of proliferation and metastasis of hepatoma cells Hep3B by Biejiajian Pill based on PI3K/AKT/GSK-3β signaling pathway[J]. China J Tradit Chin Med Pharm, 2021, 36( 3): 1361- 1365.
孙嘉玲, 文彬, 杨雪梅, 等. 基于PI3K/AKT/GSK-3β信号通路探讨鳖甲煎丸调控肝癌细胞Hep3B增殖转移的机制[J]. 中华中医药杂志, 2021, 36( 3): 1361- 1365.
|
| [24] |
HUANG JJ, HUANG HN, WANG XJ, et al. Bie Jia Jian pill enhances the amelioration of bone mesenchymal stem cells on hepatocellular carcinoma progression[J]. J Nat Med, 2022, 76( 1): 49- 58. DOI: 10.1007/s11418-021-01548-4. |
| [25] |
WANG WL, SUN Q, WU ZH, et al. Mitochondrial dysfunction-related genes in hepatocellular carcinoma[J]. Front Biosci, 2013, 18( 3): 1141- 1149. DOI: 10.2741/4169. |
| [26] |
LEE HY, NGA HT, TIAN JW, et al. Mitochondrial metabolic signatures in hepatocellular carcinoma[J]. Cells, 2021, 10( 8): 1901. DOI: 10.3390/cells10081901. |
| [27] |
van der BLIEK AM, SEDENSKY MM, MORGAN PG. Cell biology of the mitochondrion[J]. Genetics, 2017, 207( 3): 843- 871. DOI: 10.1534/genetics.117.300262. |
| [28] |
HU CX, HUANG Y, LI LJ. Drp1-dependent mitochondrial fission plays critical roles in physiological and pathological progresses in mammals[J]. Int J Mol Sci, 2017, 18( 1): 144. DOI: 10.3390/ijms18010144. |
| [29] |
SHADEL GS, HORVATH TL. Mitochondrial ROS signaling in organismal homeostasis[J]. Cell, 2015, 163( 3): 560- 569. DOI: 10.1016/j.cell.2015.10.001. |
| [30] |
TIAN CW, LIU YF, LI ZS, et al. Mitochondria related cell death modalities and disease[J]. Front Cell Dev Biol, 2022, 10: 832356. DOI: 10.3389/fcell.2022.832356. |
| [31] |
HSU CC, PENG DN, CAI Z, et al. AMPK signaling and its targeting in cancer progression and treatment[J]. Semin Cancer Biol, 2022, 85: 52- 68. DOI: 10.1016/j.semcancer.2021.04.006. |
| [32] |
TREFTS E, SHAW RJ. AMPK: Restoring metabolic homeostasis over space and time[J]. Mol Cell, 2021, 81( 18): 3677- 3690. DOI: 10.1016/j.molcel.2021.08.015. |
| [33] |
FANG GX, ZHANG PL, LIU JF, et al. Inhibition of GSK-3β activity suppresses HCC malignant phenotype by inhibiting glycolysis via activating AMPK/mTOR signaling[J]. Cancer Lett, 2019, 463: 11- 26. DOI: 10.1016/j.canlet.2019.08.003. |
| [34] |
LIU YF, XU YY, WANG F, et al. Inhibition of AMPK activity by TRIM11 facilitates cell survival of hepatocellular carcinoma under metabolic stress[J]. Clin Transl Med, 2021, 11( 12): e617. DOI: 10.1002/ctm2.617. |
| [35] |
MENG SS, GU HW, ZHANG T, et al. Gradual deterioration of fatty liver disease to liver cancer via inhibition of AMPK signaling pathways involved in energy-dependent disorders, cellular aging, and chronic inflammation[J]. Front Oncol, 2023, 13: 1099624. DOI: 10.3389/fonc.2023.1099624. |
| [36] |
JUNG TY, RYU JE, JANG MM, et al. Naa20, the catalytic subunit of NatB complex, contributes to hepatocellular carcinoma by regulating the LKB1-AMPK-mTOR axis[J]. Exp Mol Med, 2020, 52( 11): 1831- 1844. DOI: 10.1038/s12276-020-00525-3. |
| [37] |
ZHOU XJ, CHEN Y, WANG FF, et al. Artesunate induces autophagy dependent apoptosis through upregulating ROS and activating AMPK-mTOR-ULK1 axis in human bladder cancer cells[J]. Chem Biol Interact, 2020, 331: 109273. DOI: 10.1016/j.cbi.2020.109273. |
| [38] |
JIA L, LIN XR, GUO WY, et al. Salvia chinensia Benth induces autophagy in esophageal cancer cells via AMPK/ULK1 signaling pathway[J]. Front Pharmacol, 2022, 13: 995344. DOI: 10.3389/fphar.2022.995344. |
| [39] |
NAIK PP, MUKHOPADHYAY S, PRAHARAJ PP, et al. Secretory clusterin promotes oral cancer cell survival via inhibiting apoptosis by activation of autophagy in AMPK/mTOR/ULK1 dependent pathway[J]. Life Sci, 2021, 264: 118722. DOI: 10.1016/j.lfs.2020.118722. |
| [40] |
WANG CM, CIGLIANO A, JIANG LJ, et al. 4EBP1/eIF4E and p70S6K/RPS6 axes play critical and distinct roles in hepatocarcinogenesis driven by AKT and N-Ras proto-oncogenes in mice[J]. Hepatology, 2015, 61( 1): 200- 213. DOI: 10.1002/hep.27396. |
| [41] |
XU M, WANG ZJ, WU ZS, et al. Autophagy activated by the AMPK/mTOR/ULK1 pathway involves AURKB-mediated microgliosis in neuropathic pain[J]. Brain Behav Immun, 2025, 129: 948- 959. DOI: 10.1016/j.bbi.2025.07.019. |



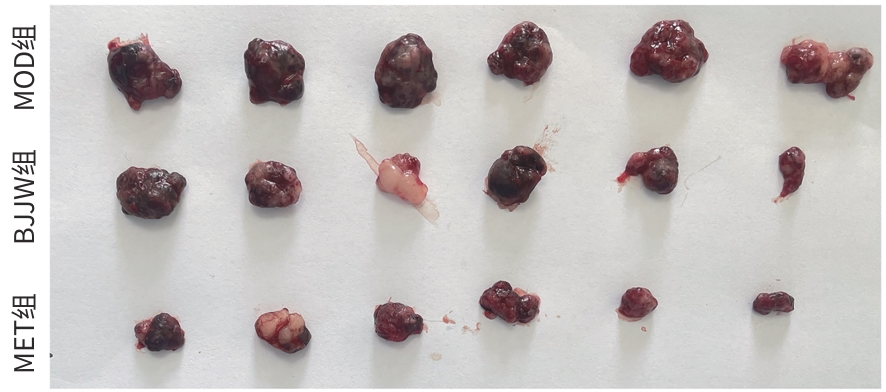




 DownLoad:
DownLoad: