| [1] |
SCHWEITZER A, HORN J, MIKOLAJCZYK RT, et al. Estimations of worldwide prevalence of chronic hepatitis B virus infection: A systematic review of data published between 1965 and 2013[J]. Lancet, 2015, 386( 10003): 1546- 1555. DOI: 10.1016/S0140-6736(15)61412-X. |
| [2] |
NASSAL M. HBV cccDNA: Viral persistence reservoir and key obstacle for a cure of chronic hepatitis B[J]. Gut, 2015, 64( 12): 1972- 1984. DOI: 10.1136/gutjnl-2015-309809. |
| [3] |
YAN H, ZHONG GC, XU GW, et al. Sodium taurocholate cotransporting polypeptide is a functional receptor for human hepatitis B and D virus[J]. eLife, 2012, 1: e00049. DOI: 10.7554/eLife.00049. |
| [4] |
XIA YC, GUO HT. Hepatitis B virus cccDNA: Formation, regulation and therapeutic potential[J]. Antiviral Res, 2020, 180: 104824. DOI: 10.1016/j.antiviral.2020.104824. |
| [5] |
ZHAO KT, WANG JJ, WANG ZC, et al. Hepatitis B virus hijacks MRE11-RAD50-NBS1 complex to form its minichromosome[J]. PLoS Pathog, 2025, 21( 1): e1012824. DOI: 10.1371/journal.ppat.1012824. |
| [6] |
XIA YC, CHENG XM, NILSSON T, et al. Nucleolin binds to and regulates transcription of hepatitis B virus covalently closed circular DNA minichromosome[J]. Proc Natl Acad Sci USA, 2023, 120( 49): e2306390120. DOI: 10.1073/pnas.2306390120. |
| [7] |
TENG Y, XU ZC, ZHAO KT, et al. Novel function of SART1 in HNF4α transcriptional regulation contributes to its antiviral role during HBV infection[J]. J Hepatol, 2021, 75( 5): 1072- 1082. DOI: 10.1016/j.jhep.2021.06.038. |
| [8] |
WANG JJ, HUANG HY, ZHAO KT, et al. G-quadruplex in hepatitis B virus pregenomic RNA promotes its translation[J]. J Biol Chem, 2023, 299( 9): 105151. DOI: 10.1016/j.jbc.2023.105151. |
| [9] |
ZHENG YC, WANG MF, LI ST, et al. Hepatitis B virus hijacks TSG101 to facilitate egress via multiple vesicle bodies[J]. PLoS Pathog, 2023, 19( 5): e1011382. DOI: 10.1371/journal.ppat.1011382. |
| [10] |
ZHAO KT, GUO FT, WANG JJ, et al. Limited disassembly of cytoplasmic hepatitis B virus nucleocapsids restricts viral infection in murine hepatic cells[J]. Hepatology, 2023, 77( 4): 1366- 1381. DOI: 10.1002/hep.32622. |
| [11] |
LEMPP FA, WIEDTKE E, QU BQ, et al. Sodium taurocholate cotransporting polypeptide is the limiting host factor of hepatitis B virus infection in macaque and pig hepatocytes[J]. Hepatology, 2017, 66( 3): 703- 716. DOI: 10.1002/hep.29112. |
| [12] |
XU ZC, ZHAO KT, WANG JJ, et al. Screening of different species reveals cat hepatocytes support HBV infection[J]. PLoS Pathog, 2025, 21( 8): e1013390. DOI: 10.1371/journal.ppat.1013390. |
| [13] |
CHEN FW, WETTENGEL JM, GEGENFURTNER F, et al. Identification of NTCP animal orthologs supporting hepatitis B virus binding and infection[J]. J Virol, 2025, 99( 4): e01833-24. DOI: 10.1128/jvi.01833-24. |
| [14] |
ASABE S, WIELAND SF, CHATTOPADHYAY PK, et al. The size of the viral inoculum contributes to the outcome of hepatitis B virus infection[J]. J Virol, 2009, 83( 19): 9652- 9662. DOI: 10.1128/JVI.00867-09. |
| [15] |
WIELAND SF, SPANGENBERG HC, THIMME R, et al. Expansion and contraction of the hepatitis B virus transcriptional template in infected chimpanzees[J]. Proc Natl Acad Sci USA, 2004, 101( 7): 2129- 2134. DOI: 10.1073/pnas.0308478100. |
| [16] |
GUIDOTTI LG, ROCHFORD R, CHUNG J, et al. Viral clearance without destruction of infected cells during acute HBV infection[J]. Science, 1999, 284( 5415): 825- 829. DOI: 10.1126/science.284.5415.825. |
| [17] |
KOMIYA Y, KATAYAMA K, YUGI H, et al. Minimum infectious dose of hepatitis B virus in chimpanzees and difference in the dynamics of viremia between genotype A and genotype C[J]. Transfusion, 2008, 48( 2): 286- 294. DOI: 10.1111/j.1537-2995.2007.01522.x. |
| [18] |
BURWITZ BJ, WETTENGEL JM, MÜCK-HÄUSL MA, et al. Hepatocytic expression of human sodium-taurocholate cotransporting polypeptide enables hepatitis B virus infection of macaques[J]. Nat Commun, 2017, 8( 1): 2146. DOI: 10.1038/s41467-017-01953-y. |
| [19] |
ZHOU M, QIN B, DENG XS, et al. hNTCP-expressing primary pig hepatocytes are a valuable tool for investigating hepatitis B virus infection and antiviral drugs[J]. Mol Med Rep, 2019, 20( 4): 3820- 3828. DOI: 10.3892/mmr.2019.10628. |
| [20] |
ZHANG H, LIU YN, LIU CD, et al. The feasibility of establishing a Hamster model for HBV infection: In vitro evidence[J]. mBio, 2024, 15( 11): e02615-24. DOI: 10.1128/mbio.02615-24. |
| [21] |
YAN H, PENG B, HE WH, et al. Molecular determinants of hepatitis B and D virus entry restriction in mouse sodium taurocholate cotransporting polypeptide[J]. J Virol, 2013, 87( 14): 7977- 7991. DOI: 10.1128/JVI.03540-12. |
| [22] |
WANG Q, SCHWARZENBERGER P, YANG F, et al. Experimental chronic hepatitis B infection of neonatal tree shrews( Tupaia belangeri Chinensis): A model to study molecular causes for susceptibility and disease progression to chronic hepatitis in humans[J]. Virol J, 2012, 9: 170. DOI: 10.1186/1743-422X-9-170. |
| [23] |
KÖCK J, NASSAL M, MACNELLY S, et al. Efficient infection of primary Tupaia Hepatocytes with purified human and woolly monkey hepatitis B virus[J]. J Virol, 2001, 75( 11): 5084- 5089. DOI: 10.1128/JVI.75.11.5084-5089.2001. |
| [24] |
GHEIT T, SEKKAT S, COVA L, et al. Experimental transfection of Macaca sylvanus with cloned human hepatitis B virus[J]. J Gen Virol, 2002, 83( Pt 7): 1645- 1649. DOI: 10.1099/0022-1317-83-7-1645. |
| [25] |
MÜLLER SF, KÖNIG A, DÖRING B, et al. Characterisation of the hepatitis B virus cross-species transmission pattern via Na +/taurocholate co-transporting polypeptides from 11 New World and Old World primate species[J]. PLoS One, 2018, 13( 6): e0199200. DOI: 10.1371/journal.pone.0199200. |
| [26] |
BISWAS S, RUST LN, WETTENGEL JM, et al. Long-term hepatitis B virus infection of Rhesus macaques requires suppression of host immunity[J]. Nat Commun, 2022, 13( 1): 2995. DOI: 10.1038/s41467-022-30593-0. |
| [27] |
RUST LN, WETTENGEL JM, BISWAS S, et al. Liver-specific transgenic expression of human NTCP in Rhesus macaques confers HBV susceptibility on primary hepatocytes[J]. Proc Natl Acad Sci USA, 2025, 122( 7): e2413771122. DOI: 10.1073/pnas.2413771122. |
| [28] |
HE WH, REN BJ, MAO FF, et al. Hepatitis D virus infection of mice expressing human sodium taurocholate co-transporting polypeptide[J]. PLoS Pathog, 2015, 11( 4): e1004840. DOI: 10.1371/journal.ppat.1004840. |
| [29] |
GUIDOTTI LG, MATZKE B, SCHALLER H, et al. High-level hepatitis B virus replication in transgenic mice[J]. J Virol, 1995, 69( 10): 6158- 6169. DOI: 10.1128/JVI.69.10.6158-6169.1995. |
| [30] |
YANG PL, ALTHAGE A, CHUNG J, et al. Hydrodynamic injection of viral DNA: A mouse model of acute hepatitis B virus infection[J]. Proc Natl Acad Sci USA, 2002, 99( 21): 13825- 13830. DOI: 10.1073/pnas.202398599. |
| [31] |
SPRENGERS D, VAN DER MOLEN RG, KUSTERS JG, et al. Analysis of intrahepatic HBV-specific cytotoxic T-cells during and after acute HBV infection in humans[J]. J Hepatol, 2006, 45( 2): 182- 189. DOI: 10.1016/j.jhep.2005.12.022. |
| [32] |
YANG D, LIU LC, ZHU DM, et al. A mouse model for HBV immunotolerance and immunotherapy[J]. Cell Mol Immunol, 2014, 11( 1): 71- 78. DOI: 10.1038/cmi.2013.43. |
| [33] |
KO C, SU JP, FESTAG J, et al. Intramolecular recombination enables the formation of hepatitis B virus(HBV) cccDNA in mice after HBV genome transfer using recombinant AAV vectors[J]. Antiviral Res, 2021, 194: 105140. DOI: 10.1016/j.antiviral.2021.105140. |
| [34] |
QI ZH, LI GY, HU H, et al. Recombinant covalently closed circular hepatitis B virus DNA induces prolonged viral persistence in immunocompetent mice[J]. J Virol, 2014, 88( 14): 8045- 8056. DOI: 10.1128/JVI.01024-14. |
| [35] |
MA KAY, HE CY, CHEN ZY. A robust system for production of minicircle DNA vectors[J]. Nat Biotechnol, 2010, 28( 12): 1287- 1289. DOI: 10.1038/nbt.1708. |
| [36] |
ZHOU ZM, LI C, TAN ZX, et al. A spatiotemporally controlled recombinant cccDNA mouse model for studying HBV and developing drugs against the virus[J]. Antiviral Res, 2023, 216: 105642. DOI: 10.1016/j.antiviral.2023.105642. |
| [37] |
XU ZC, ZHAO L, ZHONG YQ, et al. A novel mouse model harboring hepatitis B virus covalently closed circular DNA[J]. Cell Mol Gastroenterol Hepatol, 2022, 13( 4): 1001- 1017. DOI: 10.1016/j.jcmgh.2021.11.011. |
| [38] |
BILITY MT, ZHANG LG, WASHBURN ML, et al. Generation of a humanized mouse model with both human immune system and liver cells to model hepatitis C virus infection and liver immunopathogenesis[J]. Nat Protoc, 2012, 7( 9): 1608- 1617. DOI: 10.1038/nprot.2012.083. |
| [39] |
WASHBURN ML, BILITY MT, ZHANG LG, et al. A humanized mouse model to study hepatitis C virus infection, immune response, and liver disease[J]. Gastroenterology, 2011, 140( 4): 1334- 1344. DOI: 10.1053/j.gastro.2011.01.001. |
| [40] |
JESKE SD, WETTENGEL JM, GEGENFURTNER F, et al. Identification of amino acids restricting HBV receptor function in porcine NTCP[J]. NPJ Viruses, 2024, 2: 30. DOI: 10.1038/s44298-024-00041-5. |
| [41] |
DUPINAY T, GHEIT T, ROQUES P, et al. Discovery of naturally occurring transmissible chronic hepatitis B virus infection among Macaca fascicularis from Mauritius Island[J]. Hepatology, 2013, 58( 5): 1610- 1620. DOI: 10.1002/hep.26428. |
| [42] |
LANFORD RE, CHAVEZ D, BRASKY KM, et al. Isolation of a hepadnavirus from the woolly monkey, a New World primate[J]. Proc Natl Acad Sci USA, 1998, 95( 10): 5757- 5761. DOI: 10.1073/pnas.95.10.5757. |
| [43] |
CHEN CY, WINER BY, CHAVEZ D, et al. Woolly monkey-HBV infection in squirrel monkeys as a surrogate nonhuman primate model of HBV infection[J]. Hepatol Commun, 2020, 4( 3): 371- 386. DOI: 10.1002/hep4.1471. |
| [44] |
LIU YZ, CAFIERO TR, PARK D, et al. Targeted viral adaptation generates a Simian-tropic hepatitis B virus that infects marmoset cells[J]. Nat Commun, 2023, 14: 3582. DOI: 10.1038/s41467-023-39148-3. |
| [45] |
de CARVALHO DOMINGUEZ SOUZA BF, KÖNIG A, RASCHE A, et al. A novel hepatitis B virus species discovered in capuchin monkeys sheds new light on the evolution of primate hepadnaviruses[J]. J Hepatol, 2018, 68( 6): 1114- 1122. DOI: 10.1016/j.jhep.2018.01.029. |
| [46] |
SUMMERS J, SMOLEC JM, SNYDER R. A virus similar to human hepatitis B virus associated with hepatitis and hepatoma in woodchucks[J]. Proc Natl Acad Sci USA, 1978, 75( 9): 4533- 4537. DOI: 10.1073/pnas.75.9.4533. |
| [47] |
DANDRI M, BURDA MR, WILL H, et al. Increased hepatocyte turnover and inhibition of woodchuck hepatitis B virus replication by adefovir in vitro do not lead to reduction of the closed circular DNA[J]. Hepatology, 2000, 32( 1): 139- 146. DOI: 10.1053/jhep.2000.8701. |
| [48] |
MENNE S, TUMAS DB, LIU KH, et al. Sustained efficacy and seroconversion with the Toll-like receptor 7 agonist GS-9620 in the Woodchuck model of chronic hepatitis B[J]. J Hepatol, 2015, 62( 6): 1237- 1245. DOI: 10.1016/j.jhep.2014.12.026. |
| [49] |
MASON WS, SEAL G, SUMMERS J. Virus of Pekin ducks with structural and biological relatedness to human hepatitis B virus[J]. J Virol, 1980, 36( 3): 829- 836. DOI: 10.1128/JVI.36.3.829-836.1980. |
| [50] |
|
| [51] |
FUNK A, MHAMDI M, WILL H, et al. Avian hepatitis B viruses: Molecular and cellular biology, phylogenesis, and host tropism[J]. World J Gastroenterol, 2007, 13( 1): 91- 103. DOI: 10.3748/wjg.v13.i1.91. |
| [52] |
AGHAZADEH M, SHI M, BARRS VR, et al. A novel hepadnavirus identified in an immunocompromised domestic cat in Australia[J]. Viruses, 2018, 10( 5): 269. DOI: 10.3390/v10050269. |
| [53] |
LANAVE G, CAPOZZA P, DIAKOUDI G, et al. Identification of hepadnavirus in the sera of cats[J]. Sci Rep, 2019, 9( 1): 10668. DOI: 10.1038/s41598-019-47175-8. |
| [54] |
SHOFA M, OHKAWA A, KANEKO Y, et al. Conserved use of the sodium/bile acid cotransporter(NTCP) as an entry receptor by hepatitis B virus and domestic cat hepadnavirus[J]. Antiviral Res, 2023, 217: 105695. DOI: 10.1016/j.antiviral.2023.105695. |
| [55] |
SHOFA M, FUKUSHIMA YV, SAITO A. Conserved yet divergent Smc5/6 complex degradation by mammalian hepatitis B virus X proteins[J]. Int J Mol Sci, 2025, 26( 14): 6786. DOI: 10.3390/ijms26146786. |
| [56] |
CAVASIN JP, CHEN MC, AJOYAN H, et al. Recurrent integration of domestic cat hepatitis B virus DNA near feline CCNE1 supports an oncogenic role in hepatocellular carcinoma in cats[J]. Tumour Virus Res, 2025, 20: 200324. DOI: 10.1016/j.tvr.2025.200324. |



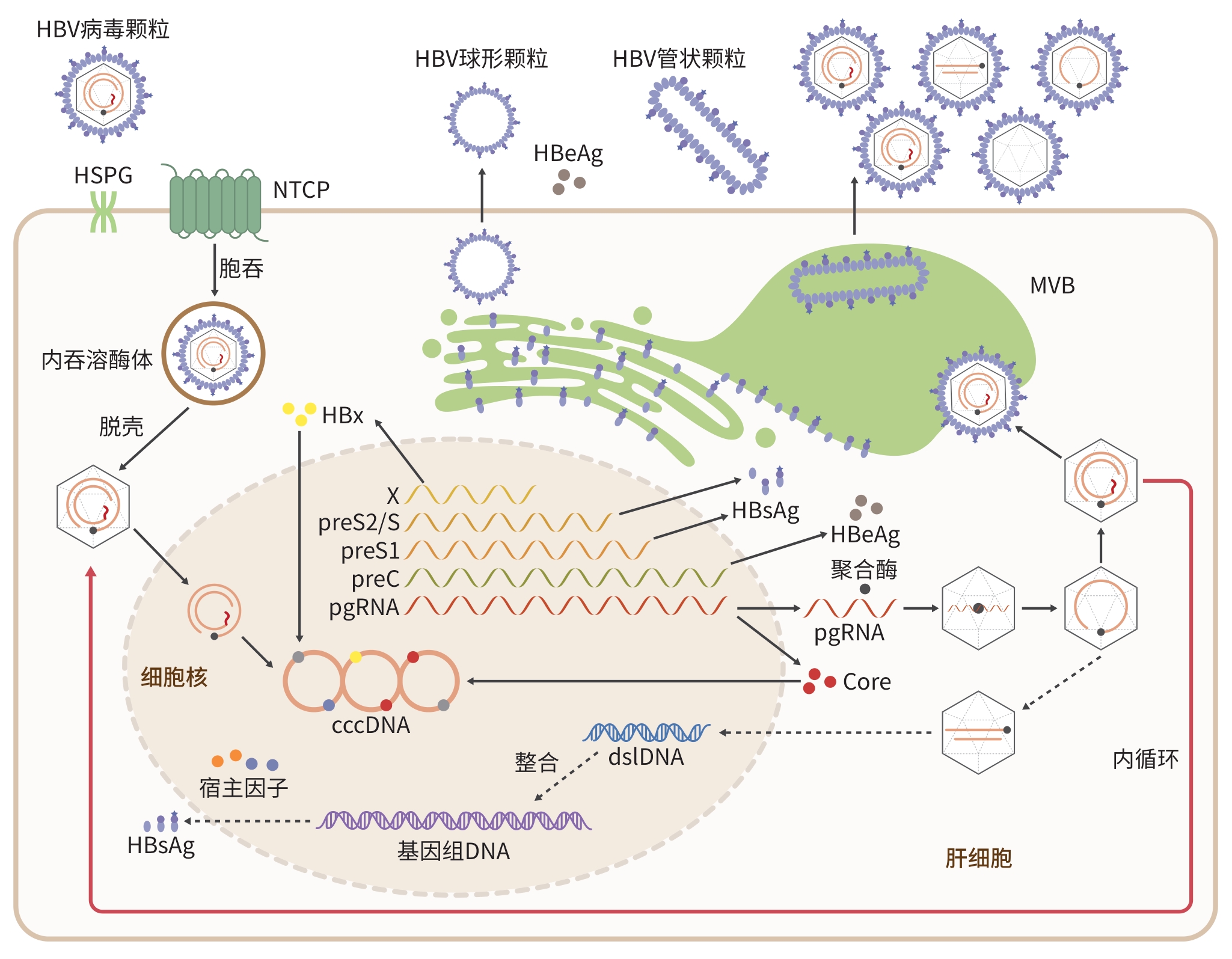




 DownLoad:
DownLoad: