| [1] |
YAN RY, SUN MH, YANG HY, et al. 2024 latest report on hepatitis B virus epidemiology in China: Current status, changing trajectory, and challenges[J]. Hepatobiliary Surg Nutr, 2025, 14( 1): 66- 77. DOI: 10.21037/hbsn-2024-754. |
| [2] |
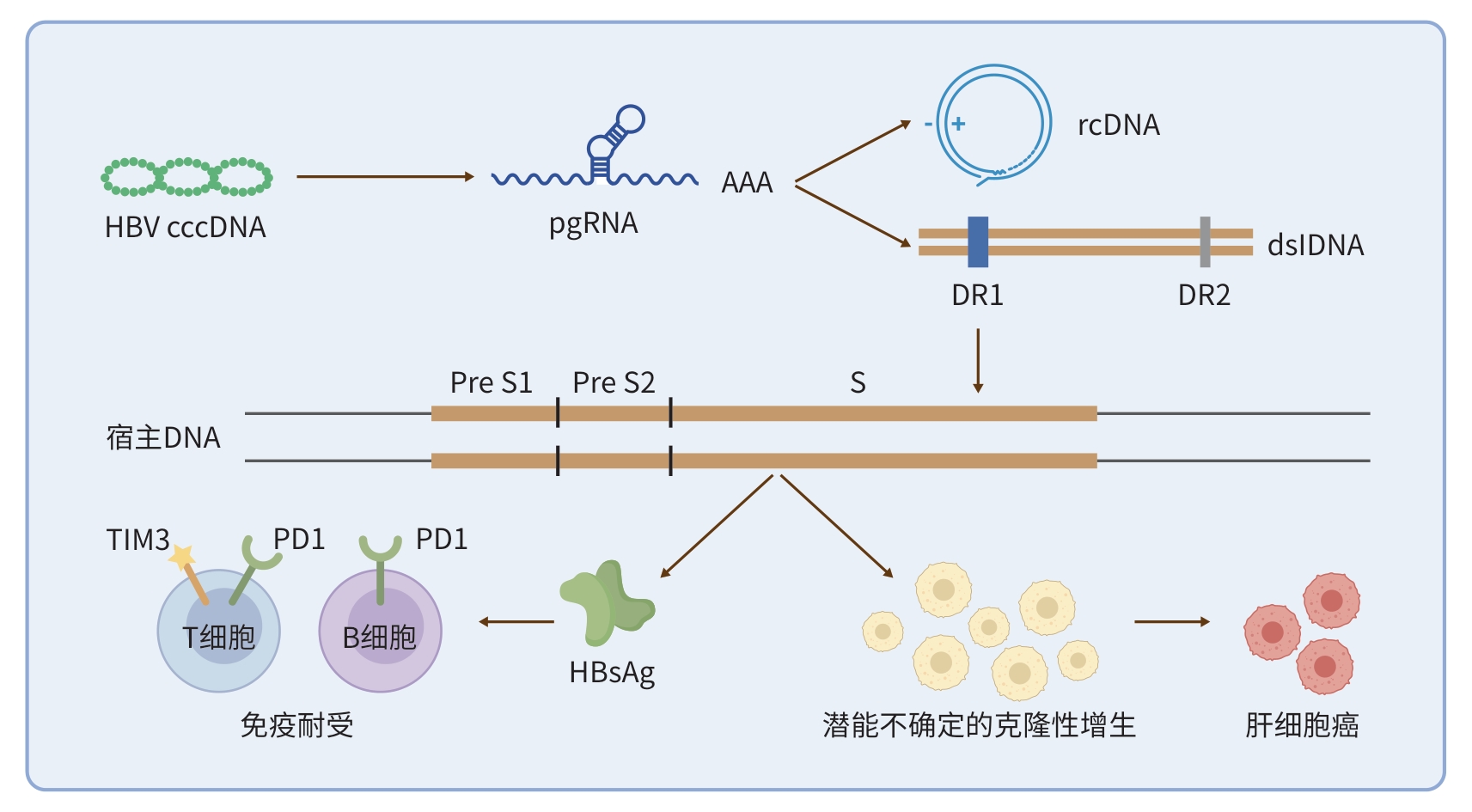
MA LL, CHEN SZ, WANG HY, et al. Hepatitis B virus integration and hepatocarcinogenesis[J]. Liver Res, 2025, 9( 3): 189- 198. DOI: 10.1016/j.livres.2025.09.002. |
| [3] |
ZHANG MM, CHEN H, LIU H, et al. The impact of integrated hepatitis B virus DNA on oncogenesis and antiviral therapy[J]. Biomark Res, 2024, 12( 1): 84. DOI: 10.1186/s40364-024-00611-y. |
| [4] |
BOUSALI M, PAPATHEODORIDIS G, PARASKEVIS D, et al. Hepatitis B virus DNA integration, chronic infections and hepatocellular carcinoma[J]. Microorganisms, 2021, 9( 8): 1787. DOI: 10.3390/microorganisms9081787. |
| [5] |
ZOULIM F, CHEN PJ, DANDRI M, et al. Hepatitis B virus DNA integration: Implications for diagnostics, therapy, and outcome[J]. J Hepatol, 2024, 81( 6): 1087- 1099. DOI: 10.1016/j.jhep.2024.06.037. |
| [6] |
GU ZQ, JIANG QQ, ABULAITI A, et al. Hepatitis B virus enhancer 1 activates preS1 and preS2 promoters of integrated HBV DNA impairing HBsAg secretion[J]. JHEP Rep, 2024, 6( 9): 101144. DOI: 10.1016/j.jhepr.2024.101144. |
| [7] |
POLLICINO T, CAMINITI G. HBV-integration studies in the clinic: Role in the natural history of infection[J]. Viruses, 2021, 13( 3): 368. DOI: 10.3390/v13030368. |
| [8] |
CHEN Y, DONG Y, WEI SZ, et al. Genomic integration of hepatitis B virus into human hepatocytes in early childhood cirrhosis[J]. Liver Int, 2025, 45( 4): e70080. DOI: 10.1111/liv.70080. |
| [9] |
KIMBI GC, KRAMVIS A, KEW MC. Integration of hepatitis B virus DNA into chromosomal DNA during acute hepatitis B[J]. World J Gastroenterol, 2005, 11( 41): 6416- 6421. DOI: 10.3748/wjg.v11.i41.6416. |
| [10] |
LAU DT, KIM ES, WANG ZL, et al. Differential intrahepatic integrated HBV DNA patterns between HBeAg-positive and HBeAg-negative chronic hepatitis B[J]. medRxiv, 2025. DOI: 10.1101/2025.02.28.25322668.[Preprint] |
| [11] |
LI CL, HSU CL, LIN YY, et al. HBV DNA integration into telomerase or MLL4 genes and TERT promoter point mutation as three independent signatures in subgrouping HBV-related HCC with distinct features[J]. Liver Cancer, 2024, 13( 1): 41- 55. DOI: 10.1159/000530699. |
| [12] |
LI MG, WU SS, LUO HQ, et al. HBV DNA integration gene CCDC91 is oncogenic and a potential therapeutic target for hepatocellular carcinoma[J]. Commun Biol, 2025, 8( 1): 1079. DOI: 10.1038/s42003-025-08369-1. |
| [13] |
BURTON AR, PALLETT LJ, MCCOY LE, et al. Circulating and intrahepatic antiviral B cells are defective in hepatitis B[J]. J Clin Invest, 2018, 128( 10): 4588- 4603. DOI: 10.1172/JCI121960. |
| [14] |
YU F, ZHU Y, LI SH, et al. Dysfunction and regulatory interplay of T and B cells in chronic hepatitis B: Immunotherapy and emerging antiviral strategies[J]. Front Cell Infect Microbiol, 2024, 14: 1488527. DOI: 10.3389/fcimb.2024.1488527. |
| [15] |
MARRAPU S, SONI JR, KAMAL K, et al. Hepatitis B functional cure: Current and future perspective[J]. World J Hepatol, 2025, 17( 10): 110107. DOI: 10.4254/wjh.v17.i10.110107. |
| [16] |
PAN DZ, SOULETTE CM, AGGARWAL A, et al. Effects of tenofovir disoproxil fumarate on intrahepatic viral burden and liver immune microenvironment in patients with chronic hepatitis B[J]. Gut, 2025, 74( 4): 628- 638. DOI: 10.1136/gutjnl-2024-332526. |
| [17] |
ZHANG MY, ZHANG HK, CHENG XM, et al. Liver biopsy of chronic hepatitis B patients indicates HBV integration profile may complicate the endpoint and effect of entecavir treatment[J]. Antiviral Res, 2022, 204: 105363. DOI: 10.1016/j.antiviral.2022.105363. |
| [18] |
TADDESE M, GRUDDA T, BELLUCCINI G, et al. Transcription of hepatitis B surface antigen shifts from cccDNA to integrated HBV DNA during treatment[J]. J Clin Invest, 2025, 135( 6): e184243. DOI: 10.1172/JCI184243. |
| [19] |
GAO N, GUAN GW, XU GL, et al. Integrated HBV DNA and cccDNA maintain transcriptional activity in intrahepatic HBsAg-positive patients with functional cure following PEG-IFN-based therapy[J]. Aliment Pharmacol Ther, 2023, 58( 10): 1086- 1098. DOI: 10.1111/apt.17670. |
| [20] |
KILANY MM, SONNEVELD MJ, FELD JJ, et al. Management of immune-tolerant chronic hepatitis B[J]. Hepatology, 2025. DOI: 10.1097/HEP.0000000000001407. DOI: 10.1097/HEP.0000000000001407.
|
| [21] |
YUEN MF, WONG DK, SCHLUEP T, et al. Long-term serological, virological and histological responses to RNA inhibition by ARC-520 in Chinese chronic hepatitis B patients on entecavir treatment[J]. Gut, 2022, 71( 4): 789- 797. DOI: 10.1136/gutjnl-2020-323445. |
| [22] |
YUEN MF, LIM SG, PLESNIAK R, et al. Efficacy and safety of bepirovirsen in chronic hepatitis B infection[J]. N Engl J Med, 2022, 387( 21): 1957- 1968. DOI: 10.1056/nejmoa2210027. |







 DownLoad:
DownLoad: