| [1] |
LINK JO, TAYLOR JG, XU LH, et al. Discovery of ledipasvir(GS-5885): A potent, once-daily oral NS5A inhibitor for the treatment of hepatitis C virus infection[J]. J Med Chem, 2014, 57( 5): 2033- 2046. DOI: 10.1021/jm401499g. |
| [2] |
CHATTERJEE A, SMITH PF, PERELSON AS. Hepatitis C viral kinetics: The past, present, and future[J]. Clin Liver Dis, 2013, 17( 1): 13- 26. DOI: 10.1016/j.cld.2012.09.003. |
| [3] |
NGUYEN THT, GUEDJ J, UPRICHARD SL, et al. The paradox of highly effective sofosbuvir-based combination therapy despite slow viral decline: Can we still rely on viral kinetics?[J]. Sci Rep, 2017, 7( 1): 10233. DOI: 10.1038/s41598-017-09776-z. |
| [4] |
NOWAK MA, BONHOEFFER S, HILL AM, et al. Viral dynamics in hepatitis B virus infection[J]. Proc Natl Acad Sci USA, 1996, 93( 9): 4398- 4402. DOI: 10.1073/pnas.93.9.4398. |
| [5] |
IMAM H, KHAN M, GOKHALE NS, et al. N6-methyladenosine modification of hepatitis B virus RNA differentially regulates the viral life cycle[J]. Proc Natl Acad Sci USA, 2018, 115( 35): 8829- 8834. DOI: 10.1073/pnas.1808319115. |
| [6] |
LIU F, LEE ACH, GUO F, et al. Host poly(A) polymerases PAPD5 and PAPD7 provide two layers of protection that ensure the integrity and stability of hepatitis B virus RNA[J]. J Virol, 2021, 95( 18): e00574-21. DOI: 10.1128/JVI.00574-21. |
| [7] |
TABATA K, NEUFELDT CJ, BARTENSCHLAGER R. Hepatitis C virus replication[J]. Cold Spring Harb Perspect Med, 2020, 10( 3): a037093. DOI: 10.1101/cshperspect.a037093. |
| [8] |
MATA M, NEBEN S, MAJZOUB K, et al. Impact of a patient-derived hepatitis C viral RNA genome with a mutated microRNA binding site[J]. PLoS Pathog, 2019, 15( 5): e1007467. DOI: 10.1371/journal.ppat.1007467. |
| [9] |
|
| [11] |
HU JL, HUANG AL. Classifying hepatitis B therapies with insights from covalently closed circular DNA dynamics[J]. Virol Sin, 2024, 39( 1): 9- 23. DOI: 10.1016/j.virs.2023.12.005. |
| [12] |
ASABE S, WIELAND SF, CHATTOPADHYAY PK, et al. The size of the viral inoculum contributes to the outcome of hepatitis B virus infection[J]. J Virol, 2009, 83( 19): 9652- 9662. DOI: 10.1128/JVI.00867-09. |
| [13] |
GUIDOTTI LG, ROCHFORD R, CHUNG J, et al. Viral clearance without destruction of infected cells during acute HBV infection[J]. Science, 1999, 284( 5415): 825- 829. DOI: 10.1126/science.284.5415.825. |
| [14] |
|
| [15] |
FRELIN L, WAHLSTRÖM T, TUCKER AE, et al. A mechanism to explain the selection of the hepatitis E antigen-negative mutant during chronic hepatitis B virus infection[J]. J Virol, 2009, 83( 3): 1379- 1392. DOI: 10.1128/JVI.01902-08. |
| [16] |
VOLZ T, LUTGEHETMANN M, WACHTLER P, et al. Impaired intrahepatic hepatitis B virus productivity contributes to low viremia in most HBeAg-negative patients[J]. Gastroenterology, 2007, 133( 3): 843- 852. DOI: 10.1053/j.gastro.2007.06.057. |
| [17] |
WALKER A, SCHWARZ T, BRINKMANN-PAULUKAT J, et al. Immune escape pathways from the HBV core(18-27) CD8 T cell response are driven by individual HLA class I alleles[J]. Front Immunol, 2022, 13: 1045498. DOI: 10.3389/fimmu.2022.1045498. |
| [18] |
LI GX, YANG DL, LIU X, et al. Precore mutation enhances viral replication to facilitate persistent infection especially in HBeAg-negative patients[J]. Virol Sin, 2024, 39( 2): 319- 330. DOI: 10.1016/j.virs.2024.03.003. |
| [19] |
UCHIDA T, IMAMURA M, HAYES CN, et al. HBV with precore and basal core promoter mutations exhibits a high replication phenotype and causes ER stress-mediated cell death in humanized liver chimeric mice[J]. Hepatology, 2023, 78( 3): 929- 942. DOI: 10.1097/HEP.0000000000000335. |
| [20] |
ROSSI M, VECCHI A, TIEZZI C, et al. Phenotypic CD8 T cell profiling in chronic hepatitis B to predict HBV-specific CD8 T cell susceptibility to functional restoration in vitro[J]. Gut, 2023, 72( 11): 2123- 2137. DOI: 10.1136/gutjnl-2022-327202. |
| [21] |
HOOGEVEEN RC, ROBIDOUX MP, SCHWARZ T, et al. Phenotype and function of HBV-specific T cells is determined by the targeted epitope in addition to the stage of infection[J]. Gut, 2019, 68( 5): 893- 904. DOI: 10.1136/gutjnl-2018-316644. |
| [22] |
BAULU E, GARDET C, CHUVIN N, et al. TCR-engineered T cell therapy in solid tumors: State of the art and perspectives[J]. Sci Adv, 2023, 9( 7): eadf3700. DOI: 10.1126/sciadv.adf3700. |
| [23] |
|
| [24] |
WU XA, QUAN DM, LI W, et al. Clinical results of an HBV-specific T-cell receptor-T-cell therapy(SCG101) in patients with HBV-related hepatocellular carcinoma treated in an investigator-initiated, interventional trial[J]. Gut, 2025, 75( 1): 147- 160. DOI: 10.1136/gutjnl-2025-335456. |
| [25] |
WAN XS, WISSKIRCHEN K, JIN T, et al. Genetically-modified, redirected T cells target hepatitis B surface antigen-positive hepatocytes and hepatocellular carcinoma lesions in a clinical setting[J]. Clin Mol Hepatol, 2024, 30( 4): 735- 755. DOI: 10.3350/cmh.2024.0058. |
| [26] |
|
| [27] |
WALTER E, KEIST R, NIEDERÖST B, et al. Hepatitis B virus infection of Tupaia hepatocytes in vitro and in vivo[J]. Hepatology, 1996, 24( 1): 1- 5. DOI: 10.1002/hep.510240101. |
| [28] |
YUEN MF, HEO J, KUMADA H, et al. Phase IIa, randomised, double-blind study of GSK3389404 in patients with chronic hepatitis B on stable nucleos(t)ide therapy[J]. J Hepatol, 2022, 77( 4): 967- 977. DOI: 10.1016/j.jhep.2022.05.031. |
| [29] |
JOSHI S, FREUDENBERG JM, SINGH JM, et al. Immunomodulation by bepirovirsen may induce killing of infected hepatocytes(B-Together study)[J]. Hepatol Int, 2025. DOI: 10.1007/s12072-025-10917-0.[ Epub ahead of print] |
| [30] |
YUEN MF, LIM SG, PLESNIAK R, et al. Efficacy and safety of bepirovirsen in chronic hepatitis B infection[J]. N Engl J Med, 2022, 387( 21): 1957- 1968. DOI: 10.1056/NEJMoa2210027. |



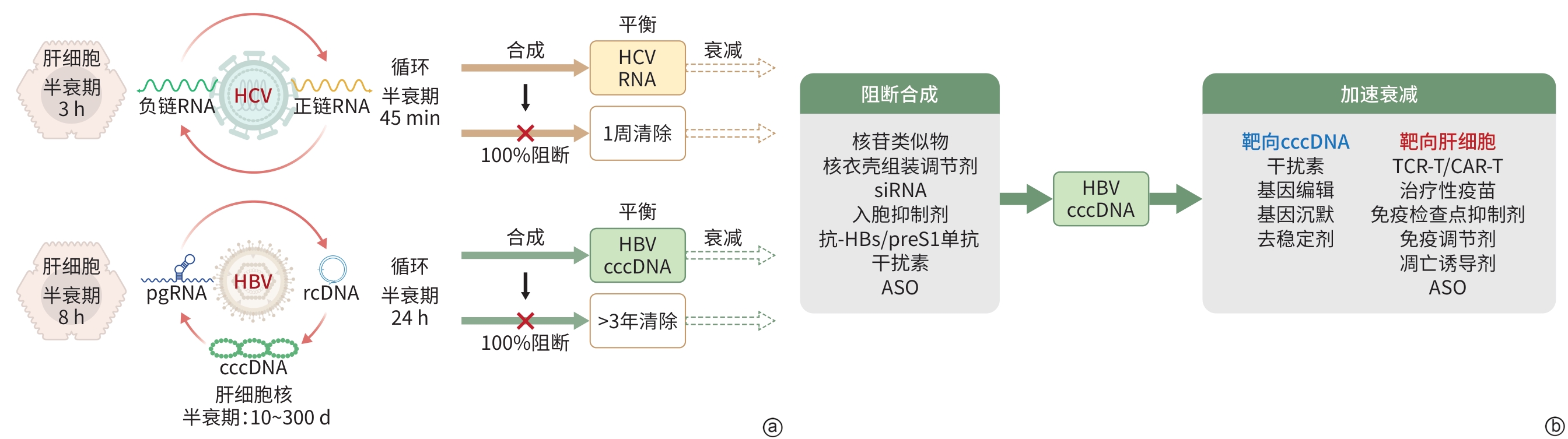




 DownLoad:
DownLoad: