| [1] |
GBD 2019 Hepatitis B Collaborators. Global, regional, and national burden of hepatitis B, 1990-2019: A systematic analysis for the Global Burden of Disease Study 2019[J]. Lancet Gastroenterol Hepatol, 2022, 7( 9): 796- 829. DOI: 10.1016/S2468-1253(22)00124-8. |
| [2] |
YAN H, ZHONG GC, XU GW, et al. Sodium taurocholate cotransporting polypeptide is a functional receptor for human hepatitis B and D virus[J]. eLife, 2012, 1: e00049. DOI: 10.7554/eLife.00049. |
| [3] |
WEI L, PLOSS A. Core components of DNA lagging strand synthesis machinery are essential for hepatitis B virus cccDNA formation[J]. Nat Microbiol, 2020, 5( 5): 715- 726. DOI: 10.1038/s41564-020-0678-0. |
| [4] |
ZOULIM F, CHEN PJ, DANDRI M, et al. Hepatitis B virus DNA integration: Implications for diagnostics, therapy, and outcome[J]. J Hepatol, 2024, 81( 6): 1087- 1099. DOI: 10.1016/j.jhep.2024.06.037. |
| [5] |
QU BQ, BROWN RJP. Strategies to inhibit hepatitis B virus at the transcript level[J]. Viruses, 2021, 13( 7): 1327. DOI: 10.3390/v13071327. |
| [6] |
YUAN HF, ZHAO LN, YANG G, et al. HBx-induced HSPB1 is a potential therapeutic target owing to its modulation of HBV cccDNA and hepatic immune responses[J]. J Hepatol, 2025. DOI: 10.1016/j.jhep.2025.09.033.[ Epub ahead of print] |
| [7] |
YE JY, LI FH, HUA T, et al. Liver mechanosignaling as a natural anti-hepatitis B virus mechanism[J]. Nat Commun, 2024, 15( 1): 8375. DOI: 10.1038/s41467-024-52718-3. |
| [8] |
|
| [9] |
MAO RC, ZHANG JM, JIANG D, et al. Indoleamine 2, 3-dioxygenase mediates the antiviral effect of gamma interferon against hepatitis B virus in human hepatocyte-derived cells[J]. J Virol, 2011, 85( 2): 1048- 1057. DOI: 10.1128/JVI.01998-10. |
| [10] |
SINHA P, THIO CL, BALAGOPAL A. Intracellular host restriction of hepatitis B virus replication[J]. Viruses, 2024, 16( 5): 764. DOI: 10.3390/v16050764. |
| [11] |
ZHANG W, CHEN JL, WU M, et al. PRMT5 restricts hepatitis B virus replication through epigenetic repression of covalently closed circular DNA transcription and interference with pregenomic RNA encapsidation[J]. Hepatology, 2017, 66( 2): 398- 415. DOI: 10.1002/hep.29133. |
| [12] |
REN JH, HU JL, CHENG ST, et al. SIRT3 restricts hepatitis B virus transcription and replication through epigenetic regulation of covalently closed circular DNA involving suppressor of variegation 3-9 homolog 1 and SET domain containing 1A histone methyltransferases[J]. Hepatology, 2018, 68( 4): 1260- 1276. DOI: 10.1002/hep.29912. |
| [13] |
DECORSIÈRE A, MUELLER H, van BREUGEL PC, et al. Hepatitis B virus X protein identifies the Smc5/6 complex as a host restriction factor[J]. Nature, 2016, 531( 7594): 386- 389. DOI: 10.1038/nature17170. |
| [14] |
YAO QY, PENG B, LI C, et al. SLF2 interacts with the SMC5/6 complex to direct hepatitis B virus episomal DNA to promyelocytic leukemia bodies for transcriptional repression[J]. J Virol, 2023, 97( 7): e00328-23. DOI: 10.1128/jvi.00328-23. |
| [15] |
WANG YX, NIKLASCH M, LIU TT, et al. Interferon-inducible MX2 is a host restriction factor of hepatitis B virus replication[J]. J Hepatol, 2020, 72( 5): 865- 876. DOI: 10.1016/j.jhep.2019.12.009. |
| [16] |
SHEN ZL, ZHANG SY, GAO ZX, et al. Intrahepatic homeobox protein MSX-1 is a novel host restriction factor of hepatitis B virus[J]. J Virol, 2024, 98( 2): e01345-23. DOI: 10.1128/jvi.01345-23. |
| [17] |
CHENG ST, HU JL, REN JH, et al. Dicoumarol, an NQO1 inhibitor, blocks cccDNA transcription by promoting degradation of HBx[J]. J Hepatol, 2021, 74( 3): 522- 534. DOI: 10.1016/j.jhep.2020.09.019. |
| [18] |
ALLWEISS L, GIERSCH K, PIROSU A, et al. Therapeutic shutdown of HBV transcripts promotes reappearance of the SMC5/6 complex and silencing of the viral genome in vivo[J]. Gut, 2022, 71( 2): 372- 381. DOI: 10.1136/gutjnl-2020-322571. |
| [19] |
KAJINO K, JILBERT AR, SAPUTELLI J, et al. Woodchuck hepatitis virus infections: Very rapid recovery after a prolonged viremia and infection of virtually every hepatocyte[J]. J Virol, 1994, 68( 9): 5792- 5803. DOI: 10.1128/JVI.68.9.5792-5803.1994. |
| [20] |
GUIDOTTI LG, ANDO K, HOBBS MV, et al. Cytotoxic T lymphocytes inhibit hepatitis B virus gene expression by a noncytolytic mechanism in transgenic mice[J]. Proc Natl Acad Sci USA, 1994, 91( 9): 3764- 3768. DOI: 10.1073/pnas.91.9.3764. |
| [21] |
XIA YC, STADLER D, LUCIFORA J, et al. Interferon-γ and tumor necrosis factor-α produced by T cells reduce the HBV persistence form, cccDNA, without cytolysis[J]. Gastroenterology, 2016, 150( 1): 194- 205. DOI: 10.1053/j.gastro.2015.09.026. |
| [22] |
SHEN F, LI YM, WANG Y, et al. Hepatitis B virus sensitivity to interferon-α in hepatocytes is more associated with cellular interferon response than with viral genotype[J]. Hepatology, 2018, 67( 4): 1237- 1252. DOI: 10.1002/hep.29609. |
| [23] |
DING JH, YI ZG, ZAI WJ, et al. Illuminating the live-cell dynamics of hepatitis B virus covalently closed circular DNA using the CRISPR-tag system[J]. mBio, 2023, 14( 2): e03550-22. DOI: 10.1128/mbio.03550-22. |
| [24] |
GEHRING AJ, PROTZER U. Targeting innate and adaptive immune responses to cure chronic HBV infection[J]. Gastroenterology, 2019, 156( 2): 325- 337. DOI: 10.1053/j.gastro.2018.10.032. |
| [25] |
LI XF, SUN WX, XU XL, et al. Hepatitis B virus surface antigen drives T cell immunity through non-canonical antigen presentation in mice[J]. Nat Commun, 2025, 16( 1): 4591. DOI: 10.1038/s41467-025-59985-8. |
| [26] |
ANDREATA F, LAURA C, RAVÀ M, et al. Therapeutic potential of co-signaling receptor modulation in hepatitis B[J]. Cell, 2024, 187( 15): 4078- 4094. e 21. DOI: 10.1016/j.cell.2024.05.038. |
| [27] |
BOSCH M, KALLIN N, DONAKONDA S, et al. A liver immune rheostat regulates CD8 T cell immunity in chronic HBV infection[J]. Nature, 2024, 631( 8022): 867- 875. DOI: 10.1038/s41586-024-07630-7. |
| [28] |
HEIM K, SAGAR, SOGUKPINAR Ö, et al. Attenuated effector T cells are linked to control of chronic HBV infection[J]. Nat Immunol, 2024, 25( 9): 1650- 1662. DOI: 10.1038/s41590-024-01928-4. |
| [29] |
LI YY, TANG LB, GUO L, et al. CXCL13-mediated recruitment of intrahepatic CXCR5(+)CD8 + T cells favors viral control in chronic HBV infection[J]. J Hepatol, 2020, 72( 3): 420- 430. DOI: 10.1016/j.jhep.2019.09.031. |
| [30] |
WANG Q, MICHAILIDIS E, YU YP, et al. A combination of human broadly neutralizing antibodies against hepatitis B virus HBsAg with distinct epitopes suppresses escape mutations[J]. Cell Host Microbe, 2020, 28( 2): 335- 349. e 6. DOI: 10.1016/j.chom.2020.05.010. |
| [31] |
LE BERT N, SALIMZADEH L, GILL US, et al. Comparative characterization of B cells specific for HBV nucleocapsid and envelope proteins in patients with chronic hepatitis B[J]. J Hepatol, 2020, 72( 1): 34- 44. DOI: 10.1016/j.jhep.2019.07.015. |
| [32] |
LI J, MA X, XUAN QK, et al. Modulation of monocyte activity by hepatocellular microRNA delivery through HBsAg particles: Implications for pathobiology of chronic hepatitis B[J]. Hepatology, 2025, 81( 3): 990- 1005. DOI: 10.1097/HEP.0000000000000972. |
| [33] |
PUBLICOVER J, GAGGAR A, JESPERSEN JM, et al. An OX40/OX40L interaction directs successful immunity to hepatitis B virus[J]. Sci Transl Med, 2018, 10( 433): eaah5766. DOI: 10.1126/scitranslmed.aah5766. |
| [34] |
ZHANG C, LI JS, CHENG YQ, et al. Single-cell RNA sequencing reveals intrahepatic and peripheral immune characteristics related to disease phases in HBV-infected patients[J]. Gut, 2023, 72( 1): 153- 167. DOI: 10.1136/gutjnl-2021-325915. |
| [35] |
QI RY, FU R, LEI X, et al. Therapeutic vaccine-induced plasma cell differentiation is defective in the presence of persistently high HBsAg levels[J]. J Hepatol, 2024, 80( 5): 714- 729. DOI: 10.1016/j.jhep.2023.12.032. |
| [36] |
PROTZER U, MAINI MK, KNOLLE PA. Living in the liver: Hepatic infections[J]. Nat Rev Immunol, 2012, 12( 3): 201- 213. DOI: 10.1038/nri3169. |
| [37] |
XU L, YIN WW, SUN R, et al. Kupffer cell-derived IL-10 plays a key role in maintaining humoral immune tolerance in hepatitis B virus-persistent mice[J]. Hepatology, 2014, 59( 2): 443- 452. DOI: 10.1002/hep.26668. |
| [38] |
FANG Z, ZHANG Y, ZHU ZQ, et al. Monocytic MDSCs homing to thymus contribute to age-related CD8 + T cell tolerance of HBV[J]. J Exp Med, 2022, 219( 4): e20211838. DOI: 10.1084/jem.20211838. |
| [39] |
THIMME R, HOFMANN M, BERTOLETTI A, et al. Decoding HBV-specific adaptive immunity: From natural clearance to cure[J]. Gut, 2025. DOI: 10.1136/gutjnl-2025-337129.[ Epub ahead of print] |
| [40] |
LIM SG, BAUMERT TF, BONI C, et al. The scientific basis of combination therapy for chronic hepatitis B functional cure[J]. Nat Rev Gastroenterol Hepatol, 2023, 20( 4): 238- 253. DOI: 10.1038/s41575-022-00724-5. |
| [41] |
JI Y, LE BERT N, WONG GL, et al. The impact of hepatitis B surface antigen reduction via small interfering RNA treatment on natural and vaccine(BRII-179)-induced hepatitis B virus-specific humoral and cellular immune responses[J]. Gastroenterology, 2025, 169( 1): 136- 149. DOI: 10.1053/j.gastro.2025.02.016. |
| [42] |
IANNACONE M, BECCARIA CG, ALLWEISS L, et al. Targeting HBV with RNA interference: Paths to cure[J]. Sci Transl Med, 2025, 17( 805): eadv3678. DOI: 10.1126/scitranslmed.adv3678. |
| [43] |
HU KY, ZAI WJ, XU MZ, et al. Augmented epigenetic repression of hepatitis B virus covalently closed circular DNA by interferon-α and small-interfering RNA synergy[J]. mBio, 2024, 15( 12): e02415-24. DOI: 10.1128/mbio.02415-24. |
| [44] |
LIN CR, HUANG Y, RAN N, et al. Therapeutic inhibition of HBsAg and HBV cccDNA through a novel phased combination treatment: Glycine and interferon-α[J]. Gut, 2025, 74( 12): 2035- 2049. DOI: 10.1136/gutjnl-2025-334813. |
| [45] |
ASHUO A, LIU J, YUAN ZH, et al. Interferon-α for immune modulation in chronic hepatitis B toward functional cure[J]. Viruses, 2025, 17( 10): 1358. DOI: 10.3390/v17101358. |



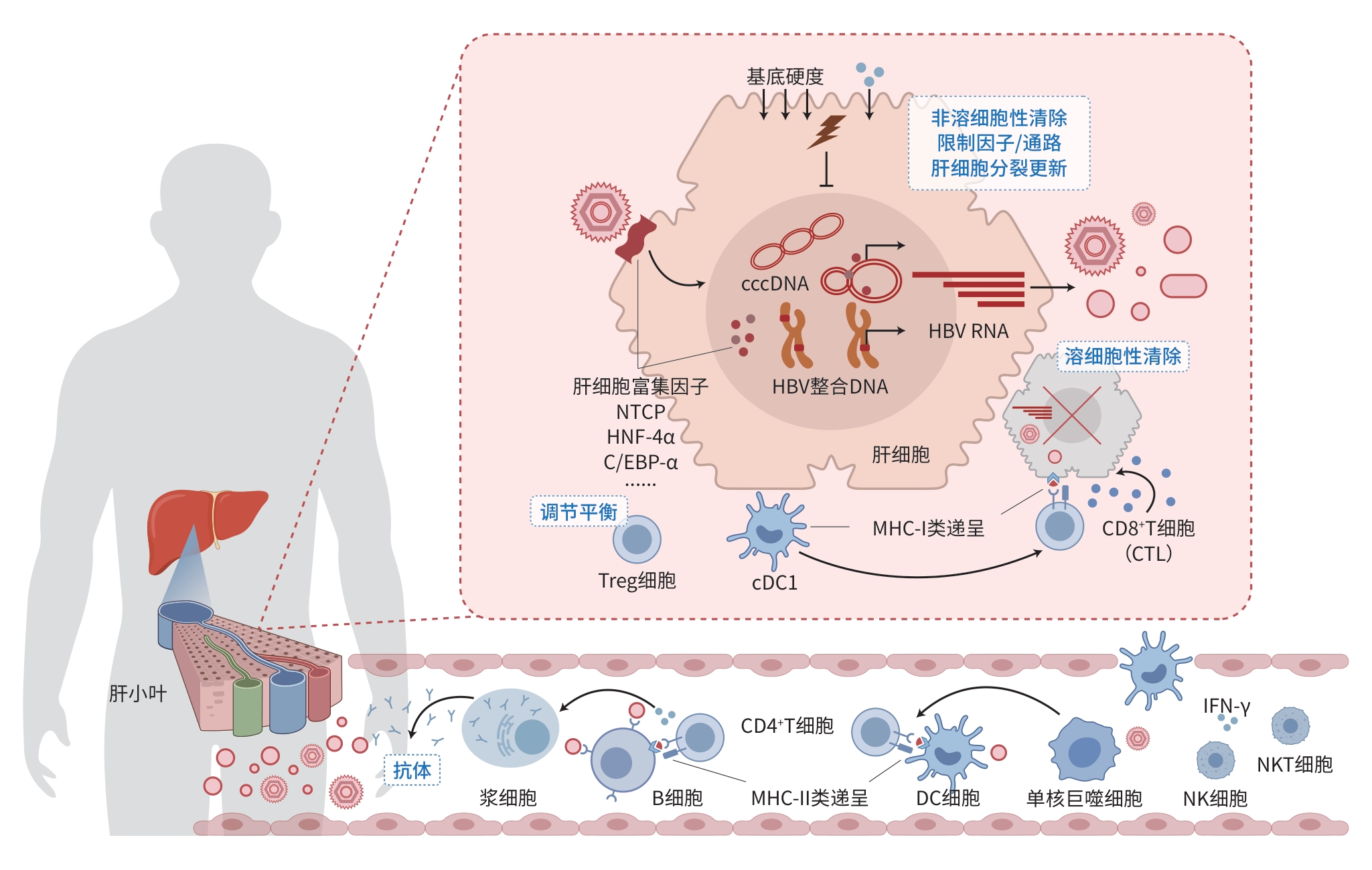




 DownLoad:
DownLoad: