他汀类药物在代谢相关脂肪性肝病治疗中的应用
DOI: 10.12449/JCH260322
Application of statins in the treatment of metabolic dysfunction-associated fatty liver disease
-
摘要: 代谢相关脂肪性肝病(MAFLD)及其进展形式——代谢相关性脂肪性肝炎(MASH)已成为全球慢性肝病的主要病因,其发病率持续上升,与肥胖、2型糖尿病等代谢紊乱密切相关。MAFLD的核心发病机制涉及胰岛素抵抗、脂代谢异常及慢性炎症反应,可进展为MASH,导致肝纤维化、肝硬化甚至肝细胞癌。目前,针对MAFLD的有效药物治疗仍有限。本文基于PRISMA指南,系统综述他汀类药物在MAFLD治疗中的作用。研究表明,他汀类药物不仅能改善血脂谱和肝酶水平,还对合并心血管疾病或2型糖尿病等患者具有良好获益,且长期使用还可以降低肝细胞癌风险。然而,其潜在的肝毒性及肌病风险需警惕,因此临床应个体化用药并定期监测肝功能。
-
关键词:
- 羟甲基戊二酰基CoA还原酶抑制剂 /
- 代谢相关脂肪性肝病 /
- 治疗学
Abstract: Metabolic dysfunction-associated fatty liver disease (MAFLD) and its progressive form metabolic dysfunction-associated steatohepatitis (MASH) have become the leading causes of chronic liver diseases worldwide, and the incidence rate of MAFLD continues to rise, which is closely associated with metabolic disorders such as obesity and type 2 diabetes. The core pathogenesis of MAFLD involves insulin resistance, abnormal lipid metabolism, and chronic inflammation, which can progress to MASH and lead to liver fibrosis, liver cirrhosis, and even hepatocellular carcinoma (HCC). At present, there are still limited effective pharmacotherapies for MAFLD. Based on the PRISMA guidelines, this article systematically reviews the role of statins in MAFLD. Studies have shown that statins not only improve blood lipid profiles and the levels of liver enzyme, but also bring good benefits to patients comorbid with cardiovascular disease or type 2 diabetes, and long-term use can also reduce the risk of HCC. However, the potential risks of hepatotoxicity and myopathy should be taken seriously, which, therefore, requires individualized medication and regular monitoring of liver function in clinical practice. -
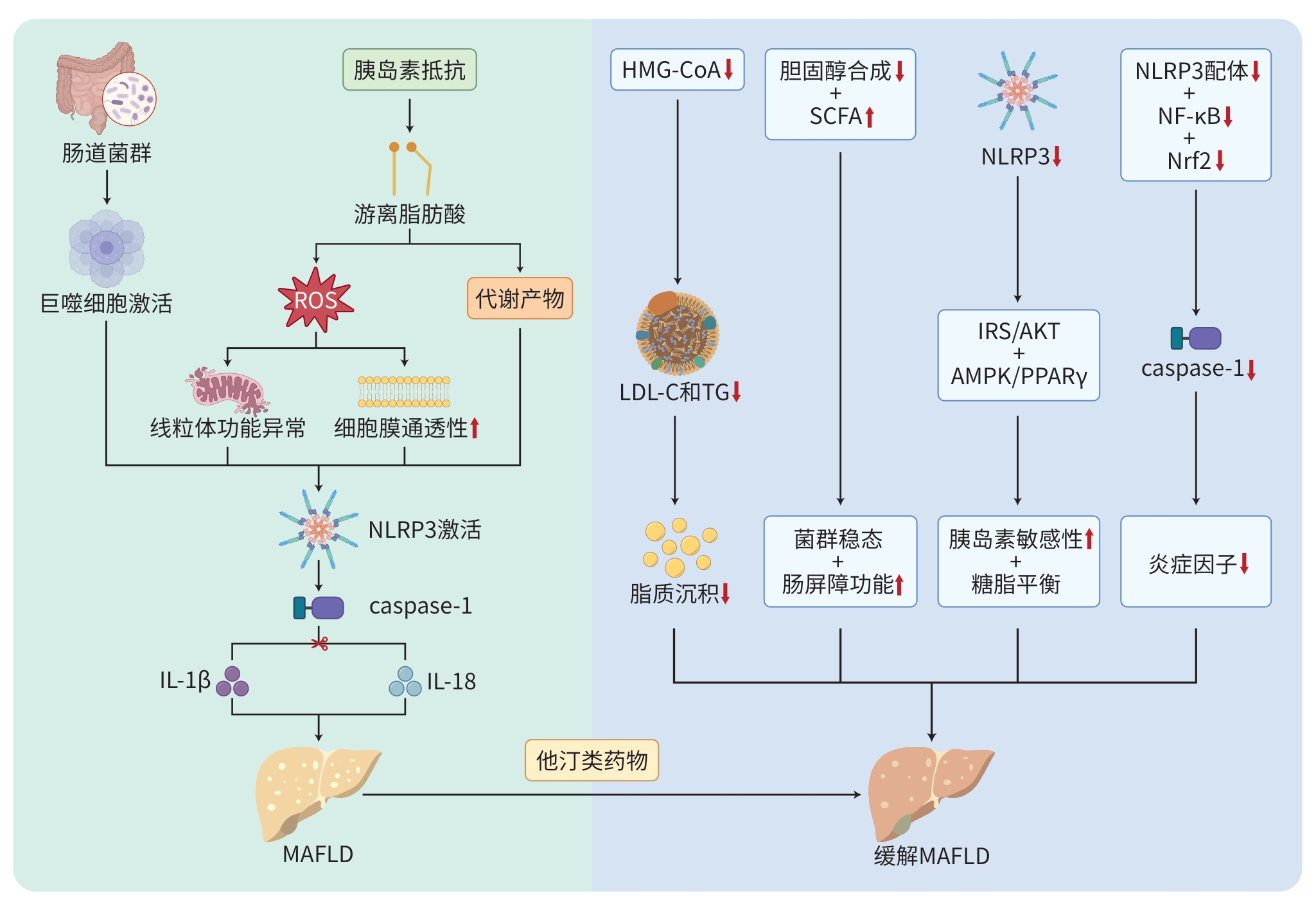
注: ROS,活性氧;NLRP3,核苷酸结合寡聚结构域样受体家族热蛋白结构域相关蛋白3;caspase-1,胱天蛋白酶1;IL-1β,白细胞介素1β;IL-18,白细胞介素18;MAFLD,代谢相关脂肪性肝病;HMG-CoA,羟基甲基戊二酸酰辅酶A;SCFA,短链脂肪酸;NF-κB,核因子κB;Nrf2,核因子红系2相关因子2;IRS,胰岛素受体底物;AMPK,AMP活化的蛋白质激酶;PPARγ,过氧化物酶体增殖物激活受体γ;LDL-C,低密度脂蛋白胆固醇;TG,甘油三酯。
图 1 他汀类药物治疗MAFLD的作用机制
Figure 1. The mechanism of action of statins in the treatment of MAFLD
-
[1] RINELLA ME, LAZARUS JV, RATZIU V, et al. A multisociety Delphi consensus statement on new fatty liver disease nomenclature[J]. Hepatology, 2023, 78( 6): 1966- 1986. DOI: 10.1097/HEP.0000000000000520. [2] FAN JG, XU XY, YANG RX, et al. Guideline for the prevention and treatment of metabolic dysfunction-associated fatty liver disease(version 2024)[J]. J Clin Transl Hepatol, 2024, 12( 11): 955- 974. DOI: 10.14218/jcth.2024.00311 [3] MOHAMED ABDELGAWWAD EL-SEHRAWY A ALI, MOHAMMED AN, GUPTA J, et al. Combating oxidative stress in non-alcoholic fatty liver disease: From mechanisms to therapeutic strategies[J]. Pathol Res Pract, 2025, 272: 156053. DOI: 10.1016/j.prp.2025.156053. [4] MARTÍN-MATEOS R, ALBILLOS A. The role of the gut-liver axis in metabolic dysfunction-associated fatty liver disease[J]. Front Immunol, 2021, 12: 660179. DOI: 10.3389/fimmu.2021.660179. [5] VALLIANOU N, CHRISTODOULATOS GS, KARAMPELA I, et al. Understanding the role of the gut microbiome and microbial metabolites in non-alcoholic fatty liver disease: Current evidence and perspectives[J]. Biomolecules, 2021, 12( 1): 56. DOI: 10.3390/biom-12010056. [6] RAMOS-TOVAR E, MURIEL P. NLRP3 inflammasome in hepatic diseases: A pharmacological target[J]. Biochem Pharmacol, 2023, 217: 115861. DOI: 10.1016/j.bcp.2023.115861. [7] QIN WW, WENG JP. Hepatocyte NLRP3 interacts with PKCε to drive hepatic insulin resistance and steatosis[J]. Sci Bull, 2023, 68( 13): 1413- 1429. DOI: 10.1016/j.scib.2023.06.003. [8] YUAN XX, BHAT OM, ZOU Y, et al. Contribution of hepatic steatosis-intensified extracellular vesicle release to aggravated inflammatory endothelial injury in liver-specific Asah1 gene knockout mice[J]. Am J Pathol, 2023, 193( 4): 493- 508. DOI: 10.1016/j.ajpath.2022.12.007. [9] de DEUS IJ, MARTINS-SILVA AF, de ANDRADE FAGUNDES MM, et al. Role of NLRP3 inflammasome and oxidative stress in hepatic insulin resistance and the ameliorative effect of phytochemical intervention[J]. Front Pharmacol, 2023, 14: 1188829. DOI: 10.3389/fphar.2023.1188829. [10] YU TT, LUO L, XUE J, et al. Gut microbiota-NLRP3 inflammasome crosstalk in metabolic dysfunction-associated steatotic liver disease[J]. Clin Res Hepatol Gastroenterol, 2024, 48( 8): 102458. DOI: 10.1016/j.clinre.2024.102458. [11] INIA JA, STOKMAN G, PIETERMAN EJ, et al. Atorvastatin attenuates diet-induced non-alcoholic steatohepatitis in APOE*3-leiden mice by reducing hepatic inflammation[J]. Int J Mol Sci, 2023, 24( 9): 7818. DOI: 10.3390/ijms24097818. [12] ZHANG X, COKER OO, CHU ES, et al. Dietary cholesterol drives fatty liver-associated liver cancer by modulating gut microbiota and metabolites[J]. Gut, 2021, 70( 4): 761- 774. DOI: 10.1136/gutjnl-2019-319664. [13] TAKAGURI A. Elucidation of a new mechanism of onset of insulin resistance: Effects of statins and tumor necrosis factor-α on insulin signal transduction[J]. Yakugaku Zasshi, 2018, 138( 11): 1329- 1334. DOI: 10.1248/yakushi.18-00116. [14] CLIMENT E, BENAIGES D, PEDRO-BOTET J. Hydrophilic or lipophilic statins[J]. Front Cardiovasc Med, 2021, 8: 687585. DOI: 10.3389/fcvm.2021.687585. [15] SIZAR O, KHARE S, PATEL P, TALATI R. Statin Medications[M]. StatPearls. Treasure Island(FL): StatPearls Publishing, 2024. [16] FOSTER T, BUDOFF MJ, SAAB S, et al. Atorvastatin and antioxidants for the treatment of nonalcoholic fatty liver disease: The St Francis Heart Study randomized clinical trial[J]. Am J Gastroenterol, 2011, 106( 1): 71- 77. DOI: 10.1038/ajg.2010.299. [17] HADZI-PETRUSHEV N, DIMOVSKA K, JANKULOVSKI N, et al. Supplementation with alpha-tocopherol and ascorbic acid to nonalcoholic fatty liver disease’s statin therapy in men[J]. Adv Pharmacol Sci, 2018, 2018: 4673061. DOI: 10.1155/2018/4673061. [18] WANG X, LYU L, LI W, et al. Impact of rosuvastatin on metabolic syndrome patients with moderate to severe metabolic associated fatty liver disease without overt diabetes: A randomized clinical trial[J]. Diabetes Metab Syndr, 2024, 18( 9): 103126. DOI: 10.1016/j.dsx.2024.103126. [19] SFIKAS G, PSALLAS M, KOUMARAS C, et al. Prevalence, diagnosis, and treatment with 3 different statins of non-alcoholic fatty liver disease/non-alcoholic steatohepatitis in military personnel. Do genetics play a role?[J]. Curr Vasc Pharmacol, 2021, 19( 5): 572- 581. DOI: 10.2174/1570161118666201015152921. [20] FATIMA K, MOEED A, WAQAR E, et al. Efficacy of statins in treatment and development of non-alcoholic fatty liver disease and steatohepatitis: A systematic review and meta-analysis[J]. Clin Res Hepatol Gastroenterol, 2022, 46( 4): 101816. DOI: 10.1016/j.clinre.2021.101816. [21] ABDALLAH M, BROWN L, PROVENZA J, et al. Safety and efficacy of dyslipidemia treatment in NAFLD patients: A meta-analysis of randomized controlled trials[J]. Ann Hepatol, 2022, 27( 6): 100738. DOI: 10.1016/j.aohep.2022.100738. [22] DENG YF, XU QQ, CHEN TQ, et al. Kinsenoside alleviates inflammation and fibrosis in experimental NASH mice by suppressing the NF-κB/NLRP3 signaling pathway[J]. Phytomedicine, 2022, 104: 154241. DOI: 10.1016/j.phymed.2022.154241. [23] LIN HZ, QI JW, LIN SW, et al. Inhibition of cholesterol synthesis in T cells protects the liver against steatosis, inflammation, and oxidative stress in metabolic dysfunction-associated steatohepatitis[J]. FASEB J, 2025, 39( 11): e70665. DOI: 10.1096/fj.202500115R. [24] MCGLONE ER, ANSELL TB, DUNSTERVILLE C, et al. Hepatocyte cholesterol content modulates glucagon receptor signalling[J]. Mol Metab, 2022, 63: 101530. DOI: 10.1016/j.molmet.2022.101530. [25] da SILVA PEREIRA ENG, de ARAUJO BP, RODRIGUES KL, et al. Simvastatin improves microcirculatory function in nonalcoholic fatty liver disease and downregulates oxidative and ALE-RAGE stress[J]. Nutrients, 2022, 14( 3): 716. DOI: 10.3390/nu14030716. [26] LASTUVKOVA H, FARADONBEH FA, SCHREIBEROVA J, et al. Atorvastatin modulates bile acid homeostasis in mice with diet-induced nonalcoholic steatohepatitis[J]. Int J Mol Sci, 2021, 22( 12): 6468. DOI: 10.3390/ijms22126468. [27] ESLAMI Z, MOGHANLOU AE, KANDI YMNP, et al. Atorvastatin and flaxseed effects on biochemical indices and hepatic fat of NAFLD model in rats[J]. Adv Biomed Res, 2023, 12: 98. DOI: 10.4103/abr.abr_21_22. [28] ESLAMI Z, JOSHAGHANI H, EGHBAL MOGHANLOU A, et al. Atorvastatin and flaxseed dietary treatments improve dyslipidemia and liver injuries in a diet-induced rat model of non-alcoholic fatty liver disease[J]. Avicenna J Phytomed, 2025, 15( 3): 1102- 1112. DOI: 10.22038/ajp.2024.25220. [29] EGHDAMI S, AFRASHTEH F, SHOJAII A, et al. The therapeutic effect of alcoholic extract of Fumaria parviflora on high-fat diet-induced nonalcoholic fatty liver in rats: An animal experiment[J]. Ann Med Surg, 2024, 86( 5): 2657- 2664. DOI: 10.1097/MS9.0000000000001890. [30] AYADA I, VAN KLEEF LA, ZHANG H, et al. Dissecting the multifaceted impact of statin use on fatty liver disease: A multidimensional study[J]. EBioMedicine, 2023, 87: 104392. DOI: 10.1016/j.ebiom.2022.104392. [31] HO A, KIENER T, NGUYEN QN, et al. Effect of statin use on liver enzymes and lipid profile in patients with non-alcoholic fatty liver disease(NAFLD)[J]. J Clin Lipidol, 2024, 18( 4): e501- e508. DOI: 10.1016/j.jacl.2024.03.003. [32] THOMSON MJ, SERPER M, KHUNGAR V, et al. Prevalence and factors associated with statin use among patients with nonalcoholic fatty liver disease in the TARGET-NASH study[J]. Clin Gastroenterol Hepatol, 2022, 20( 2): 458- 460. e 4. DOI: 10.1016/j.cgh.2021.03.031. [33] NAKAGAWA C, YOKOYAMA S, HOSOMI K. Association of statin adherence with the development of nonalcoholic fatty liver disease: A nested case-control study using a Japanese claims database[J]. Ann Pharmacother, 2023, 57( 6): 637- 645. DOI: 10.1177/10600280221126971. [34] SCHREINER AD, ZHANG JW, PETZ CA, et al. Statin prescriptions and progression of advanced fibrosis risk in primary care patients with MAFLD[J]. BMJ Open Gastroenterol, 2024, 11( 1): e001404. DOI: 10.1136/bmjgast-2024-001404. [35] ZHOU XD, KIM SU, YIP TC, et al. Long-term liver-related outcomes and liver stiffness progression of statin usage in steatotic liver disease[J]. Gut, 2024, 73( 11): 1883- 1892. DOI: 10.1136/gutjnl-2024-333074. [36] TARAR ZI, FAROOQ U, INAYAT F, et al. Statins decrease the risk of hepatocellular carcinoma in metabolic dysfunction-associated steatotic liver disease: A systematic review and meta-analysis[J]. World J Exp Med, 2024, 14( 4): 98543. DOI: 10.5493/wjem.v14.i4.98543. [37] ISLAM MM, POLY TN, WALTHER BA, et al. Statin use and the risk of hepatocellular carcinoma: A meta-analysis of observational studies[J]. Cancers, 2020, 12( 3): 671. DOI: 10.3390/cancers12030671. [38] ZHANG J, FU SF, LIU DL, et al. Statin can reduce the risk of hepatocellular carcinoma among patients with nonalcoholic fatty liver disease: A systematic review and meta-analysis[J]. Eur J Gastroenterol Hepatol, 2023, 35( 4): 353- 358. DOI: 10.1097/MEG.0000000000002517. [39] TOMAH S, ALKHOURI N, HAMDY O. Nonalcoholic fatty liver disease and type 2 diabetes: Where do Diabetologists stand?[J]. Clin Diabetes Endocrinol, 2020, 6: 9. DOI: 10.1186/s40842-020-00097-1. [40] NASCIMBENI F, ARON-WISNEWSKY J, PAIS R, et al. Statins, antidiabetic medications and liver histology in patients with diabetes with non-alcoholic fatty liver disease[J]. BMJ Open Gastroenterol, 2016, 3( 1): e000075. DOI: 10.1136/bmjgast-2015-000075. [41] BRIL F, PORTILLO SANCHEZ P, LOMONACO R, et al. Liver safety of statins in prediabetes or T2DM and nonalcoholic steatohepatitis: Post hoc analysis of a randomized trial[J]. J Clin Endocrinol Metab, 2017, 102( 8): 2950- 2961. DOI: 10.1210/jc.2017-00867. [42] YU HZ, ZHANG SD, LI JC, et al. Effects of statins on progressive liver fibrosis,liver cirrhosis and metabolic associated fatty liver disease in patients with type 2 diabetes[J/CD]. Chin J Liver Dis(Electronic Version), 2025, 17( 1): 35- 43. DOI: 10.3969/j.issn.1674-7380.2025.01.006.余海忠, 张松达, 李进财, 等. 他汀类药物对2型糖尿病患者发生进展期肝纤维化、肝硬化和代谢相关脂肪性肝病的影响[J/CD]. 中国肝脏病杂志(电子版), 2025, 17( 1): 35- 43. DOI: 10.3969/j.issn.1674-7380.2025.01.006. [43] WU SY, CHEN WM, CHIANG MF, et al. Protective effects of statins on the incidence of NAFLD-related decompensated cirrhosis in T2DM[J]. Liver Int, 2023, 43( 10): 2232- 2244. DOI: 10.1111/liv.15656. [44] ATHYROS VG, POLYZOS SA, KOUNTOURAS J, et al. Non-alcoholic fatty liver disease treatment in patients with type 2 diabetes mellitus; new kids on the block[J]. Curr Vasc Pharmacol, 2020, 18( 2): 172- 181. DOI: 10.2174/1570161117666190405164313. [45] CIARDULLO S, PERSEGHIN G. Statin use is associated with lower prevalence of advanced liver fibrosis in patients with type 2 diabetes[J]. Metabolism, 2021, 121: 154752. DOI: 10.1016/j.metabol.2021.154752. [46] ZISIS M, CHONDROGIANNI ME, ANDROUTSAKOS T, et al. Linking cardiovascular disease and metabolic dysfunction-associated steatotic liver disease(MAFLD): The role of cardiometabolic drugs in MAFLD treatment[J]. Biomolecules, 2025, 15( 3): 324. DOI: 10.3390/biom15030324. [47] SAFAROVA MS, WEINTRAUB S, SADANIANTZ K, et al. Statin use in special populations for the prevention of cardiovascular disease in adults[J]. Curr Atheroscler Rep, 2025, 27( 1): 54. DOI: 10.1007/s11883-025-01298-8. [48] GRUNDY SM, STONE NJ, BAILEY AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: A report of the American college of cardiology/American heart association task force on clinical practice guidelines[J]. J Am Coll Cardiol, 2019, 73( 24): e285- e350. DOI: 10.1016/j.jacc.2018.11.003. [49] YOO J, JEON J, BAIK M, et al. Effect of statins for primary prevention of cardiovascular disease according to the fatty liver index[J]. J Epidemiol Glob Health, 2024, 14( 3): 710- 719. DOI: 10.1007/s44197-024-00205-9. [50] SÁNCHEZ-POLO MT, CASTELLS MT, GARCÍA-PÉREZ B, et al. Effect of diet/atorvastatin on atherosclerotic lesions associated to nonalcoholic fatty liver disease in chickens[J]. Histol Histopathol, 2015, 30( 12): 1439- 1446. DOI: 10.14670/HH-11-639. [51] CAI T, ABEL L, LANGFORD O, et al. Associations between statins and adverse events in primary prevention of cardiovascular disease: Systematic review with pairwise, network, and dose-response meta-analyses[J]. BMJ, 2021, 374: n1537. DOI: 10.1136/bmj.n1537. [52] ZHANG HB, PLUTZKY J, SHUBINA M, et al. Continued statin prescriptions after adverse reactions and patient outcomes: A cohort study[J]. Ann Intern Med, 2017, 167( 4): 221- 227. DOI: 10.7326/M16-0838. [53] SARKAR M, KUSHNER T. Metabolic dysfunction-associated steatotic liver disease and pregnancy[J]. J Clin Invest, 2025, 135( 10): e186426. DOI: 10.1172/JCI186426. [54] LINS SERAFIM J, LUCAS SANTOS DE MENEZES TELES P, SOUZA LIMA AK, et al. Clinical repercussions of statin use during pregnancy: A review of the literature[J]. Rev Bras Ginecol Obstet, 2025, 47: e-rbgo 2. DOI: 10.61622/rbgo/2025rbgo2. [55] CHANG JC, CHEN YJ, CHEN IC, et al. Perinatal outcomes after statin exposure during pregnancy[J]. JAMA Netw Open, 2021, 4( 12): e214‑ 1321. DOI: 10.1001/jamanetworkopen.2021.41321. [56] ZHANG Y, BU YF, ZHAO R, et al. Metabolic-associated fatty liver disease and pregnancy complications: New challenges and clinical perspectives[J]. Ther Adv Endocrinol Metab, 2024, 15: 204201882412743‑ 50. DOI: 10.1177/20420188241274350. [57] HUANG SY, HU WT, FANG TY. The global impact of non-alcoholic fatty liver disease(including cirrhosis) in the elderly from 1990 to 2021 and future projections of disease burden[J]. PLoS One, 2025, 20( 6): e0325961. DOI: 10.1371/journal.pone.0325961. [58] GUO MZ, ZHAO JL, ZHAI YJ, et al. A prospective study of hepatic safety of statins used in very elderly patients[J]. BMC Geriatr, 2019, 19( 1): 352. DOI: 10.1186/s12877-019-1361-2. [59] XU WC, YAU YK, PAN YY, et al. Effectiveness and safety of using statin therapy for the primary prevention of cardiovascular diseases in older patients with chronic kidney disease who are hypercholesterolemic: A target trial emulation study[J]. Lancet Healthy Longev, 2025, 6( 3): 100683. DOI: 10.1016/j.lanhl.2025.100683. [60] PASTORI D, PANI A, DI ROCCO A, et al. Statin liver safety in non-alcoholic fatty liver disease: A systematic review and metanalysis[J]. Br J Clin Pharmacol, 2022, 88( 2): 441- 451. DOI: 10.1111/bcp.14943. [61] LI Z, WEI D, WAN MX, et al. Research progress on drug-induced liver injury induced by statins[J]. Drug Eval Res, 2024, 47( 5): 941- 950. DOI: 10.7501/j.issn.1674-6376.2024.05.004.李智, 魏栋, 万梅绪, 等. 他汀类药物致药物性肝损伤的研究进展[J]. 药物评价研究, 2024, 47( 5): 941- 950. DOI: 10.7501/j.issn.1674-6376.2024.05.004. [62] NEWMAN CB. Safety of statins and nonstatins for treatment of dyslipidemia[J]. Endocrinol Metab Clin North Am, 2022, 51( 3): 655- 679. DOI: 10.1016/j.ecl.2022.01.004. [63] KAMMOUN R, CHARFI O, LAKHOUA G, et al. Statin associated muscular adverse effects[J]. Curr Drug Saf, 2024, 19( 1): 114- 116. DOI: 10.2174/1574886318666230227143627. [64] MORRIS R, BU K, HAN WR, et al. The association between statin drugs and rhabdomyolysis: An analysis of FDA adverse event reporting system(FAERS) data and transcriptomic profiles[J]. Genes, 2025, 16( 3): 248. DOI: 10.3390/genes16030248. [65] XIAO M, LI L, ZHU WW, et al. Statin-related neurocognitive disorder: A real-world pharmacovigilance study based on the FDA adverse event reporting system[J]. Expert Rev Clin Pharmacol, 2024, 17( 3): 255- 261. DOI: 10.1080/17512433.2024.2311875. [66] KIM H, KIM N, LEE DH, et al. Analysis of national pharmacovigilance data associated with statin use in Korea[J]. Basic Clin Pharmacol Toxicol, 2017, 121( 5): 409- 413. DOI: 10.1111/bcpt.12808. [67] KARAHALIL B, HARE E, KOÇ G, et al. Hepatotoxicity associated with statins[J]. Arh Hig Rada Toksikol, 2017, 68( 4): 254- 260. DOI: 10.1515/aiht-2017-68-2994. [68] AVERBUKH LD, TURSHUDZHYAN A, WU DC, et al. Statin-induced liver injury patterns: A clinical review[J]. J Clin Transl Hepatol, 2022, 10( 3): 543- 552. DOI: 10.14218/JCTH.2021.00271. [69] WANG HJ, LIU SY, ZHOU CJ, et al. Fatal hepatic failure following atorvastatin treatment: A case report[J]. Medicine, 2023, 102( 19): e33743. DOI: 10.1097/MD.0000000000033743. -



 PDF下载 ( 1043 KB)
PDF下载 ( 1043 KB)

 下载:
下载:


