木犀草素治疗肝损伤的作用机制
DOI: 10.12449/JCH260231
-
摘要: 肝损伤已成为一项日益严峻的全球性健康难题,而现有化学药物存在疗效局限与不良反应等问题,亟需研发安全高效的新型治疗药物。近年研究发现,天然药用植物中的黄酮类化合物在肝损伤防治领域中展现出良好潜力。木犀草素作为典型天然黄酮成分,对多病因肝损伤均表现出良好的保护作用,但其作用机制尚未形成系统性阐释。本文归纳近年国内外相关研究进展,综述木犀草素在抑制氧化应激、抗炎、调控细胞死亡、抗肝纤维化、调节脂质代谢紊乱和调控肠-肝轴等方面的作用机制,并对其在肝损伤治疗中的应用前景进行展望,以期为该成分的深入研究提供科学参考。
-
关键词:
- 木犀草素 /
- 化学与药物性肝损伤 /
- 药理作用分子作用机制
Abstract: Liver injury has become an increasingly serious global health problem, and existing chemical drugs face the limitations in efficacy and adverse reactions, resulting in the urgent need to develop safe and effective drugs. Recent studies have highlighted the potential of flavonoids from natural medicinal plants in the prevention and treatment of liver injury. As a typical natural flavonoid, luteolin shows a good protective effect against liver injury due to various etiologies, but there is still a lack of systematic elaboration on its mechanism of action. This article summarizes related research advances in China and globally and reviews the mechanism of action of luteolin in inhibiting oxidative stress, exerting an anti-inflammatory effect, regulating cell death, alleviating hepatic fibrosis, modulating lipid metabolism disorders, and regulating the gut-liver axis, as well as the application prospect of luteolin in the treatment of liver injury, in order to provide a scientific reference for further research on this compound. -
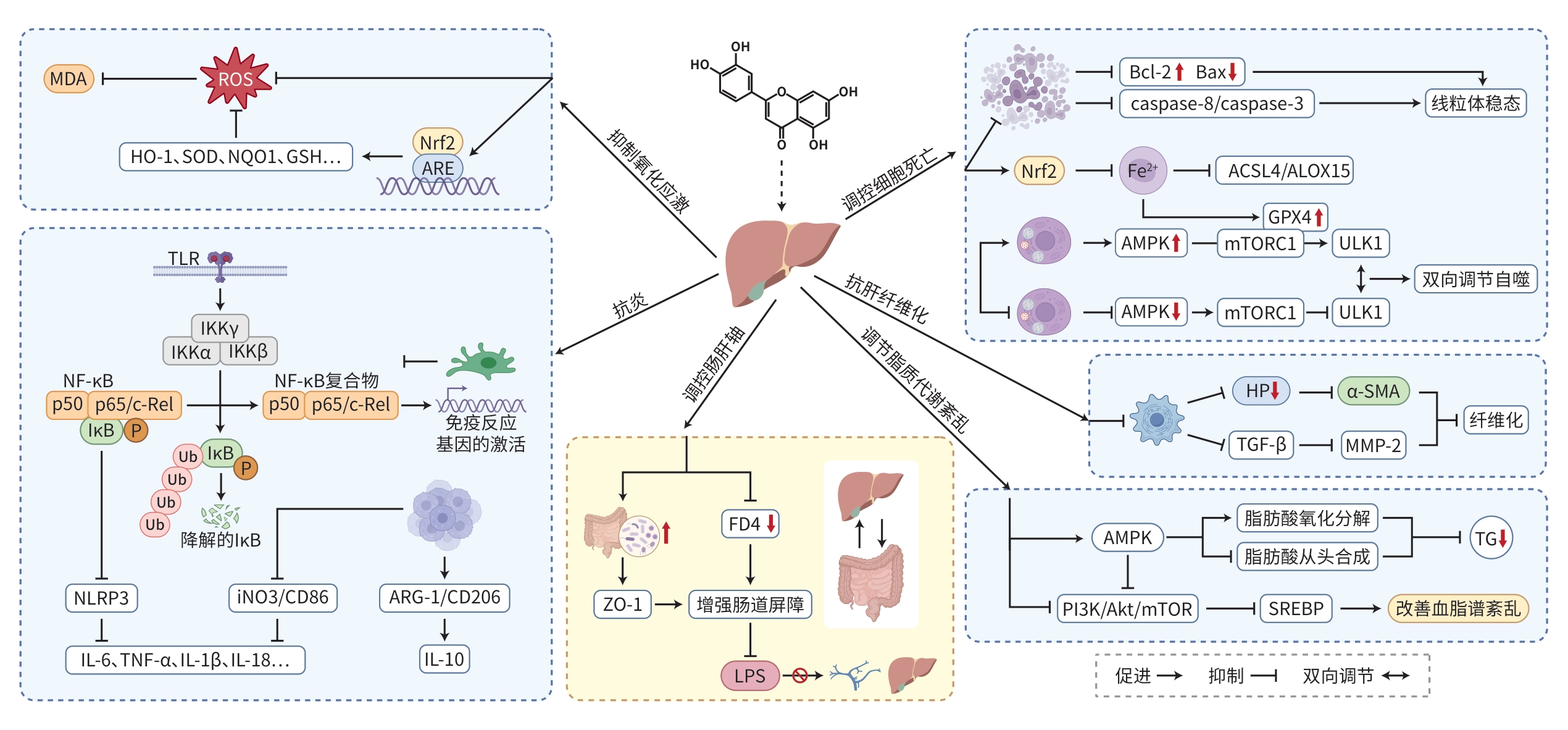
注: MDA,丙二醛;ROS,活性氧;HO-1,血红素加氧酶-1;SOD,超氧化物歧化酶;NQO1,醌氧化还原酶1;GSH,谷胱甘肽;Nrf2,核因子E2相关因子2;ARE,抗氧化反应元件;TLR,Toll样受体;IKK,κB抑制因子激酶;NF-κB,核因子κB;NLRP3,NOD样受体热蛋白结构域相关蛋白3;IL,白细胞介素;TNF-α,肿瘤坏死因子-α;iNo3,诱导型一氧化氮合酶;CD86,分化簇86;ARG-1,精氨酸酶1;CD206,分化簇206;ZO-1,紧密连接蛋白1;FD4,肝脂肪变程度;LPS,脂多糖;Bcl-2,B细胞淋巴瘤因子-2;Bax,Bcl-2相关X蛋白;Caspase,半胱氨酸天冬氨酸蛋白酶家族;mTOR,雷帕霉素靶蛋白;ACSL4,长链脂肪酸辅酶A合成酶4;ALOX15,花生四烯酸15-脂加氧酶;AMPK,腺苷酸活化蛋白激酶;GPX4,谷胱甘肽过氧化物酶4;ULK1,丝氨酸/苏氨酸特异性蛋白激酶;HP,羟脯氨酸;α-SMA,α-平滑肌肌动蛋白;TGF-β,转化生长因子-β;MMP-2,基质金属蛋白酶-2;TG,甘油三酯;Akt,3-激酶-蛋白激酶B;PI3K,磷脂酰肌醇-3-激酶;SREBP,甾醇调节元件结合蛋白。
图 1 木犀草素对肝损伤的保护作用机制
Figure 1. Diagram of the protective mechanism of luteolin on liver injury
-
[1] MAO JX, TAN LH, TIAN C, et al. Research progress on rodent models and its mechanisms of liver injury[J]. Life Sci, 2024, 337: 122343. DOI: 10.1016/j.lfs.2023.122343. [2] WEI HW, ZHAO T, LIU XL, et al. Mechanism of action of dihydroquercetin in the prevention and therapy of experimental liver injury[J]. Molecules, 2024, 29( 15): 3537. DOI: 10.3390/molecules29153537. [3] WANG HT, ZHAO ZT, SONG MM, et al. Luteolin detoxifies DEHP and prevents liver injury by degrading Uroc1 protein in mice[J]. EMBO Mol Med, 2024, 16( 11): 2699- 2724. DOI: 10.1038/s44321-024-00160-9. [4] WANG XH, WANG L, DONG RC, et al. Luteolin ameliorates LPS-induced acute liver injury by inhibiting TXNIP-NLRP3 inflammasome in mice[J]. Phytomedicine, 2021, 87: 153586. DOI: 10.1016/j.phymed.2021.153586. [5] TAWEESAP P, POTUE P, KHAMSEEKAEW J, et al. Luteolin relieves metabolic dysfunction-associated fatty liver disease caused by a high-fat diet in rats through modulating the AdipoR1/AMPK/PPARγ signaling pathway[J]. Int J Mol Sci, 2025, 26( 8): 3804. DOI: 10.3390/ijms26083804. [6] DAI HT. The protective effect and mechanism of luteolin on carbon tetrachloride-induced acute liver injury in mice[D]. Changchun: Jilin University, 2023. DOI: 10.27162/d.cnki.gjlin.2023.003139.代惠婷. 木犀草素对四氯化碳诱导小鼠急性肝损伤的保护作用及机制研究[D]. 长春: 吉林大学, 2023. DOI: 10.27162/d.cnki.gjlin.2023.003139. [7] JIANG YH, YANG WJ, DING JM, et al. Luteolin pretreatment attenuates hepatic ischemia-reperfusion injury in mice by inhibiting inflammation, autophagy, and apoptosis via the ERK/PPARα pathway[J]. PPAR Res, 2022, 2022: 8161946. DOI: 10.1155/2022/8161946. [8] ÇETINKAYA M, BARAN Y. Therapeutic potential of luteolin on cancer[J]. Vaccines, 2023, 11( 3): 554. DOI: 10.3390/vaccines11030554. [9] JAYAWICKREME DK, EKWOSI C, ANAND A, et al. Luteolin for neurodegenerative diseases: A review[J]. Pharmacol Rep, 2024, 76( 4): 644- 664. DOI: 10.1007/s43440-024-00610-8. [10] LV JL, SONG XY, LUO ZX, et al. Luteolin: Exploring its therapeutic potential and molecular mechanisms in pulmonary diseases[J]. Front Pharmacol, 2025, 16: 1535555. DOI: 10.3389/fphar.2025.1535555. [11] ALMATROODI SA, ALMATROUDI A, ALHARBI HOA, et al. Effects and mechanisms of luteolin, a plant-based flavonoid, in the prevention of cancers via modulation of inflammation and cell signaling molecules[J]. Molecules, 2024, 29( 5): 1093. DOI: 10.3390/molecules29051093. [12] SADASIVAM N, KIM YJ, RADHAKRISHNAN K, et al. Oxidative stress, genomic integrity, and liver diseases[J]. Molecules, 2022, 27( 10): 3159. DOI: 10.3390/molecules27103159. [13] ZHENG XY, SHU ZH, LI Y, et al. Traditional Chinese medicine regulates oxidative stress-related signaling pathways to prevent and treat bronchial asthma: A review[J]. Chin J Exp Tradit Med Formulae, 2024, 30( 18): 260- 269. DOI: 10.13422/j.cnki.syfjx.20231818.郑旭阳, 舒臻辉, 李怡, 等. 中医药调控氧化应激相关信号通路防治支气管哮喘研究进展[J]. 中国实验方剂学杂志, 2024, 30( 18): 260- 269. DOI: 10.13422/j.cnki.syfjx.20231818. [14] RAJPUT S ALI, SHAUKAT A, WU KT, et al. Luteolin alleviates AflatoxinB1-induced apoptosis and oxidative stress in the liver of mice through activation of Nrf2 signaling pathway[J]. Antioxidants, 2021, 10( 8): 1268. DOI: 10.3390/antiox10081268. [15] WU WY, LI KX, RAN XH, et al. Combination of resveratrol and luteolin ameliorates α-naphthylisothiocyanate-induced cholestasis by regulating the bile acid homeostasis and suppressing oxidative stress[J]. Food Funct, 2022, 13( 13): 7098- 7111. DOI: 10.1039/d2fo00521b. [16] BAIRD L, YAMAMOTO M. The molecular mechanisms regulating the KEAP1-NRF2 pathway[J]. Mol Cell Biol, 2020, 40( 13): e00099-20. DOI: 10.1128/MCB.00099-20. [17] BARDALLO R G, PANISELLO-ROSELLÓ A, SANCHEZ-NUNO S, et al. Nrf2 and oxidative stress in liver ischemia/reperfusion injury[J]. FEBS J, 2022, 289( 18): 5463- 5479. DOI: 10.1111/febs.16336. [18] SARCINELLI C, DRAGIC H, PIECYK M, et al. ATF4-dependent NRF2 transcriptional regulation promotes antioxidant protection during endoplasmic reticulum stress[J]. Cancers, 2020, 12( 3): 569. DOI: 10.3390/cancers12030569. [19] ZHU MT, SUN YP, SU Y, et al. Luteolin: A promising multifunctional natural flavonoid for human diseases[J]. Phytother Res, 2024, 38( 7): 3417- 3443. DOI: 10.1002/ptr.8217. [20] LIU Y, GUO XY, YU L, et al. Luteolin alleviates inorganic mercury-induced liver injury in quails by resisting oxidative stress and promoting mercury ion excretion[J]. Mol Biol Rep, 2023, 50( 1): 399- 408. DOI: 10.1007/s11033-022-08049-x. [21] TARU V, SZABO G, MEHAL W, et al. Inflammasomes in chronic liver disease: Hepatic injury, fibrosis progression and systemic inflammation[J]. J Hepatol, 2024, 81( 5): 895- 910. DOI: 10.1016/j.jhep.2024.06.016. [22] HUI L, KWAN M. New insights in mechanisms and therapeutics for short-and long-term impacts of hepatic ischemia reperfusion injury post liver transplantation[J]. Int J Mol Sci, 2021, 22( 15): 8210. DOI: 10.3390/ijms22158210. [23] WEN YK, LAMBRECHT J, JU C, et al. Hepatic macrophages in liver homeostasis and diseases-diversity, plasticity and therapeutic opportunities[J]. Cell Mol Immunol, 2021, 18( 1): 45- 56. DOI: 10.1038/s41423-020-00558-8. [24] WANG SX, CAO M, XU SH, et al. Luteolin alters macrophage polarization to inhibit inflammation[J]. Inflammation, 2020, 43( 1): 95- 108. DOI: 10.1007/s10753-019-01099-7. [25] DENG LY, YU QL, KUANG G, et al. Luteolin modulates liver macrophage subtype polarization and play protective role in sepsis induced acute hepatic injury[J]. Inflamm Res, 2025, 74( 1): 59. DOI: 10.1007/s00011-025-02026-3. [26] LI ZC, LI DD, CHEN RC, et al. Cell death regulation: A new way for natural products to treat osteoporosis[J]. Pharmacol Res, 2023, 187: 106635. DOI: 10.1016/j.phrs.2022.106635. [27] ZHAO X, NIU Q, DONG HJ, et al. Research progress on regulation of cell death by traditional Chinese medicine in treatment of liver fibrosis[J]. Chin Tradit Herb Drugs, 2025, 56( 3): 1016- 1027. DOI: 10.7501/j.issn.0253-2670.2025.03.026.赵芯, 牛群, 董海舰, 等. 中药调控细胞死亡治疗肝纤维化的研究进展[J]. 中草药, 2025, 56( 3): 1016- 1027. DOI: 10.7501/j.issn.0253-2670.2025.03.026. [28] YU CQ, CHEN P, MIAO LY, et al. The role of the NLRP3 inflammasome and programmed cell death in acute liver injury[J]. Int J Mol Sci, 2023, 24( 4): 3067. DOI: 10.3390/ijms24043067. [29] CARNEIRO BA, EL-DEIRY WS. Targeting apoptosis in cancer therapy[J]. Nat Rev Clin Oncol, 2020, 17( 7): 395- 417. DOI: 10.1038/s41571-020-0341-y. [30] TAN X. Effects of luteolin on quails and its mitigation of liver and kidney injury induced by inorganic mercury[D]. Harbin: Northeast Agricultural University, 2020. DOI: 10.27010/d.cnki.gdbnu.2020.000043.谭斅. 木犀草素对鹌鹑机体影响及其缓解无机汞致肝肾损伤作用[D]. 哈尔滨: 东北农业大学, 2020. DOI: 10.27010/d.cnki.gdbnu.2020.000043. [31] LI XY. Effect of luteolin on proliferation, apoptosis and glycolysis of hepatocellular carcinoma cells(HepG2 and Huh7)[D]. Guiyang: Guizhou Medical University, 2024. DOI: 10.27045/d.cnki.ggyyc.2024.000414.李雪彦. 木犀草素对肝癌细胞(HepG2、Huh7)增殖、凋亡及糖酵解的影响[D]. 贵阳: 贵州医科大学, 2024. DOI: 10.27045/d.cnki.ggyyc.2024.000414. [32] DAR AA, FEHAID A, ALKHATANI S, et al. The protective role of luteolin against the methotrexate-induced hepato-renal toxicity via its antioxidative, anti-inflammatory, and anti-apoptotic effects in rats[J]. Hum Exp Toxicol, 2021, 40( 7): 1194- 1207. DOI: 10.1177/0960327121991905. [33] FENG QZ. Baicalein combined with luteolin against liver cancer by inhibiting the EGFR/ERK/NFκB signaling pathway[D]. Hefei: Anhui Medical University, 2024. DOI: 10.26921/d.cnki.ganyu.2024.000158.冯其柱. 黄芩素联合木犀草素靶向EGFR/ERK/NFκB信号通路抗肝癌机制研究[D]. 合肥: 安徽医科大学, 2024. DOI: 10.26921/d.cnki.ganyu.2024.000158. [34] PAN Q, LUO Y, XIA Q, et al. Ferroptosis and liver fibrosis[J]. Int J Med Sci, 2021, 18( 15): 3361- 3366. DOI: 10.7150/ijms.62903. [35] CAPELLETTI MM, MANCEAU H, PUY H, et al. Ferroptosis in liver diseases: An overview[J]. Int J Mol Sci, 2020, 21( 14): 4908. DOI: 10.3390/ijms21144908. [36] LI J, CAO F, YIN HL, et al. Ferroptosis: Past, present and future[J]. Cell Death Dis, 2020, 11( 2): 88. DOI: 10.1038/s41419-020-2298-2. [37] HAN ZQ, BATUDELIGEN, CHEN HM, et al. Luteolin attenuates CCl4-induced hepatic injury by inhibiting ferroptosis via SLC7A11[J]. BMC Complement Med Ther, 2024, 24( 1): 193. DOI: 10.1186/s12906-024-04486-2. [38] HAN ZQ, BATU DLG, CHEN HM, et al. Luteolin inhibits ferroptosis by down-regulating CISD1 in the treatment of chronic liver injury with Mongolian drug Dedu Gurigumu-7[R]. Inner Mongolia Autonomous Region: Inner Mongolia Minzu University, 2023-03-31.韩志强, 巴图德力根, 陈红梅, 等. 木犀草素通过下调CISD1抑制铁死亡参与蒙药德都古日古木-7治疗慢性肝损伤的机理研究[R]. 内蒙古自治区: 内蒙古民族大学, 2023-03-31. [39] FENG JQ, YE SF, HAI B, et al. RNF115/BCA2 deficiency alleviated acute liver injury in mice by promoting autophagy and inhibiting inflammatory response[J]. Cell Death Dis, 2023, 14( 12): 855. DOI: 10.1038/s41419-023-06379-7. [40] QIAN H, CHAO XJ, WILLIAMS J, et al. Autophagy in liver diseases: A review[J]. Mol Aspects Med, 2021, 82: 100973. DOI: 10.1016/j.mam.2021.100973. [41] ASHRAFIZADEH M, AHMADI Z, FARKHONDEH T, et al. Autophagy regulation using luteolin: New insight into its anti-tumor activity[J]. Cancer Cell Int, 2020, 20( 1): 537. DOI: 10.1186/s12935-020-01634-9. [42] ZHANG MF, SERNA-SALAS S, DAMBA T, et al. Hepatic stellate cell senescence in liver fibrosis: Characteristics, mechanisms and perspectives[J]. Mech Ageing Dev, 2021, 199: 111572. DOI: 10.1016/j.mad.2021.111572. [43] MAO AJ, TIAN HJ, ZHAO Y, et al. Mechanism of luteolin in the treatment of non-alcoholic fatty liver disease: A review[J]. World Chin Med, 2025, 20( 3): 513- 518. DOI: 10.3969/j.issn.1673-7202.2025.03.024.毛傲洁, 田华捷, 赵瑜, 等. 木犀草素治疗非酒精性脂肪性肝病作用机制研究进展[J]. 世界中医药, 2025, 20( 3): 513- 518. DOI: 10.3969/j.issn.1673-7202.2025.03.024. [44] LIAO ZH, XIE ZY. Research progress in molecular mechanism of hepatic fibrosis and related therapeutic targets[J]. J Jilin Univ Med Ed, 2024, 50( 5): 1450- 1456. DOI: 10.13481/j.1671-587X.20240532.廖昭辉, 谢正元. 肝纤维化发病的分子机制及其相关治疗靶点的研究进展[J]. 吉林大学学报(医学版), 2024, 50( 5): 1450- 1456. DOI: 10.13481/j.1671-587X.20240532. [45] ZHOU X, WANG Z, HE XR, et al. Research advances in signaling pathways associated with potential anti-liver fibrosis drugs and targets[J]. J Clin Hepatol, 2023, 39( 12): 2932- 2941. DOI: 10.3969/j.issn.1001-5256.2023.12.027.周鑫, 王智, 何雪茹, 等. 潜在抗肝纤维化药物与靶点相关信号通路研究进展[J]. 临床肝胆病杂志, 2023, 39( 12): 2932- 2941. DOI: 10.3969/j.issn.1001-5256.2023.12.027. [46] ASHOUR AA, EL-KAMEL AH, MEHANNA RA, et al. Luteolin-loaded exosomes derived from bone marrow mesenchymal stem cells: A promising therapy for liver fibrosis[J]. Drug Deliv, 2022, 29( 1): 3270- 3280. DOI: 10.1080/10717544.2022.2142700. [47] BATUDELIGEN, HAN ZQ, CHEN HM, et al. Luteolin alleviates liver fibrosis in rat hepatic stellate cell HSC-T6: A proteomic analysis[J]. Drug Des Devel Ther, 2023, 17: 1819- 1829. DOI: 10.2147/DDDT.S402864. [48] LIU YD, SUN ZY, DONG RX, et al. Rutin ameliorated lipid metabolism dysfunction of diabetic NAFLD via AMPK/SREBP1 pathway[J]. Phytomedicine, 2024, 126: 155437. DOI: 10.1016/j.phymed.2024.155437. [49] LIU YS, YUAN MH, ZHANG CY, et al. Puerariae Lobatae Radix flavonoids and puerarin alleviate alcoholic liver injury in zebrafish by regulating alcohol and lipid metabolism[J]. Biomed Pharmacother, 2021, 134: 111121. DOI: 10.1016/j.biopha.2020.111121. [50] LI N, LI XD, DING YF, et al. SREBP regulation of lipid metabolism in liver disease, and therapeutic strategies[J]. Biomedicines, 2023, 11( 12): 3280. DOI: 10.3390/biomedicines11123280. [51] GUO W, LUO LX, MENG Y, et al. Luteolin alleviates methionine-choline-deficient diet-induced non-alcoholic steatohepatitis by modulating host serum metabolome and gut microbiome[J]. Front Nutr, 2022, 9: 936237. DOI: 10.3389/fnut.2022.936237. [52] LIU Y, LI GY, LU FY, et al. Excess iron intake induced liver injury: The role of gut-liver axis and therapeutic potential[J]. Biomed Pharmacother, 2023, 168: 115728. DOI: 10.1016/j.biopha.2023.115728. [53] ZHANG P, ZHENG LY, DUAN YT, et al. Gut microbiota exaggerates triclosan-induced liver injury via gut-liver axis[J]. J Hazard Mater, 2022, 421: 126707. DOI: 10.1016/j.jhazmat.2021.126707. [54] SUN WL, YANG JW, DOU HY, et al. Anti-inflammatory effect of luteolin is related to the changes in the gut microbiota and contributes to preventing the progression from simple steatosis to nonalcoholic steatohepatitis[J]. Bioorg Chem, 2021, 112: 104966. DOI: 10.1016/j.bioorg.2021.104966. [55] ZOU H, ALI W, DENG K, et al. The protective effect of luteolin on cadmium induced liver intestinal toxicity in chicken by Gut-liver axis regulation[J]. Poult Sci, 2024, 103( 11): 104242. DOI: 10.1016/j.psj.2024.104242. [56] LAI XJ, LIN LW, LIU HL, et al. Protective effect of luteolin on liver function in rats with autoimmune hepatitis and its mechanism[J]. Immunol J, 2024, 40( 5): 452- 457. DOI: 10.13431/j.cnki.immunol.j.20240059.赖玺杰, 林列威, 刘洪亮, 等. 木犀草素对大鼠自身免疫性肝炎的肝功能保护作用及其机制研究[J]. 免疫学杂志, 2024, 40( 5): 452- 457. DOI: 10.13431/j.cnki.immunol.j.20240059. [57] GAO L, SONG KX, REN JP, et al. Effects of luteolin on the activities of cytochrome P450 enzymes in rats by Cocktail probe approach[J]. Chin J New Drugs, 2022, 31( 6): 590- 595. DOI: 10.3969/j.issn.1003-3734.2022.06.011.高琳, 宋可心, 任纪平, 等. Cocktail探针药物法评价木犀草素对大鼠CYP450酶活性影响[J]. 中国新药杂志, 2022, 31( 6): 590- 595. DOI: 10.3969/j.issn.1003-3734.2022.06.011. -



 PDF下载 ( 1713 KB)
PDF下载 ( 1713 KB)

 下载:
下载:


