线粒体钙单向转运体在急性胰腺炎小鼠模型胰腺导管上皮细胞骨架中的作用及其机制
DOI: 10.12449/JCH260220
Role and mechanism of mitochondrial calcium uniporter in the cytoskeleton of pancreatic ductal epithelial cells in a mouse model of acute pancreatitis
-
摘要:
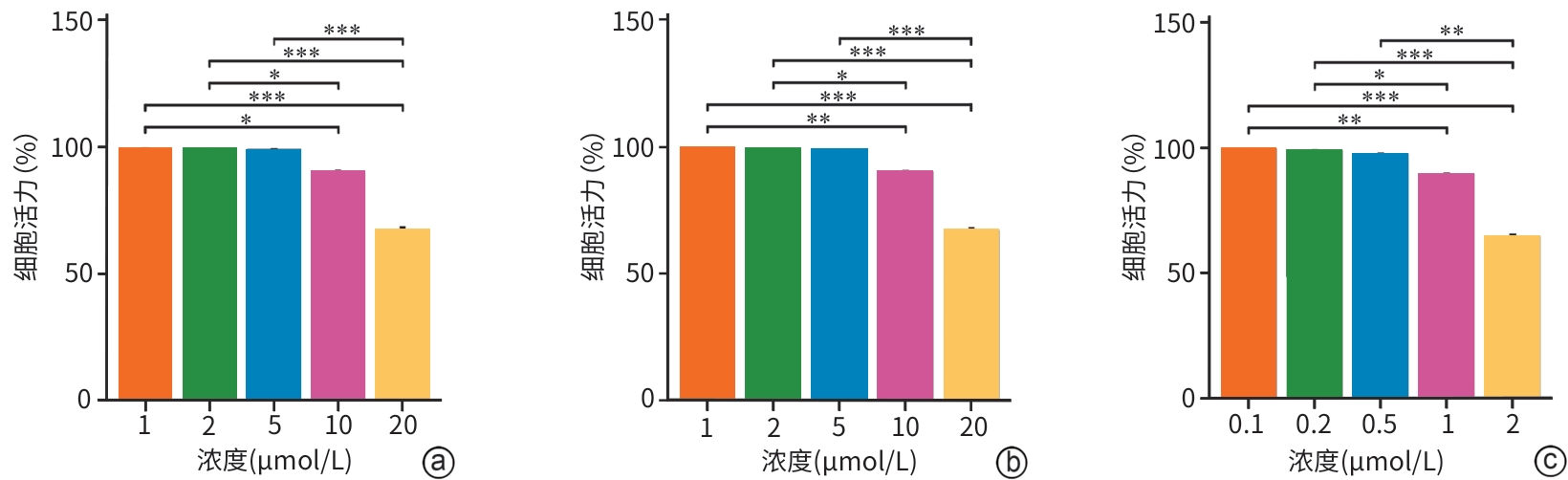
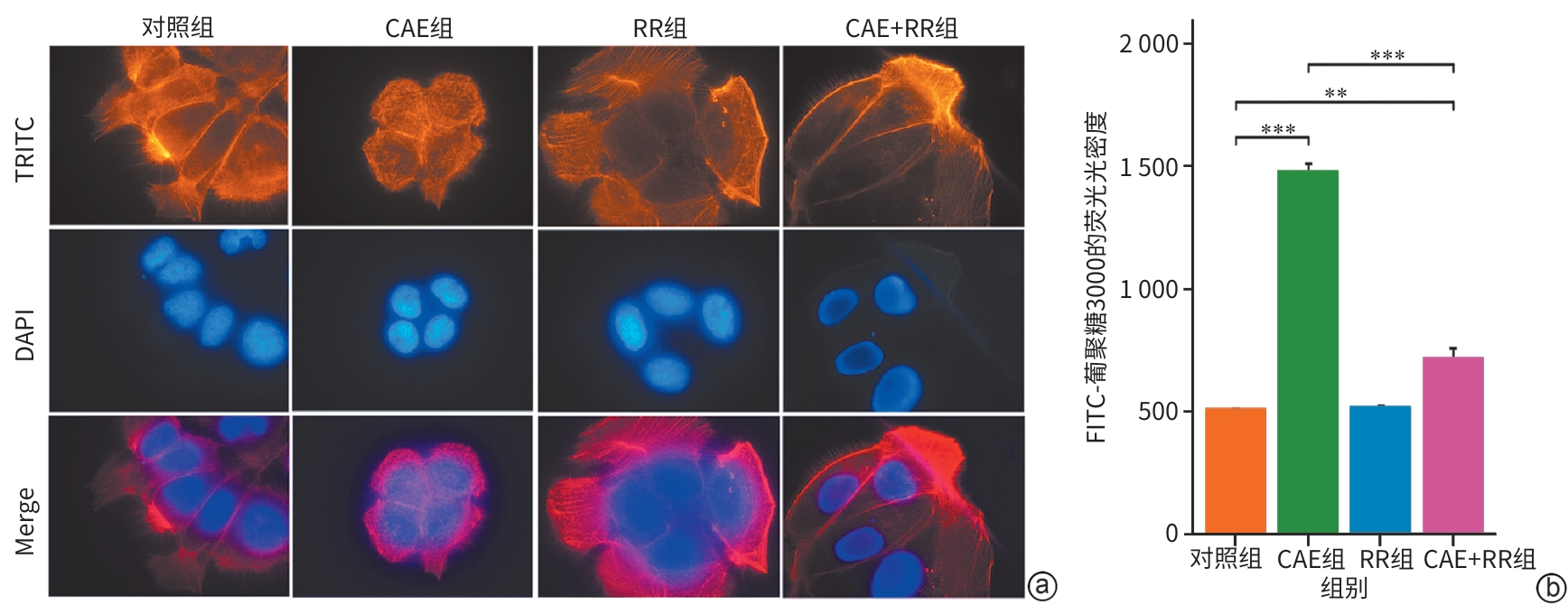
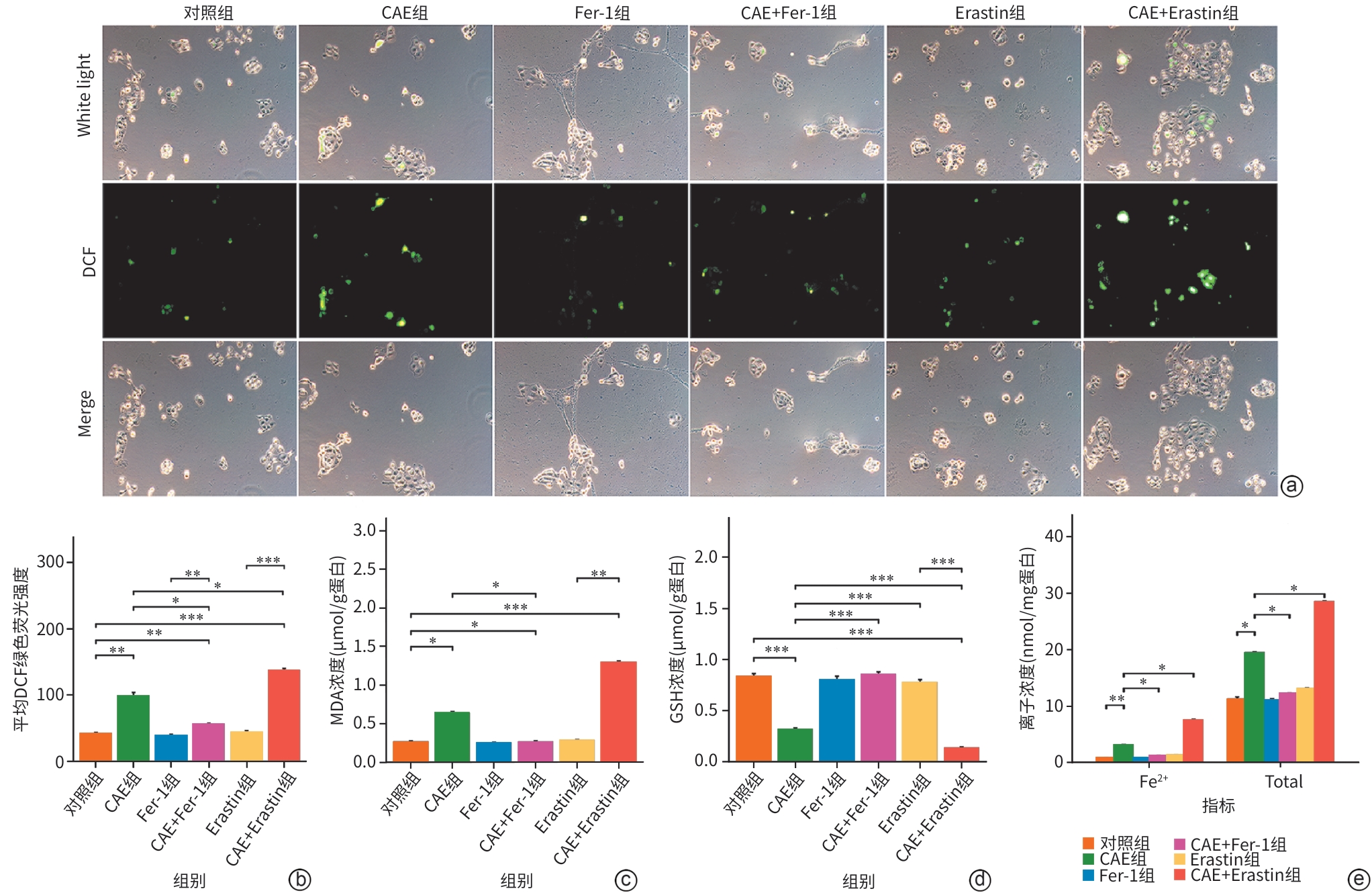
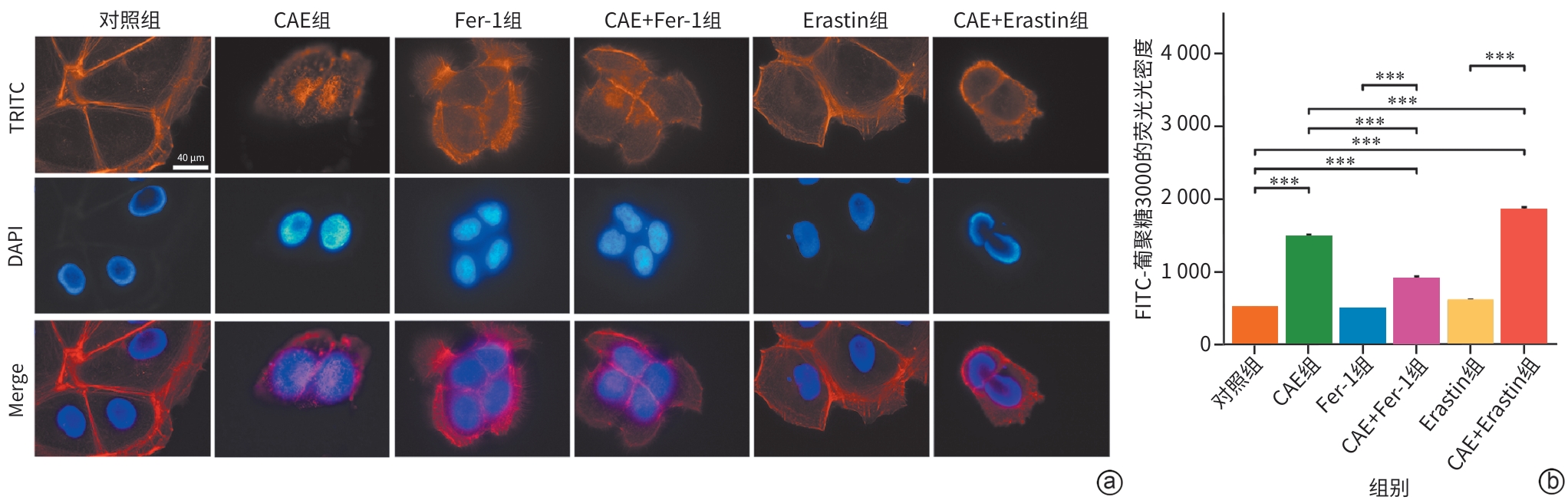
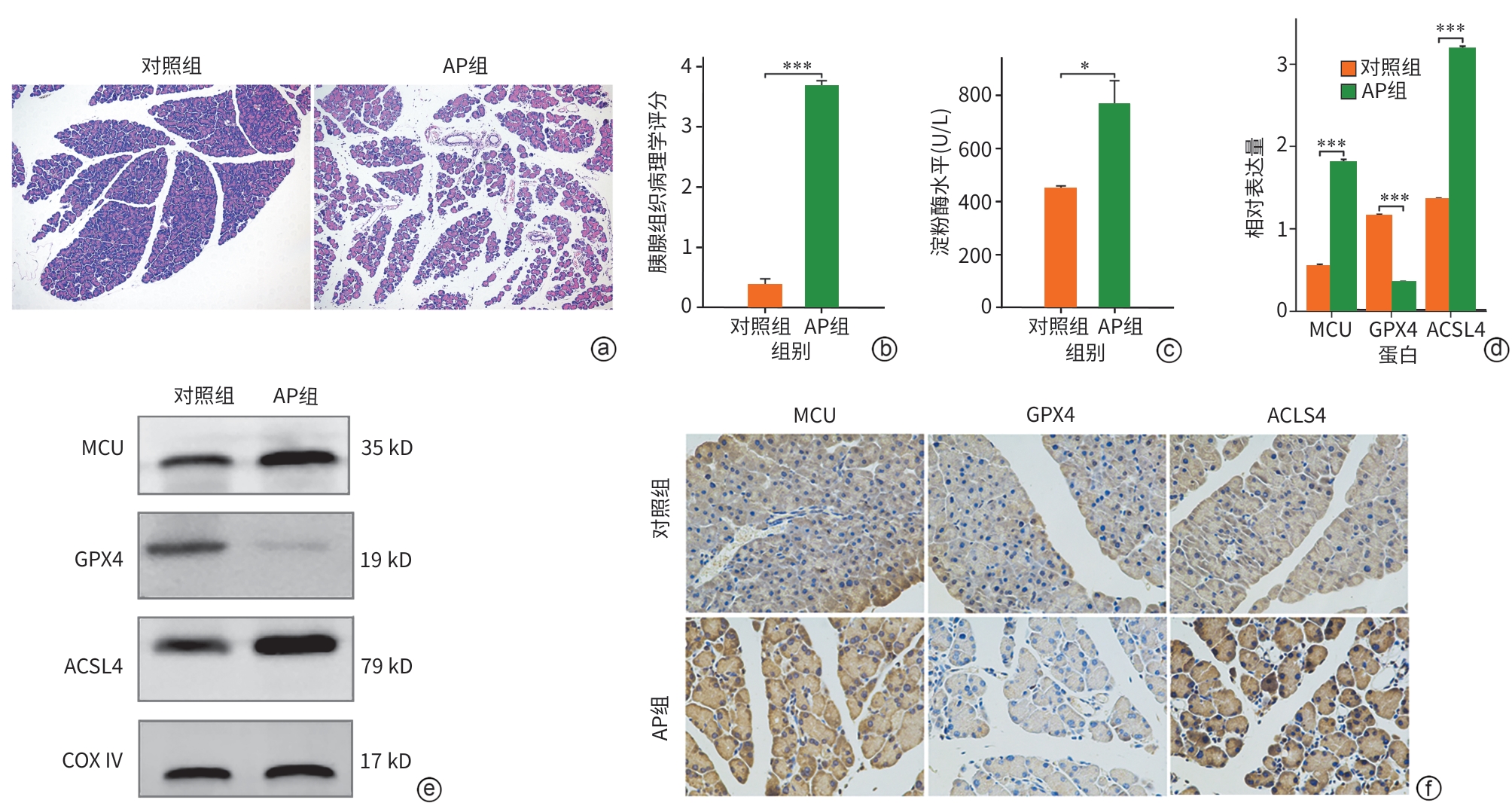
目的 探讨线粒体钙单向转运体(MCU)调控铁死亡对雨蛙肽(CAE)诱导的急性胰腺炎(AP)小鼠模型胰腺导管上皮细胞骨架的影响,了解MCU在AP发病过程中的作用,进而为临床治疗提供理论基础。 方法 动物实验:将4周龄野生型雄性C57BL6/J小鼠随机分为对照组和AP组,每组6只。AP组通过腹腔注射CAE构建AP模型,对照组腹腔注射等量的生理盐水。造模24 h后收集小鼠血清和胰腺组织。通过HE染色观察胰腺组织病理变化。蛋白免疫印迹法检测MCU、谷胱甘肽过氧化物酶4(GPX4)和长链脂酰辅酶A合成酶4(ASCL4)的表达水平。使用试剂盒检测小鼠血清淀粉酶水平。细胞实验:人胰腺导管上皮细胞系HPDE6-C7与CAE共培养24 h建立体外AP模型,分为对照组、CAE组、RR(MCU活性抑制剂)组、CAE+RR组、Fer-1(铁死亡抑制剂)组、CAE+Fer-1组、Erastin(铁死亡诱导剂)组、CAE+Erastin组。CCK-8检测不同干预剂对细胞活力的影响。蛋白免疫印迹法检测MCU、GPX4和ASCL4的表达水平。免疫荧光检测活性氧(ROS)、微丝细胞骨架和单层细胞通透性。试剂盒检测丙二醛(MDA)、谷胱甘肽(GSH)、Fe2+和总铁浓度。计量资料多组间比较采用单因素方差分析,进一步两两比较采用LSD-t检验。 结果 动物实验:与对照组相比,AP组小鼠胰腺组织学病理评分和淀粉酶水平均显著升高,MCU和ASCL4表达显著升高、GPX4表达显著下降(P值均<0.05)。细胞实验:与对照组相比,CAE组MCU和ASCL4表达升高、GPX4表达下降,Fe2+和总铁浓度、MDA浓度、ROS绿色荧光强度、单层细胞通透性均显著升高,GSH浓度下降(P值均<0.05),并出现细胞微丝骨架破坏。与CAE组相比,CAE+RR组GPX4表达升高、ASCL4表达下降,Fe2+和总铁浓度、MDA浓度、ROS绿色荧光强度、单层细胞通透性均显著性下降,GSH浓度上升(P值均<0.05),细胞微丝骨架破坏程度减轻。与CAE组相比,CAE+Fer-1组Fe2+和总铁浓度、MDA浓度、ROS绿色荧光强度、单层细胞通透性均显著下降,GSH浓度显著上升(P值均<0.05),细胞微丝骨架破坏程度减轻。与CAE组相比,CAE+Erastin组Fe2+和总铁浓度、MDA浓度、ROS绿色荧光强度、单层细胞通透性均显著升高,GSH浓度显著下降(P值均<0.05),细胞微丝骨架破坏程度减轻。 结论 在AP发病时,MCU介导氧化应激引发铁死亡导致胰腺导管上皮细胞屏障破坏,可能是AP的发病机制之一。 Abstract:Objective To investigate the effect of mitochondrial calcium uniporter (MCU) on the cytoskeleton of pancreatic ductal epithelial cells in a mouse model of acute pancreatitis (AP) induced by caerulein (CAE), to analyze the role of MCU in the development of AP, and to provide a theoretical basis for clinical treatment. Methods In the in vivo experiment, wild-type male C57BL6/J mice, aged 4 weeks, were randomly divided into control group and AP group, with 6 mice in each group. The mice in the AP group were given intraperitoneal injection of CAE to establish a model of AP, and those in the control group were given intraperitoneal injection of an equal volume of normal saline. Serum and pancreatic tissue samples were collected after 24 hours of modeling. HE staining was used to observe pancreatic histopathological changes; Western Blot was used to measure the expression levels of MCU, glutathione peroxidase 4 (GPX4), and acyl-CoA synthetase long chain family member 4 (ASCL4); kits were used to measure the serum level of amylase. In the in vitro experiment, the human pancreatic ductal epithelial cell line HPDE6-C7 was co-cultured with CAE for 24 hours to establish an in vitro AP model, and the cells were divided into control group, CAE group, RR (an MCU activity inhibitor) group, CAE+RR group, Fer-1 (an ferroptosis inhibitor) group, CAE+Fer-1 group, Erastin (an ferroptosis inducer) group, and CAE+Erastin group. CCK-8 assay was used to observe the influence of different agents on cell viability; Western Blot was used to measure the expression levels of MCU, GPX4, and ASCL4; immunofluorescence assay was used to measure reactive oxygen species (ROS), actin cytoskeleton, and monolayer permeability; kits were used to measure the concentrations of malondialdehyde (MDA), glutathione (GSH), Fe2+, and total iron. A one-way analysis of variance was used for comparison of continuous data between multiple groups, and the least significant difference t-test was used for comparison between two groups. Results In the in vivo experiment, compared with the control group, the AP group had significant increases in pancreatic histopathological score, the serum level of amylase, and the expression levels of MCU and ASCL4, as well as a significant reduction in the expression of GPX4 (all P<0.05). In the in vitro experiment, compared with the control group, the CAE group had significant increases in the expression levels of MCU and ASCL4, a significant reduction in the expression of GPX4, and significant increases in the concentrations of Fe2+, total iron, and MDA, the green fluorescence intensity of ROS, and monolayer permeability, as well as a significant reduction in the concentration of GSH (all P<0.05), with the presence of actin cytoskeleton disruption. Compared with the CAE group, the CAE+RR group had a significant increase in the expression level of GPX4, a significant reduction in the expression level of ASCL4, and significant reductions in the concentrations of Fe2+, total iron, and MDA, the green fluorescence intensity of ROS, and monolayer permeability and a significant increase in the concentration of GSH (all P<0.05), with alleviation of actin cytoskeleton disruption. Compared with the CAE group, the CAE+Fer-1 group had significant reductions in the concentrations of Fe2+, total iron, and MDA, the green fluorescence intensity of ROS, and monolayer permeability and a significant increase in the concentration of GSH (all P<0.05), with alleviation of actin cytoskeleton disruption. Compared with the CAE group, the CAE+Erastin group had significant increases in the concentrations of Fe2+, total iron, and MDA, the green fluorescence intensity of ROS, and monolayer permeability and a significant reduction in the concentration of GSH (all P<0.05), with aggravation of actin cytoskeleton disruption. Conclusion During the onset of AP, MCU mediates oxidative stress-induced ferroptosis and leads to the disruption of the pancreatic ductal epithelial barrier, which may be one of the possible pathogeneses of AP. -
Key words:
- Pancreatitis /
- Cytoskeleton /
- Models, Animal
-
注: a,各组小鼠胰腺组织HE染色(×200);b,各组小鼠胰腺组织病理学评分;c,各组小鼠血清淀粉酶水平;d,各组小鼠MCU、GPX4和ACSL4的Western Blot条带;e,各组MCU、GPX4和ACSL4较内参的相对表达量;f,各组小鼠胰腺组织MCU、GPX4和ACSL4的免疫组化染色(×400)。*P<0.05,***P<0.001。AP,急性胰腺炎;MCU,线粒体钙单向转运体;GPX4,谷胱甘肽过氧化物酶4;ACSL4,长链脂酰辅酶A合成酶4;COX Ⅳ,线粒体内参抗体。
图 1 各组小鼠胰腺导管组织中MCU及铁死亡相关蛋白的变化
Figure 1. Changes in MCU and ferroptosis-related proteins in pancreatic ductal tissues of mice from each group
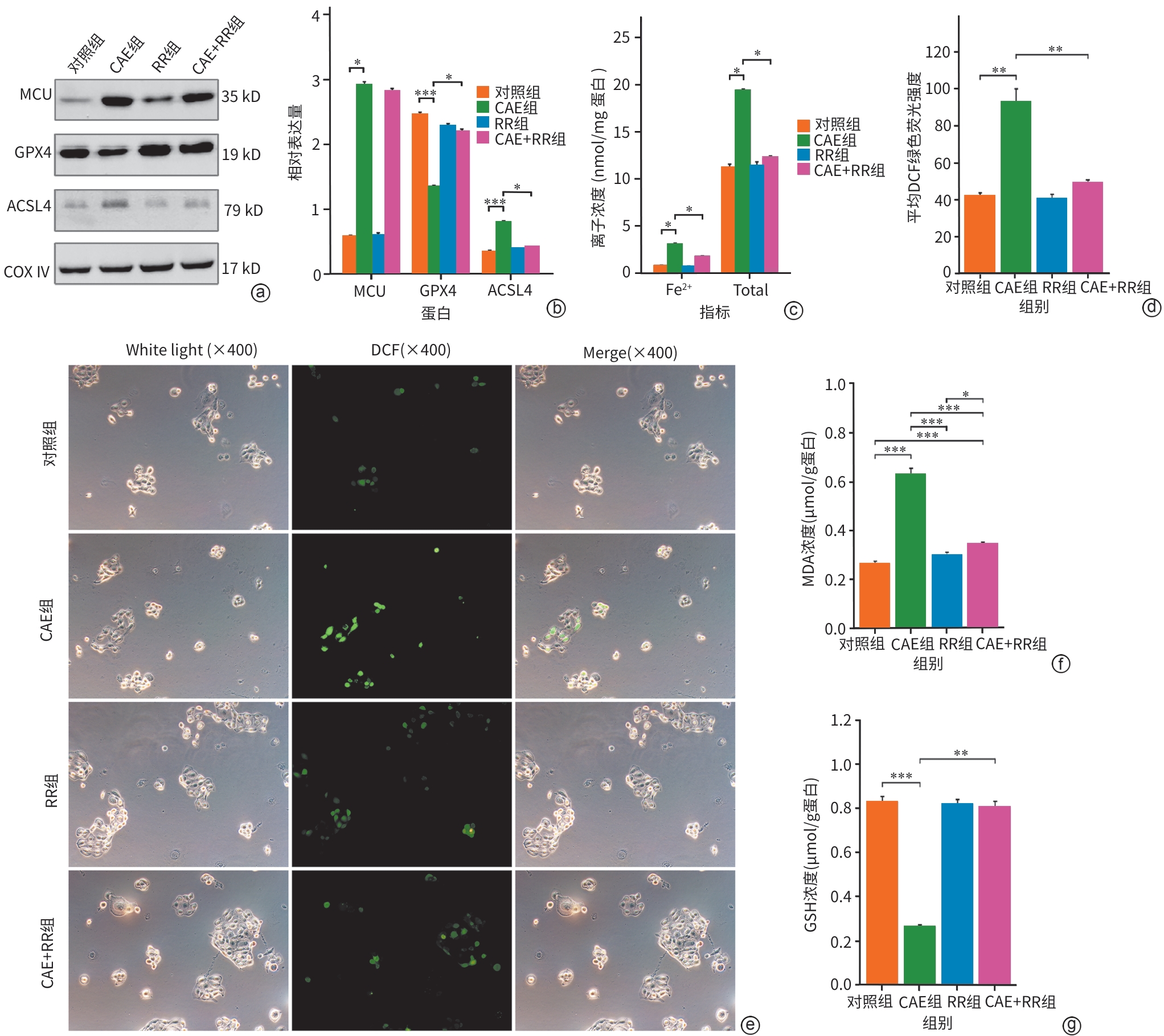
注: a,Western Blot条带图;b,各组MCU、GPX4和ACSL4较内参的相对表达量;c,各组细胞Fe2+和总铁浓度;d,各组细胞DCF绿色荧光强度;e,不同组细胞的ROS免疫荧光图(×400);f,各组细胞MDA浓度;g,各组细胞GSH浓度。*P<0.05,**P<0.01,***P<0.001。MCU,线粒体钙单向转运体;GPX4,谷胱甘肽过氧化物酶4;ACSL4,长链脂酰辅酶A合成酶4;COX Ⅳ,线粒体内参抗体;CAE,雨蛙肽;RR,线粒体钙单向转运体活性抑制剂;MDA,丙二醛;GSH,谷胱甘肽。
图 3 各组MCU、铁死亡特征性蛋白、Fe2+浓度、氧化应激指标的变化
Figure 3. Changes in MCU expression, characteristic ferroptosis proteins, iron ion concentration, and oxidative stress indicators across groups
-
[1] BOXHOORN L, VOERMANS RP, BOUWENSE SA, et al. Acute pancreatitis[J]. Lancet, 2020, 396( 10252): 726- 734. DOI: 10.1016/S0140-6736(20)31310-6. [2] PETROV MS, YADAV D. Global epidemiology and holistic prevention of pancreatitis[J]. Nat Rev Gastroenterol Hepatol, 2019, 16( 3): 175- 184. DOI: 10.1038/s41575-018-0087-5. [3] SCHEPERS NJ, BAKKER OJ, BESSELINK MG, et al. Impact of characteristics of organ failure and infected necrosis on mortality in necrotising pancreatitis[J]. Gut, 2019, 68( 6): 1044- 1051. DOI: 10.1136/gutjnl-2017-314657. [4] YIN JL, ZHAO MM, WANG Y, et al. Analysis of dlinical characteristics and inlluencing factors of disease Prognosis in severe acute pancreatitis at different stages[J]. J Clin Exp Med, 2024, 23( 7): 698- 702. DOI: 10.3969/j.issn.1671-4695.2024.07.007.殷将领, 赵茗茗, 王尧, 等. 不同时期重症急性胰腺炎临床特点及疾病转归的影响因素分析[J]. 临床和实验医学杂志, 2024, 23( 7): 698- 702. DOI: 10.3969/j.issn.1671-4695.2024.07.007. [5] HABTEZION A, GUKOVSKAYA AS, PANDOL SJ. Acute pancreatitis: A multifaceted set of organelle and cellular interactions[J]. Gastroenterology, 2019, 156( 7): 1941- 1950. DOI: 10.1053/j.gastro.2018.11.082. [6] WALDRON RT, CHEN YF, PHAM H, et al. The Orai Ca2+ channel inhibitor CM4620 targets both parenchymal and immune cells to reduce inflammation in experimental acute pancreatitis[J]. J Physiol, 2019, 597( 12): 3085- 3105. DOI: 10.1113/JP277856. [7] DELIERNEUX C, KOUBA S, SHANMUGHAPRIYA S, et al. Mitochondrial calcium regulation of redox signaling in cancer[J]. Cells, 2020, 9( 2): 432. DOI: 10.3390/cells9020432. [8] PENG TI, JOU MJ. Oxidative stress caused by mitochondrial calcium overload[J]. Ann N Y Acad Sci, 2010, 1201: 183- 188. DOI: 10.1111/j.1749-6632.2010.05634.x. [9] MUÑOZ-CASARES FC, PADILLO FJ, BRICEÑO J, et al. Melatonin reduces apoptosis and necrosis induced by ischemia/reperfusion injury of the pancreas[J]. J Pineal Res, 2006, 40( 3): 195- 203. DOI: 10.1111/j.1600-079X.2005.00291.x. [10] KONOK GP, THOMPSON AG. Pancreatic ductal mucosa as a protective barrier in the pathogenesis of pancreatitis[J]. Am J Surg, 1969, 117( 1): 18- 23. DOI: 10.1016/0002-9610(69)90280-3. [11] JIANG XJ, STOCKWELL BR, CONRAD M. Ferroptosis: Mechanisms, biology and role in disease[J]. Nat Rev Mol Cell Biol, 2021, 22( 4): 266- 282. DOI: 10.1038/s41580-020-00324-8. [12] XIE Y, HOU W, SONG X, et al. Ferroptosis: Process and function[J]. Cell Death Differ, 2016, 23( 3): 369- 379. DOI: 10.1038/cdd.2015.158. [13] YANG WS, SRIRAMARATNAM R, WELSCH ME, et al. Regulation of ferroptotic cancer cell death by GPX4[J]. Cell, 2014, 156( 1-2): 317- 331. DOI: 10.1016/j.cell.2013.12.010. [14] DIXON SJ, WINTER GE, MUSAVI LS, et al. Human haploid cell genetics reveals roles for lipid metabolism genes in nonapoptotic cell death[J]. ACS Chem Biol, 2015, 10( 7): 1604- 1609. DOI: 10.1021/acschembio.5b00245. [15] DOLL S, PRONETH B, TYURINA YY, et al. ACSL4 dictates ferroptosis sensitivity by shaping cellular lipid composition[J]. Nat Chem Biol, 2017, 13( 1): 91- 98. DOI: 10.1038/nchembio.2239. [16] KAGAN VE, MAO GW, QU F, et al. Oxidized arachidonic and adrenic PEs navigate cells to ferroptosis[J]. Nat Chem Biol, 2017, 13( 1): 81- 90. DOI: 10.1038/nchembio.2238. [17] PARK EJ, PARK YJ, LEE SJ, et al. Whole cigarette smoke condensates induce ferroptosis in human bronchial epithelial cells[J]. Toxicol Lett, 2019, 303: 55- 66. DOI: 10.1016/j.toxlet.2018.12.007. [18] DAI EY, HAN L, LIU J, et al. Ferroptotic damage promotes pancreatic tumorigenesis through a TMEM173/STING-dependent DNA sensor pathway[J]. Nat Commun, 2020, 11( 1): 6339. DOI: 10.1038/s41467-020-20154-8. [19] FAN R, SUI JD, DONG XP, et al. Wedelolactone alleviates acute pancreatitis and associated lung injury via GPX4 mediated suppression of pyroptosis and ferroptosis[J]. Free Radic Biol Med, 2021, 173: 29- 40. DOI: 10.1016/j.freeradbiomed.2021.07.009. [20] MA DL, LI C, JIANG PL, et al. Inhibition of ferroptosis attenuates acute kidney injury in rats with severe acute pancreatitis[J]. Dig Dis Sci, 2021, 66( 2): 483- 492. DOI: 10.1007/s10620-020-06225-2. [21] MA DL, JIANG PL, JIANG YJ, et al. Effects of lipid peroxidation-mediated ferroptosis on severe acute pancreatitis-induced intestinal barrier injury and bacterial translocation[J]. Oxid Med Cell Longev, 2021, 2021: 6644576. DOI: 10.1155/2021/6644576. [22] van LAETHEM JL, MARCHANT A, DELVAUX A, et al. Interleukin 10 prevents necrosis in murine experimental acute pancreatitis[J]. Gastroenterology, 1995, 108( 6): 1917- 1922. DOI: 10.1016/0016-5085(95)90158-2. [23] LEI Y, YANG HY, MENG N, et al. Mitochondrial calcium uniporter promotes mitophagy by regulating the PINK1/Parkin pathway in caerulein-treated pancreatic ductal epithelial cells in vitro[J]. Exp Ther Med, 2024, 27( 4): 147. DOI: 10.3892/etm.2024.12435. [24] YU XY, DAI C, ZHAO XM, et al. Ruthenium red attenuates acute pancreatitis by inhibiting MCU and improving mitochondrial function[J]. Biochem Biophys Res Commun, 2022, 635: 236- 243. DOI: 10.1016/j.bbrc.2022.10.044. [25] LI C, SUN JY, LING HL, et al. MCU regulating bone remodeling and osteogenic function through mitochondrial calcium homeostasis and oxidative stress alteration[J]. Free Radic Biol Med, 2025, 236: 87- 97. DOI: 10.1016/j.freeradbiomed.2025.05.001. [26] LI ZX, RAN Q, QU C, et al. Sigma-1 receptor activation attenuates DOX-induced cardiotoxicity by alleviating endoplasmic reticulum stress and mitochondrial calcium overload via PERK and IP3R-VDAC1-MCU signaling pathways[J]. Biol Direct, 2025, 20( 1): 23. DOI: 10.1186/s13062-025-00617-y. [27] XING MY, LIANG S, CAO W, et al. Annexin A3 represses endothelial permeability and inflammation during sepsis via actin cytoskeleton modulation[J]. Adv Sci, 2025, 12( 22): 2416904. DOI: 10.1002/advs.202416904. [28] WANG XC, LI YZ, LI ZK, et al. Mitochondrial calcium uniporter drives metastasis and confers a targetable cystine dependency in pancreatic cancer[J]. Cancer Res, 2022, 82( 12): 2254- 2268. DOI: 10.1158/0008-5472.CAN-21-3230. [29] STEJEREAN-TODORAN I, ZIMMERMANN K, GIBHARDT CS, et al. MCU controls melanoma progression through a redox-controlled phenotype switch[J]. EMBO Rep, 2022, 23( 11): e54746. DOI: 10.15252/embr.202254746. [30] ZHANG LL, WANG KP, JIANG WR, et al. Neferine ameliorates severe acute pancreatitis-associated intestinal injury by promoting NRF2-mediated ferroptosis[J]. Int J Biol Sci, 2025, 21( 7): 3247- 3261. DOI: 10.7150/ijbs.112888. [31] LI J, JIA YC, ZHANG HY, et al. Nrf2 ameliorates defective autophagic processes and thereby inhibits ferroptosis in acute pancreatitis by suppressing Beclin1-Slc7a11 complex formation[J]. Free Radic Biol Med, 2025, 230: 294- 308. DOI: 10.1016/j.freeradbiomed.2025.02.011. [32] REN SY, WANG JY, DONG ZC, et al. Perfluorooctane sulfonate induces ferroptosis-dependent non-alcoholic steatohepatitis via autophagy-MCU-caused mitochondrial calcium overload and MCU-ACSL4 interaction[J]. Ecotoxicol Environ Saf, 2024, 280: 116553. DOI: 10.1016/j.ecoenv.2024.116553. [33] LI Y, CUI H, XU WX, et al. Selenium represses microRNA-202-5p/MICU1 aixs to attenuate mercuric chloride-induced kidney ferroptosis[J]. Poult Sci, 2024, 103( 8): 103891. DOI: 10.1016/j.psj.2024.103891. -



 PDF下载 ( 47391 KB)
PDF下载 ( 47391 KB)

 下载:
下载: