多房棘球蚴病患者钙盐沉积的影响因素分析
DOI: 10.12449/JCH260217
Influencing factors for calcium salt deposition in patients with alveolar echinococcosis
-
摘要:
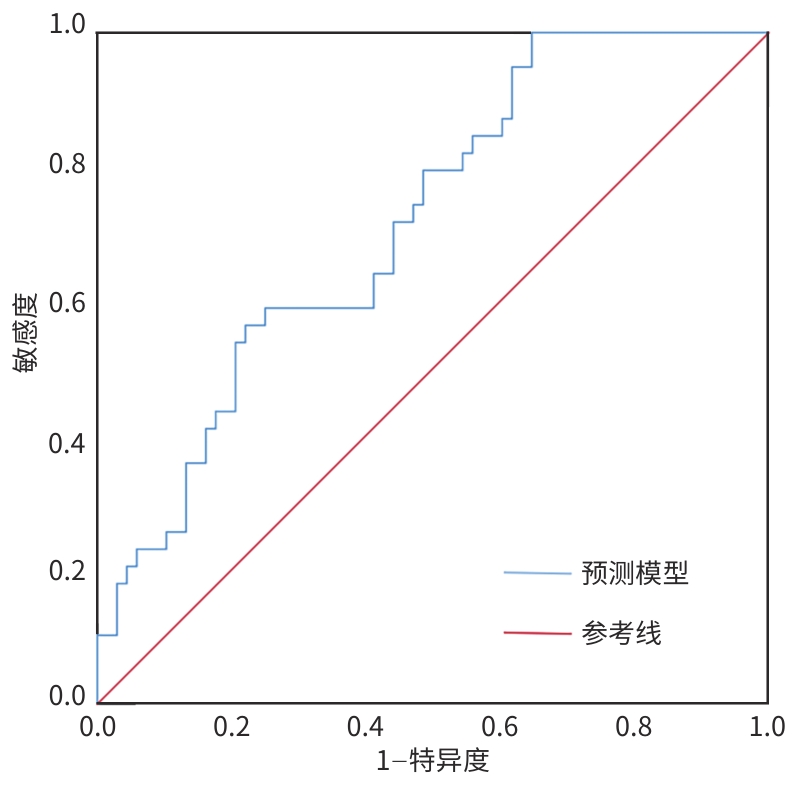
目的 回顾性分析多房棘球蚴病患者的影像学钙盐沉积特征与血清学指标,了解影响病灶钙盐沉积程度的独立影响因素,进而为评估疾病进程提供依据。 方法 回顾性收集石河子大学第一附属医院2023年12月—2025年6月收治的107例多房棘球蚴病患者的影像和临床资料,根据钙盐沉积体积比分为无沉积组(n=16)、轻度沉积组(n=52)、中度沉积组(n=16)和重度沉积组(n=23)。计量资料组间比较采用单因素方差分析或Kruskal-Wallis H检验,计数资料组间比较采用χ2检验或Fisher精确检验。进一步将4组合并为低度沉积组(无沉积组+轻度沉积组)和高度沉积组(中度沉积组+重度沉积组),采用二分类Logistic回归分析钙盐沉积的独立影响因素,并建立预测模型。通过接受者操作特征曲线评估模型预测效能,并使用Bootstrap法进行内部验证。 结果 性别分布、疾病是否累及其他部位,以及白细胞计数、淋巴细胞百分比、纤维蛋白原、尿酸、钠离子、氯离子及钙离子水平4组间比较差异均有统计学意义(P值均<0.0.5)。单因素分析显示,性别、是否累及其他部位、白细胞计数、淋巴细胞百分比、纤维蛋白原、丙氨酸氨基转移酶、白蛋白、肌酐、尿酸、钠离子、氯离子和钙离子4组间比较P值均<0.1。多重共线性诊断显示,所有连续变量的VIF值为1.104~1.760,表明共线性问题不影响建模。最终选取性别、是否累及其他部位、钙离子、淋巴细胞百分比和尿酸构建有序Logistic回归模型。多因素分析显示,淋巴细胞百分比[比值比(OR)=1.106,95%置信区间(CI):1.041~1.174,P=0.001]和血钙水平(OR=0.005,95%CI:0.000~0.230,P=0.007)是钙盐沉积程度的独立影响因素。模型回归方程为:Logit(P)=8.231+0.100×淋巴细胞百分比-5.344×钙离子。受者操作特征曲线分析显示,曲线下面积为0.716,约登指数为0.353,敏感度为1.000,特异度为0.353。Hosmer-Lemeshow检验提示模型校准度较差(χ2=20.688,P=0.008)。经Bootstrap法1 000次重复抽样验证,淋巴细胞百分比(OR=1.106,95%CI:1.049~1.186,P=0.002)和钙离子(OR=0.005,95%CI:0.000~0.214,P=0.010)的估计值与原始模型一致,置信区间均未包含1,进一步支持模型的可靠性。 结论 淋巴细胞百分比及血钙水平均为多房棘球蚴病钙盐沉积的独立影响因素,血钙越低、淋巴细胞百分比越高,多房棘球蚴病灶的钙盐沉积程度越严重。 Abstract:Objective To investigate the imaging features of calcium salt deposition and serological markers in patients with alveolar echinococcosis through a retrospective analysis, as well as independent risk factors for the degree of calcium salt deposition in lesions, and to provide a basis for assessing disease process. Methods A retrospective analysis was performed for the imaging and clinical data of 107 patients with alveolar echinococcosis who were admitted to The First Affiliated Hospital of Shihezi University from December 2023 to June 2025, and according to the volume of calcium salt deposition, they were divided into non-deposition group with 16 patients, mild deposition group with 52 patients, moderate deposition group with 16 patients, and severe deposition group with 23 patients. A one-way analysis of variance or the Kruskal-Wallis H test was used for comparison of continuous data between groups, and the χ2 test or Fisher’s exact test was used for comparison of categorical data between groups. The four groups were further combined into the low deposition group (no/mild deposition) and the high deposition group (moderate/severe deposition). A binary logistic regression analysis was used to investigate the independent influencing factors for calcium salt deposition, and a predictive model was established. The receiver operating characteristic (ROC) curve was used to assess the predictive performance of the model, and the Bootstrap method was used for internal validation. Results There were significant differences between the four groups in sex distribution, involvement of other sites, white blood cell count, lymphocyte percentage, fibrinogen, uric acid, sodium ion, chloride ion, and calcium ion (all P<0.05). The univariate analysis showed that there were significant differences between the four groups in sex, involvement of other sites, white blood cell count, lymphocyte percentage, fibrinogen, alanine aminotransferase, albumin, creatinine, uric acid, sodium ion, chloride ion, and calcium ion (all P<0.1). The multi-collinearity diagnosis showed that the VIF values for all continuous variables ranged from 1.104 to 1.760, suggesting that collinearity did not affect modeling. An ordinal logistic regression model was established based on sex, involvement of other sites, calcium ion, lymphocyte percentage, and uric acid. The multivariate analysis showed that lymphocyte percentage (odds ratio [OR]=1.106, 95% confidence interval [CI]: 1.041 — 1.174, P=0.001) and blood calcium level (OR=0.005, 95%CI: 0.000 —0.230, P=0.007) were independent influencing factors for the degree of calcium salt deposition. The regression equation was established as Logit(P)=8.231 + 0.100 × lymphocyte percentage -5.344 × calcium ion. The ROC curve analysis showed that the model had an area under the ROC curve of 0.716, with a Youden index of 0.353, a sensitivity of 1.000, and a specificity of 0.353. The Hosmer-Lemeshow test showed that the model had poor calibration (χ2=20.688, P=0.008). The Bootstrap method with 1000 repeated samples showed that the estimated values of lymphocyte percentage (OR=1.106, 95%CI: 1.049 — 1.186, P=0.002) and calcium ion (OR=0.005, 95%CI: 0.000 — 0.214, P=0.010) were consistent with the original model, and the confidence intervals did not include 1, which further supported the reliability of the model. Conclusion Both lymphocyte percentage and blood calcium level are independent influencing factors for calcium salt deposition in alveolar echinococcosis, and the degree of calcium salt deposition in alveolar echinococcosis lesions increases with the reduction in blood calcium level and the increase in lymphocyte percentage. -
Key words:
- Echinococcosis, Hepatic /
- Calcium Deposition /
- Root Cause Analysis
-
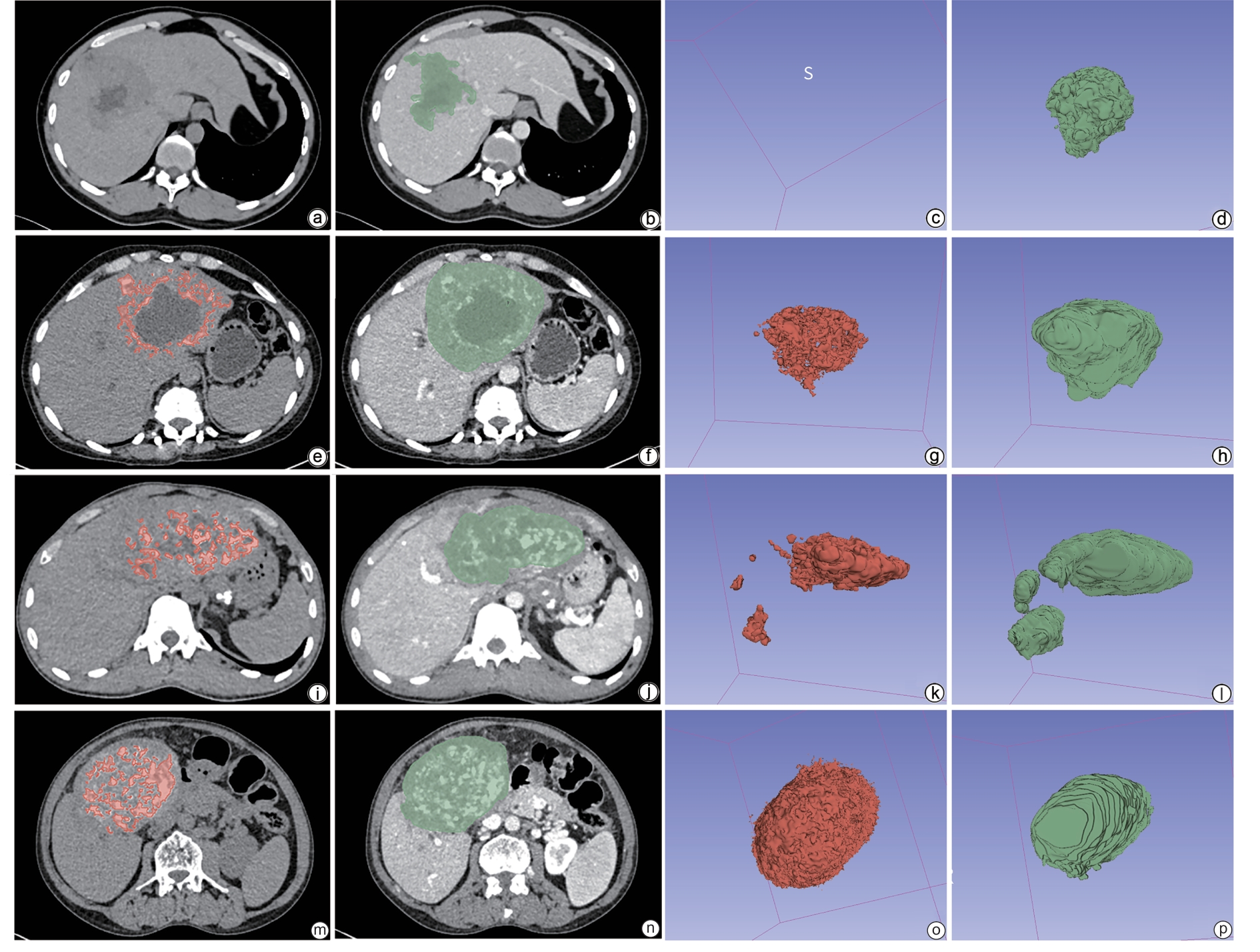
注: a~d,无钙盐沉积组;e~h,轻度钙盐沉积组;i~l,中度钙盐沉积组;m~p,重度钙盐沉积组。a、e、i、m,CT平扫钙盐沉积区(红色区域)分割图;b、f、j、n,增强CT静脉期病灶区(绿色区域)分割图;c、g、k、o,钙盐沉积体积的三维图;d、h、l、p,病灶体积的三维图。S,三维图中的方向south。CT,计算机体层成像。
图 1 各钙盐沉积组多房棘球蚴病患者CT影像及其钙盐沉积三维图
Figure 1. CT images of patients with echinococcosis multilocularis disease in each calcium deposition group and their three-dimensional images of calcium salt deposition
表 1 多房棘球蚴病患者基线资料对比
Table 1. Comparative analysis of baseline characteristics in patients with echinococcosis multilocularis
指标 无沉积组(n=16) 轻度沉积组(n=52) 中度沉积组(n=16) 重度沉积组(n=23) 统计值 P值 性别[例(%)] χ2=12.559 0.006 男 13(81.25) 22(42.31) 4(25.00) 14(60.87) 女 3(18.75) 30(57.69) 12(75.00) 9(39.13) 年龄(岁) 41.25 ± 3.47 41.00 ± 1.92 47.44 ± 2.80 38.78 ± 3.32 F=1.278 0.286 病程(年) 4.30(2.63~7.38) 3.00(1.50~5.88) 2.50(0.85~5.25) 2.00(1.00~3.70) H=5.080 0.166 累及其他部位[例(%)] 0(0.00) 16(30.77) 6(37.50) 6(26.09) 0.0101) 注:1)为Fisher’s精确检验。
表 2 各组多房棘球蚴病患者炎症与免疫相关指标比较
Table 2. Comparative analysis of inflammatory and immune-related parametersin patients with echinococcosis multilocularis in each group
指标 无沉积组(n=16) 轻度沉积组(n=52) 中度沉积组(n=16) 重度沉积组(n=23) 统计值 P值 白细胞计数
(×109/L)7.391 ± 0.349 7.080 ± 0.276 5.475 ± 0.333 5.905 ± 0.298 F=6.041 0.001 中性粒细胞
百分比(%)56.528
(49.425~66.325)58.800
(51.025~65.200)57.000
(47.300~60.175)56.300
(50.700~59.400)H=2.601 0.457 淋巴细胞百分比(%) 27.417 ± 2.262 26.240 ± 0.978 31.025 ± 2.108 31.188 ± 1.572 F=3.065 0.031 单核细胞百分比(%) 6.801 ± 0.280 7.455 ± 0.286 7.169 ± 0.396 7.872 ± 0.367 F=1.192 0.317 嗜酸性细胞
百分比(%)3.000
(2.075~8.525)4.450
(2.925~8.475)4.000
(2.350~7.875)4.300
(2.900~5.700)H=1.115 0.773 嗜碱性细胞
百分比(%)0.650
(0.40~0.871)0.800
(0.620~1.000)0.700
(0.500~1.175)0.800
(0.600~1.300)H=3.526 0.317 纤维蛋白原(g/L) 3.249 ± 0.320 3.811 ± 0.165 3.428 ± 0.220 3.056 ± 0.176 F=2.955 0.036 D-二聚体(mg/L) 0.365
(0.218~0.890)0.520
(0.167~0.845)0.760
(0.388~1.220)0.410
(0.110~0.955)H=3.556 0.314 表 3 各组多房棘球蚴病患者代谢与生化相关指标比较
Table 3. Comparative analysis of metabolic and biochemical parameters in patient with alveolar echinococcosis in each group
指标 无沉积组(n=16) 轻度沉积组(n=52) 中度沉积组(n=16) 重度沉积组(n=23) 统计值 P值 丙氨酸氨基转移酶
(U/L)44.509
(28.475~75.325)32.800
(19.000~50.750)21.250
(16.775~46.500)23.000
(15.000~48.730)H=6.668 0.083 天冬氨酸氨基转移酶
(U/L)32.750
(26.575~47.991)33.450
(21.000~45.025)27.650
(16.750~32.700)26.000
(20.000~47.700)H=3.888 0.274 碱性磷酸酶(U/L) 186.600
(92.125~300.326)196.000
(106.475~257.200)143.500
(112.450~220.860)107.000
(77.500~223.400)H=4.684 0.196 γ-谷氨酰转移酶
(U/L)146.101
(43.075~300.000)104.150
(43.200~154.107)65.950
(31.125~144.466)65.000
(36.000~150.000)H=3.872 0.276 总蛋白(g/L) 79.830 ± 2.457 78.594 ± 1.243 74.444 ± 2.094 75.780 ± 1.803 F=1.526 0.212 白蛋白(g/L) 40.141 ± 0.985 37.027 ± 0.632 36.775 ± 1.006 38.198 ± 0.905 F=2.436 0.069 球蛋白(g/L) 39.222 ± 2.734 41.170 ± 1.544 37.669 ± 2.379 37.253 ± 1.970 F=0.948 0.420 白蛋白/球蛋白 1.100
(0.825~1.475)0.900
(0.700~1.275)1.000
(0.725~1.275)1.000
(0.700~1.400)H=2.591 0.459 二氧化碳结合力
(mmol/L)21.700
(18.475~23.210)22.500
(21.000~24.675)21.300
(20.475~23.700)22.900
(20.579~24.300)H=3.426 0.330 尿素(mmol/L) 5.500
(4.100~6.615)5.100
(4.125~6.068)5.147
(4.140~5.408)5.094
(4.300~6.382)H=0.766 0.858 肌酐(μmol/L) 66.400
(58.382~74.200)62.400
(51.425~67.200)57.600
(48.775~69.500)65.300
(60.354~74.300)H=6.290 0.098 尿酸(μmol/L) 347.300
(296.663~440.400)289.073
(261.025~305.182)287.730
(235.850~320.289)301.681
(253.000~362.000)H=9.890 0.020 eGFR(CKD-EPI)
(mL/min)112.624
(109.731~124.325)110.185
(103.120~117.805)105.979
(98.955~115.333)106.077
(99.200~120.500)H=4.376 0.224 钾离子(mmol/L) 4.030
(3.800~4.345)4.100
(3.800~4.300)3.950
(3.390~4.146)4.060
(3.700~4.300)H=1.966 0.579 钠离子(mmol/L) 141.700
(139.125~142.975)139.900
(137.500~142.700)142.650
(140.650~143.750)139.900
(138.200~141.900)H=9.626 0.022 氯离子(mmol/L) 107.050
(105.700~109.375)106.150
(103.750~109.425)107.750
(106.550~111.175)106.200
(105.100~107.000)H=8.550 0.036 钙离子(mmol/L) 2.257 ± 0.027 2.197 ± 0.015 2.143 ± 0.023 2.174 ± 0.031 F=2.826 0.042 镁离子(mmol/L) 0.816 ± 0.031 0.810 ± 0.012 0.848 ± 0.014 0.822 ± 0.018 F=0.750 0.275 磷离子(mmol/L) 1.204 ± 0.052 1.164 ± 0.026 1.163 ± 0.036 1.206 ± 0.061 F=0.323 0.808 注:eGFR,估算的肾小球滤过率;CKD-EPI,慢性肾脏病流行病学合作研究。
表 4 影响多房棘球蚴病患者钙盐沉积的二元Logistic回归分析
Table 4. Binary Logistic regression analysis of calcium deposition in patients with echinococcosis multilocularis
因素 系数估计值 SE Wald χ2 P值 比值比(95%置信区间) 淋巴细胞百分比 0.100 0.031 10.593 0.001 1.106(1.041~1.174) 钙离子 -5.344 1.978 7.301 0.007 0.005(0.000~0.230) 常量 8.231 4.116 4.000 0.046 — 表 5 多房棘球蚴病患者钙盐沉积影响因素的Bootstrap验证(n=1 000)
Table 5. Factors affecting calcium deposition in patients with echinococcosis multilocularis cyst (Bootstrap validation, n=1 000)
变量 原始模型 Bootstrap验证 比值比(95%置信区间) P值 比值比(95%置信区间) P值 淋巴细胞百分比 1.106(1.041~1.174) 0.001 1.105(1.049~1.186) 0.002 钙离子 0.005(0.000~0.230) 0.007 0.005(0.000~0.214) 0.010 -
[1] ILICA AT, KOCAOGLU M, ZEYBEK N, et al. Extrahepatic abdominal hydatid disease caused by Echinococcus granulosus: Imaging findings[J]. AJR Am J Roentgenol, 2007, 189( 2): 337- 343. DOI: 10.2214/AJR.07.2255. [2] WEN H, VUITTON L, TUXUN T, et al. Echinococcosis: Advances in the 21st century[J]. Clin Microbiol Rev, 2019, 32( 2): e00075-18. DOI: 10.1128/CMR.00075-18. [3] CHEN KF, TANG YY, WANG R, et al. The choose of different surgical therapies of hepatic alveolar echinococcosis: A single-center retrospective case-control study[J]. Medicine, 2018, 97( 8): e0033. DOI: 10.1097/MD.0000000000010033. [4] MENG QY, PENG J, MA SM, et al. Progress of researches on the use of ultrasound in the diagnosis and treatment of hepatic alveolar echinococcosis[J]. Chin J Schisto Control, 2020, 32( 6): 657- 660. DOI: 10.16250/j.32.1374.2020055.孟庆杨, 彭婕, 马淑梅, 等. 超声在肝多房棘球蚴病诊疗中的应用进展[J]. 中国血吸虫病防治杂志, 2020, 32( 6): 657- 660. DOI: 10.16250/j.32.1374.2020055. [5] XU MQ, HADEER KEB, KONG CQ, et al. Imaging diagnosis and classification of hepatic cystic echinococcosis[J]. Natl Med J China, 2002, 82( 3): 176- 179.徐明谦, 哈德尔·库尔班, 孔长青, 等. 肝囊性包虫病的影像学诊断与分型[J]. 中华医学杂志, 2002, 82( 3): 176- 179. [6] MÜLLER J, ZUMKEHR B, HELLER M, et al. Host proteins in Echinococcus multilocularis metacestodes[J]. Int J Mol Sci, 2025, 26( 7): 3266. DOI: 10.3390/ijms26073266. [7] LIU HH, XIE YJ, AN XY, et al. Advances in novel diagnostic techniques for alveolar echinococcosis[J]. Diagnostics, 2025, 15( 5): 585. DOI: 10.3390/diagnostics15050585. [8] GRÜNER B, PETERS L, HILLENBRAND A, et al. Echinococcus multilocularis specific antibody, systemic cytokine, and chemokine levels, as well as antigen-specific cellular responses in patients with progressive, stable, and cured alveolar echinococcosis: A 10-year follow-up[J]. PLoS Negl Trop Dis, 2022, 16( 2): e0010099. DOI: 10.1371/journal.pntd.0010099. [9] Radiology of Infection Subbranch, Branch Radiology, Chinese Medical Association; Committee on Radiology of Infectious, Branch Radiology, Chinese Medical Doctor Association. Expert consensus on the imaging diagnosis of hepatic echinococcosis[J]. J Clin Hepatol, 2021, 37( 4): 792- 797. DOI: 10.3969/j.issn.1001-5256.2021.04.014.中华医学会放射学分会传染病学组, 中国医师协会放射医师分会感染影像专业委员会. 肝包虫病影像学诊断专家共识[J]. 临床肝胆病杂志, 2021, 37( 4): 792- 797. DOI: 10.3969/j.issn.1001-5256.2021.04.014. [10] GRAETER T, EBERHARDT N, SHI R, et al. Hepatic alveolar echinococcosis: Correlation between computed tomography morphology and inflammatory activity in positron emission tomography[J]. Sci Rep, 2020, 10( 1): 11808. DOI: 10.1038/s41598-020-68624-9. [11] OU ZH, LI L, REN PD, et al. Spatiotemporal transcriptomic profiling reveals the dynamic immunological landscape of alveolar echinococcosis[J]. Adv Sci, 2025, 12( 18): 2405914. DOI: 10.1002/advs.202405914. [12] HUANG H, ZHANG SK. Research progress on mechanisms of infiltration and metastasis for the alveolar echinococcosis[J]. Chin J Zoonoses, 2016, 32( 7): 670- 673, 678. DOI: 10.3969/j.issn.1002-2694.2016.07.016.黄红, 张淑坤. 多房棘球蚴病的浸润和转移机制研究进展[J]. 中国人兽共患病学报, 2016, 32( 7): 670- 673, 678. DOI: 10.3969/j.issn.1002-2694.2016.07.016. [13] MATUZ-MARES D, GONZÁLEZ-ANDRADE M, ARAIZA-VILLANUEVA MG, et al. Mitochondrial calcium: Effects of its imbalance in disease[J]. Antioxidants(Basel), 2022, 11( 5): 801. DOI: 10.3390/antiox11050801. [14] KASSIS AI, TANNER CE. Host serum proteins in Echinococcus multilocularis: Complement activation via the classical pathway[J]. Immunology, 1977, 33( 1): 1- 9. [15] JIANG TM, SUN W, AJI T, et al. Single-cell heterogeneity of the liver-infiltrating lymphocytes in individuals with chronic Echinococcus multilocularis infection[J]. Infect Immun, 2022, 90( 11): e00177-22. DOI: 10.1128/iai.00177-22. [16] DAWA ZM, ZHAI T, LIU CC, et al. Efficacy and safety of pseudolaric acid B against Echinococcus multilocularis in vitro and in a murine infection model[J]. Front Med, 2025, 12: 1503472. DOI: 10.3389/fmed.2025.1503472. [17] WANG JH, GOEPFERT C, MUELLER N, et al. Larval Echinococcus multilocularis infection reduces dextran sulphate sodium-induced colitis in mice by attenuating T helper type 1/type 17-mediated immune reactions[J]. Immunology, 2018, 154( 1): 76- 88. DOI: 10.1111/imm.12860. [18] TREBAK M, KINET JP. Calcium signalling in T cells[J]. Nat Rev Immunol, 2019, 19( 3): 154- 169. DOI: 10.1038/s41577-018-0110-7. [19] FONTA CM, ARNOLDINI S, JARAMILLO D, et al. Fibronectin fibers are highly tensed in healthy organs in contrast to tumors and virus-infected lymph nodes[J]. Matrix Biol Plus, 2020, 8: 100046. DOI: 10.1016/j.mbplus.2020.100046. [20] PALIT S, KENDRICK J. Vascular calcification in chronic kidney disease: Role of disordered mineral metabolism[J]. Curr Pharm Des, 2014, 20( 37): 5829- 5833. DOI: 10.2174/1381612820666140212194926. [21] LIANG SQ, HOU M. Analysis of the current situation of hypocalcemia in critically ill patients[J]. Adv Clin Med, 2023, 13( 12): 18763- 18767. DOI: 10.12677/ACM.2023.13122639.梁水清, 侯明. 危重患者低钙血症的现状分析[J]. 临床医学进展, 2023, 13( 12): 18763- 18767. DOI: 10.12677/ACM.2023.13122639. [22] PROUDFOOT D. Calcium signaling and tissue calcification[J]. Cold Spring Harb Perspect Biol, 2019, 11( 10): a035303. DOI: 10.1101/cshperspect.a035303. [23] RODRIGUES JJ, FERREIRA HB, FARIAS SE, et al. A protein with a novel calcium-binding domain associated with calcareous corpuscles in Echinococcus granulosus[J]. Biochem Biophys Res Commun, 1997, 237( 2): 451- 456. DOI: 10.1006/bbrc.1997.7025. -



 PDF下载 ( 14459 KB)
PDF下载 ( 14459 KB)

 下载:
下载: