内脏脂肪面积对非酒精性脂肪性肝病患者发生显著肝纤维化的影响及预测模型构建
DOI: 10.12449/JCH260211
Impact of visceral fat area on significant liver fibrosis in patients with nonalcoholic fatty liver disease and establishment of a predictive model
-
摘要:
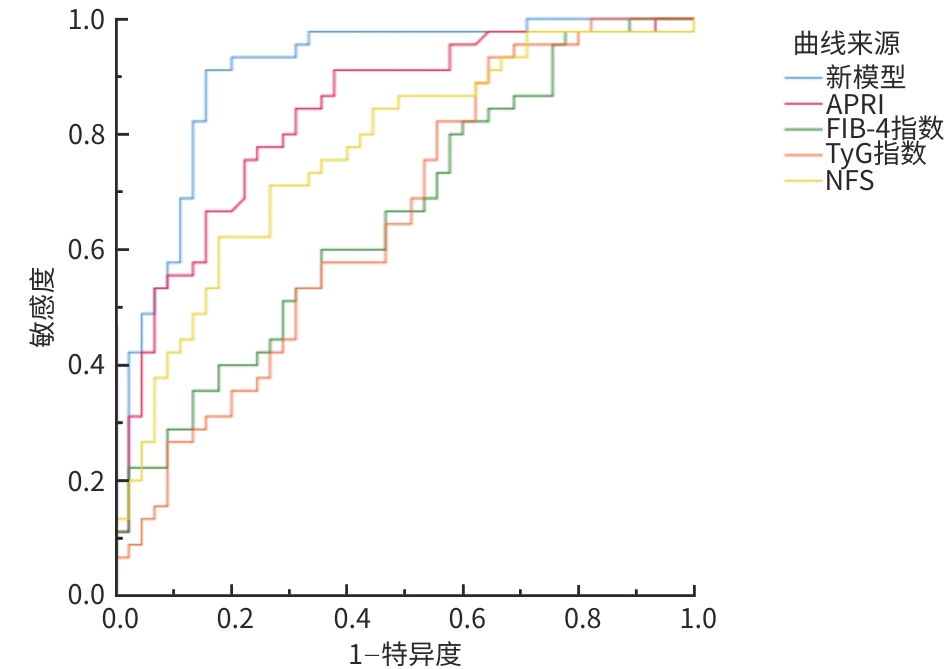
目的 本研究基于临床数据,探讨内脏脂肪面积(VFA)是否为非酒精性脂肪性肝病(NAFLD)患者发生显著肝纤维化的独立危险因素,并据此构建一个有效的诊断模型。 方法 纳入2021年1月—2025年4月于广东省中医院肝病科就诊的222例NAFLD患者,根据肝硬度值是否≥8 kPa,分为显著肝纤维化组和无显著肝纤维化组,并应用倾向性评分匹配(PSM)进行1∶1配对以平衡2组基线资料。计量资料两组比较采用成组t检验或Mann-Whitney U检验。计数资料组间比较采用χ2检验。通过Spearman相关性分析明确VFA及其他指标与显著肝纤维化的相关性;进一步采用单因素和多因素Logistic回归分析明确VFA是否是NAFLD患者发生显著肝纤维化的独立影响因素,并绘制受试者操作特征曲线(ROC曲线)评估相关指标的预测效能。 结果 共纳入显著肝纤维化45例,无显著肝纤维化177例,经PSM后最终纳入90例(45对)患者。与无显著肝纤维化组相比,显著肝纤维化组的体重指数(BMI)、空腹血糖(FBG)、糖化血红蛋白(HbA1c)、尿酸(UA)、丙氨酸氨基转移酶(ALT)、天冬氨酸氨基转移酶(AST)、γ-谷氨酰转移酶(GGT)、受控衰减指数(CAP)、VFA水平更高,内脏脂肪型肥胖、合并3种及以上的代谢危险因素比例更高(P值均<0.05)。VFA、BMI、AST、HbA1c与显著肝纤维化呈强相关(r值均>0.5,P值均<0.05),ALT、GGT、UA、FBG、CAP、内脏型肥胖亦与显著肝纤维化显著正相关(r=0.3~0.5,P值均<0.05)。VFA(OR=1.040,95%CI:1.018~1.062)、FBG(OR=2.372,95%CI:1.199~4.691)、AST(OR=1.032,95%CI:1.003~1.058)是NAFLD患者发生显著肝纤维化的独立危险因素(P值均<0.05)。由VFA、FBG、AST构建的新型诊断模型对NALFD显著肝纤维化的预测效能(AUC=0.907)优于天冬氨酸氨基转移酶/血小板比值指数(AUC=0.834)、纤维化-4指数(AUC=0.660)、甘油三酯-葡萄糖指数(AUC=0.656)、NAFLD纤维化评分(AUC=0.768),差异均具有统计学意义(P值均<0.05)。 结论 VFA是NAFLD患者发生显著肝纤维化的独立影响因素,基于VFA、FBG及AST构建的无创诊断模型能有效预测NAFLD患者发生显著肝纤维化。 Abstract:Objective To investigate whether visceral fat area (VFA) is an independent risk factor for significant liver fibrosis in patients with nonalcoholic fatty liver disease (NAFLD) based on clinical data, and to establish an effective diagnostic model. Methods A total of 222 NAFLD patients who attended Department of Hepatology, Guangdong Provincial Hospital of Traditional Chinese Medicine, from January 2021 to April 2025 were enrolled, and according to liver stiffness measurement (≥8 kPa or not), they were divided into significant fibrosis group and non-significant fibrosis group. Propensity score matching (PSM) was performed at a ratio of 1∶1 to balance the baseline data between the two groups. The independent-samples t test or the Mann-Whitney U test was used for comparison of continuous data between two groups; the chi-square test was used for comparison of categorical data between groups. A Spearman correlation analysis was used to determine the correlation of VFA and other indicators with significant liver fibrosis; univariate and multivariate logistic regression analyses were used to identify whether VFA was an independent risk factor for significant liver fibrosis in NAFLD patients, and the receiver operating characteristic (ROC) curve was plotted to assess the predictive performance of related indicators. Results A total of 45 patients with significant liver fibrosis and 177 patients without significant liver fibrosis were enrolled, and after PSM, 90 patients (45 pairs) were finally included in analysis. Compared with the non-significant fibrosis group, the significant fibrosis group had significantly higher levels of body mass index (BMI), fasting blood glucose (FBG), glycated hemoglobin (HbA1c), uric acid (UA), alanine aminotransferase (ALT), aspartate aminotransferase (AST), gamma-glutamyl transpeptidase (GGT), controlled attenuation parameter (CAP), and VFA, as well as a significantly higher proportion of patients with visceral fat obesity or three or more metabolic risk factors (all P<0.05). VFA, BMI, AST, and HbA1c were strongly correlated with significant liver fibrosis (all r>0.5, all P <0.05), and ALT, GGT, UA, FBG, and CAP were significantly positively correlated with significant liver fibrosis (r=0.3 — 0.5, all P<0.05). VFA (odds ratio [OR]=1.040, 95% confidence interval [CI]: 1.018 — 1.062, P<0.05), FBG (OR=2.372, 95%CI: 1.199 — 4.691, P<0.05), and AST (OR=1.032, 95%CI: 1.003 — 1.058, P<0.05) were independent risk factors for significant liver fibrosis in NAFLD patients. The new diagnostic model based on VFA, FBG, and AST (with an area under the ROC curve [AUC] of 0.907) had a significantly better performance than aspartate aminotransferase-to-platelet ratio index (AUC=0.834), fibrosis-4 (AUC=0.660), triglyceride-glucose index (AUC=0.656), and NAFLD fibrosis score (AUC=0.768) in predicting significant liver fibrosis in NAFLD patients (all P<0.05). Conclusion VFA is an independent risk factor for significant liver fibrosis in NAFLD patients, and the noninvasive diagnostic model based on VFA, FBG, and AST can effectively predict the onset of significant liver fibrosis in NAFLD patients. -
Key words:
- Non-alcoholic Fatty Liver Disease /
- Hepatic Fibrosis /
- Visceral Fat Area
-
注: ROC曲线,受试者操作特征曲线;APRI,天冬氨酸氨基转移酶/血小板比值指数;FIB-4指数,纤维化-4指数;TyG指数,甘油三酯-葡萄糖指数;NFS,非酒精性脂肪性肝病纤维化评分。
图 1 ROC曲线评估新模型、APRI、FIB-4指数、TyG指数、NFS对NAFLD患者显著肝纤维化的预测价值
Figure 1. ROC curves evaluating the predictive value of new prediction model, APRI, FIB-4, TyG index, and NFS for significant liver fibrosis in NAFLD patients
表 1 PSM前后NAFLD显著肝纤维化与无显著肝纤维化的基线及生化指标比较
Table 1. Comparison of baseline and biochemical indicators between NAFLD patients with and without significant liver fibrosis before and after PSM
指标 匹配前 匹配后 无显著肝纤维化组
(n=177)显著肝纤维化组
(n=45)统计值 P值 无显著肝纤维化组
(n=45)显著肝纤维化组
(n=45)统计值 P值 年龄(岁) 48.00(40.00~54.50) 38.00(29.00~52.50) Z=-3.115 0.002 39.00(31.50~49.50) 38.00(29.00~52.50) Z=-0.166 0.868 男[例(%)] 116(65.54) 33(73.33) χ2 =0.988 0.320 30(66.67) 33(73.33) χ2 =0.479 0.490 BMI(kg/m2) 25.47(23.64~27.61) 30.70(27.43~32.64) Z=-6.267 <0.001 24.67(22.43~26.88) 30.70(27.43~32.64) Z=-5.447 <0.001 TC(mmol/L) 5.14±1.02 5.47±1.15 t=-1.601 0.109 5.36±0.95 5.47±1.15 t=-0.456 0.650 TG(mmol/L) 1.57(1.13~2.52) 2.20(1.57~3.13) Z=-2.975 0.003 1.87(1.36~2.62) 2.2(1.57~3.13) Z=-1.477 0.140 HDL-C
(mmol/L)1.11(0.96~1.28) 1.06(0.88~1.26) Z=-1.597 0.110 1.09(0.98~1.30) 1.06(0.88~1.27) Z=-1.497 0.134 LDL-C
(mmol/L)3.23(2.73~3.84) 3.44(2.89~4.08) Z=-1.532 0.125 3.38±0.91 3.50±0.84 t=-0.673 0.503 FBG(mmol/L) 5.11(4.68~5.55) 5.64(5.05~7.36) Z=-4.064 <0.001 4.99(4.62~5.37) 5.64(5.05~7.36) Z=-3.866 <0.001 HbA1c(%) 5.70(5.40~6.10) 6.20(5.85~6.90) Z=-5.776 <0.001 5.60(5.40~6.00) 6.20(5.85~6.90) Z=-5.041 <0.001 UA(μmol/L) 396.56±100.19 482.16±113.60 t=-4.977 <0.001 407.31±103.91 482.16±113.60 t=-3.261 0.002 ALT(U/L) 26.00
(18.00~55.50)97.00
(46.50~113.00)Z=-5.900 <0.001 27.00
(20.00~65.00)97.00
(46.50~113.00)Z=-4.193 <0.001 AST(U/L) 22.00(17.50~31.00) 51.00(34.50~74.50) Z=-7.110 <0.001 23.00
(19.00~35.00)51.00
(34.50~74.50)Z=-5.022 <0.001 GGT(U/L) 42.00
(25.00~71.00)82.00
(54.50~131.50)Z=-4.679 <0.001 47.00
(24.00~82.50)82.00
(54.50~131.50)Z=-3.301 0.001 CAP(dB/m) 279.97±46.09 319.67±39.65 t=-5.298 <0.001 270.80±47.66 319.67±39.65 t=-5.287 <0.001 VFA(cm2) 91.00
(72.00~116.50)122.00
(105.50~163.00)Z=-5.391 <0.001 81.00
(59.00~112.50)122.00
(105.50~163.00)Z=-5.000 <0.001 内脏型肥胖
[例(%)]72(40.68) 36(80.00) χ2 =22.207 <0.001 17(37.78) 36(80.00) χ2 =16.568 <0.001 高血压[例
(%)]78(44.07) 21(46.67) χ2 =0.098 0.754 17(37.78) 21(46.67) χ2 =0.729 0.393 代谢风险因
素≥3[例(%)]88(49.72) 39(86.67) χ2 =20.009 <0.001 23(51.11) 39(86.67) χ2 =13.272 <0.001 注:PSM,倾向性评分匹配;NAFLD,非酒精性脂肪性肝病;BMI,体重指数;TC,总胆固醇;TG,甘油三酯;HDL-C,高密度脂蛋白胆固醇;LDL-C,低密度脂蛋白胆固醇;FBG,空腹血糖;HbA1c,糖化血红蛋白;UA,尿酸;ALT,丙氨酸氨基转移酶;AST,天冬氨酸氨基转移酶;GGT,γ-谷氨酰转移酶;CAP,受控衰减参数;VFA,内脏脂肪面积。
表 2 NAFLD显著肝纤维化与临床指标的相关性分析
Table 2. Correlation analysis between significant liver fibrosis in NAFLD and related clinical indicators
指标 r值 P值 VFA 0.530 <0.001 BMI 0.577 <0.001 TC 0.028 0.795 TG 0.157 0.141 HDL-C -0.159 0.135 LDL-C 0.053 0.622 FBG 0.410 <0.001 HbA1c 0.534 <0.001 UA 0.303 0.004 AST 0.532 0.005 ALT 0.444 0.023 GGT 0.350 0.001 CAP 0.491 <0.001 内脏型肥胖 0.429 <0.001 注:NAFLD,非酒精性脂肪性肝病;VFA,内脏脂肪面积;BMI,体重指数;TC,总胆固醇;TG,甘油三酯;HDL-C,高密度脂蛋白胆固醇;LDL-C,低密度脂蛋白胆固醇;FBG,空腹血糖;HbA1c,糖化血红蛋白;UA,尿酸;AST,天冬氨酸氨基转移酶;ALT,丙氨酸氨基转移酶;GGT,γ-谷氨酰转移酶;CAP,受控衰减参数。
表 3 VFA影响NAFLD患者显著肝纤维化的单因素、多因素Logistic回归分析
Table 3. Univariate and multivariate logistic regression analysis of the impact of VFA on significant liver fibrosis in NAFLD patients
指标 单因素分析 多因素分析 OR(95%CI) P值 OR(95%CI) P值 ALT 1.019(1.008~1.030) 0.001 AST 1.046(1.021~1.070) <0.001 1.032(1.003~1.058) 0.016 FBG 2.515(1.465~4.319) 0.001 2.372(1.199~4.691) 0.013 CAP 1.026(1.013~1.038) <0.001 BMI 1.349(1.174~1.550) <0.001 VFA 1.038(1.020~1.056) <0.001 1.040(1.018~1.062) 0.001 UA 1.006(1.002~1.011) 0.003 HbA1c 5.215(2.089~13.018) <0.001 代谢危险因素≥3 7.618(2.878~20.160) <0.001 注:VFA,内脏脂肪面积;NAFLD,非酒精性脂肪性肝病;ALT,丙氨酸氨基转移酶;AST,天冬氨酸氨基转移酶;FBG,空腹血糖;CAP,受控衰减指数;BMI,体重指数;UA,尿酸;HbA1c,糖化血红蛋白;OR,比值比;CI,置信区间。
表 4 各模型预测NAFLD患者显著肝纤维化的ROC曲线结果
Table 4. ROC curve results for significant liver fibrosis in NAFLD patients predicted by each model
指标 敏感度(%) 特异度(%) 截断值 AUC(95%CI) P值 新模型 91.1 84.4 -0.557 4 0.907(0.843~0.970) <0.001 APRI 77.8 75.6 0.330 2 0.834(0.750~0.918) <0.001 FIB-4指数 60.0 66.4 0.776 4 0.660(0.549~0.772) 0.009 TyG指数 93.3 35.6 7.052 4 0.656(0.543~0.768) 0.011 NFS 71.1 73.3 -2.491 0 0.768(0.671~0.866) <0.001 注:NAFLD,非酒精性脂肪性肝病;ROC曲线,受试者操作特征曲线;APRI,天冬氨酸氨基转移酶/血小板比值指数;FIB-4指数,纤维化-4指数;TyG指数,甘油三酯-葡萄糖指数;NFS,非酒精性脂肪性肝病纤维化评分;OR,比值比;CI,置信区间。
-
[1] Hepatology Branch of Chinese Medical Association. Guidelines for prevention and treatment of metabolic-related(nonalcoholic) fatty liver disease(2024 edition)[J]. J Pract Hepatol, 2024, 27( 4): 494- 510. DOI: 10.3760/cma.j.cn501113-20240327-00163.中华医学会肝病学分会. 代谢相关(非酒精性)脂肪性肝病防治指南(2024年版)[J]. 实用肝脏病杂志, 2024, 27( 4): 494- 510. DOI: 10.3760/cma.j.cn501113-20240327-00163. [2] ANGULO P, KLEINER DE, DAM-LARSEN S, et al. Liver fibrosis, but no other histologic features, is associated with long-term outcomes of patients with nonalcoholic fatty liver disease[J]. Gastroenterology, 2015, 149( 2): 389- 397. e 10. DOI: 10.1053/j.gastro.2015.04.043. [3] DULAI PS, SINGH S, PATEL J, et al. Increased risk of mortality by fibrosis stage in nonalcoholic fatty liver disease: Systematic review and meta-analysis[J]. Hepatology, 2017, 65( 5): 1557- 1565. DOI: 10.1002/hep.29085. [4] KIM D, CHUNG GE, KWAK MS, et al. Effect of longitudinal changes of body fat on the incidence and regression of nonalcoholic fatty liver disease[J]. Dig Liver Dis, 2018, 50( 4): 389- 395. DOI: 10.1016/j.dld.2017.12.014. [5] YU SJ, KIM W, KIM D, et al. Visceral obesity predicts significant fibrosis in patients with nonalcoholic fatty liver disease[J]. Medicine, 2015, 94( 48): e2159. DOI: 10.1097/MD.0000000000002159. [6] National Workshop on Fatty Liver and Alcoholic Liver Disease, Chinese Society of Hepatology, Chinese Medical Association, Fatty Liver Expert Committee, Chinese Medical Doctor Association. Guidelines of prevention and treatment for nonalcoholic fatty liver disease: A 2018 update[J]. J Clin Hepatol, 2018, 34( 5): 947- 957. DOI: 10.3969/j.issn.1001-5256.2018.05.007.中华医学会肝病学分会脂肪肝和酒精性肝病学组, 中国医师协会脂肪性肝病专家委员会. 非酒精性脂肪性肝病防治指南(2018年更新版)[J]. 临床肝胆病杂志, 2018, 34( 5): 947- 957. DOI: 10.3969/j.issn.1001-5256.2018.05.007. [7] Chinese Society of Health Management; Clinical Nutrition Branch of Chinese Nutrition Society; Medical Nutrition Industry Branch of the National Association of Health Industry and Enterprise Management, et al. Expert consensus on weight management process for overweight or obese people(2021)[J]. Chin J Health Manag, 2021, 15( 4): 317- 322. DOI: 10.3760/cma.j.cn115624-20210630-00368.中华医学会健康管理学分会, 中国营养学会临床营养分会, 全国卫生产业企业管理协会医学营养产业分会, 等. 超重或肥胖人群体重管理流程的专家共识(2021年)[J]. 中华健康管理学杂志, 2021, 15( 4): 317- 322. DOI: 10.3760/cma.j.cn115624-20210630-00368. [8] CHEN N, GENG N, LI J. Mechanisms and clinical practice advances in weight reduction for patients with metabolic associated fatty liver disease[J/CD]. Chin J Liver Dis: Electronic Edition, 2025, 17( 3): 10- 16. DOI: 10.3969/j.issn.1671-4695.2025.01.026.陈楠, 耿楠, 李婕. 代谢相关脂肪性肝病患者减重治疗机制与临床实践进展[J/CD]. 中国肝脏病杂志(电子版), 2025, 17( 3): 10- 16. DOI: 10.3969/j.issn.1671-4695.2025.01.026. [9] DESPRÉS JP, LEMIEUX I. Abdominal obesity and metabolic syndrome[J]. Nature, 2006, 444( 7121): 881- 887. DOI: 10.1038/nature05488. [10] EGUCHI Y, MIZUTA T, SUMIDA Y, et al. The pathological role of visceral fat accumulation in steatosis, inflammation, and progression of nonalcoholic fatty liver disease[J]. J Gastroenterol, 2011, 46( Suppl 1): 70- 78. DOI: 10.1007/s00535-010-0340-3. [11] VARGHESE J, DEVADAS K, JOSEPH RC, et al. Assessment of visceral fat volume and its correlation with the severity of hepatic fibrosis in patients with NAFLD[J]. J Assoc Physicians India, 2022, 70( 9): 11- 12. DOI: 10.5005/japi-11001-0097. [12] IDILMAN IS, LOW HM, GIDENER T, et al. Association between visceral adipose tissue and non-alcoholic steatohepatitis histology in patients with known or suspected non-alcoholic fatty liver disease[J]. J Clin Med, 2021, 10( 12): 2565. DOI: 10.3390/jcm10122565. [13] AUSTIN PC. Balance diagnostics for comparing the distribution of baseline covariates between treatment groups in propensity-score matched samples[J]. Stat Med, 2009, 28( 25): 3083- 3107. DOI: 10.1002/sim.3697. [14] ROSENBAUM PR, RUBIN DB. The central role of the propensity score in observational studies for causal effects[J]. Biometrika, 1983, 70( 1): 41- 55. DOI: 10.1093/biomet/70.1.41. [15] LONARDO A, NASCIMBENI F, MAURANTONIO M, et al. Nonalcoholic fatty liver disease: Evolving paradigms[J]. World J Gastroenterol, 2017, 23( 36): 6571- 6592. DOI: 10.3748/wjg.v23.i36.6571. [16] XUAN YY, WU DT, ZHANG Q, et al. Elevated ALT/AST ratio as a marker for NAFLD risk and severity: Insights from a cross-sectional analysis in the United States[J]. Front Endocrinol, 2024, 15: 1457598. DOI: 10.3389/fendo.2024.1457598. [17] ZHANG YX, WANG Y, YOU CL, et al. Analysis of related factors of abnormal liver function in patients with non-alcoholic fatty liver disease[J]. Chin J Med Offic, 2025, 53( 5): 522- 524, 528. DOI: 10.16680/j.1671-3826.2025.05.21.张月霞, 王宇, 尤丛蕾, 等. 非酒精性脂肪肝患者肝功能异常相关因素分析[J]. 临床军医杂志, 2025, 53( 5): 522- 524, 528. DOI: 10.16680/j.1671-3826.2025.05.21. [18] JI XQ, LIU A, SUN YB, et al. Correlation analysis of visceral fat area and liver function related indexes based on quantitative CT[J]. J Clin Radiol, 2025, 44( 6): 1107- 1111. DOI: 10.13437/j.cnki.jcr.2025.06.024.季晓琪, 刘澳, 孙永兵, 等. 基于定量CT对内脏脂肪面积与肝功能相关指标的相关性分析[J]. 临床放射学杂志, 2025, 44( 6): 1107- 1111. DOI: 10.13437/j.cnki.jcr.2025.06.024. [19] BENNETT NR, FERGUSON TS, BENNETT FI, et al. High-sensitivity C-reactive protein is related to central obesity and the number of metabolic syndrome components in Jamaican young adults[J]. Front Cardiovasc Med, 2014, 1: 12. DOI: 10.3389/fcvm.2014.00012. [20] PONCE-DE-LEON M, HANNEMANN A, LINSEISEN J, et al. Links between ectopic and abdominal fat and systemic inflammation: New insights from the SHIP-Trend study[J]. Dig Liver Dis, 2022, 54( 8): 1030- 1037. DOI: 10.1016/j.dld.2022.02.003. [21] LUO JQ, WANG Y, MAO JX, et al. Features, functions, and associated diseases of visceral and ectopic fat: A comprehensive review[J]. Obesity, 2025, 33( 5): 825- 838. DOI: 10.1002/oby.24239. [22] GUO L, TANG QQ. Research progress of the mechanism and therapy of non-alcoholic fatty liver disease[J]. Chin Bull Life Sci, 2018, 30( 11): 1165- 1172. DOI: 10.13376/j.cbls/2018141.郭亮, 汤其群. 非酒精性脂肪肝发病机制和治疗的研究进展[J]. 生命科学, 2018, 30( 11): 1165- 1172. DOI: 10.13376/j.cbls/2018141. [23] SI KY, HU Y, WANG ML, et al. Weight loss strategies, weight change, and type 2 diabetes in US health professionals: A cohort study[J]. PLoS Med, 2022, 19( 9): e1004094. DOI: 10.1371/journal.pmed.1004094. [24] SZTANEK F, TÓTH LI, PETŐ A, et al. New developments in pharmacological treatment of obesity and type 2 diabetes-beyond and within GLP-1 receptor agonists[J]. Biomedicines, 2024, 12( 6): 1320. DOI: 10.3390/biomedicines12061320. [25] YANG B, ZHANG R. Progress on the treatment of metabolic associated fatty liver disease[J/OL]. Chin J Liver Dis: Electronic Edition, 2024, 16( 4): 25- 30. DOI: 10.3969/j.issn.1674-7380.2024.04.004.杨彬, 张瑞. 代谢相关脂肪性肝病治疗进展[J/OL]. 中国肝脏病杂志(电子版), 2024, 16( 4): 25- 30. DOI: 10.3969/j.issn.1674-7380.2024.04.004. [26] DUAN YJ, FENG WH, SUN XT, et al. Cognitive improvement following bariatric surgery is associated with enhanced hippocampal activation and reduction in visceral and ectopic fat deposition[J]. Obes Surg, 2025, 35( 8): 2867- 2876. DOI: 10.1007/s11695-025-07964-9. -



 PDF下载 ( 1019 KB)
PDF下载 ( 1019 KB)

 下载:
下载:


