八宝丹胶囊治疗慢性乙型肝炎病毒感染合并胆囊息肉的效果及安全性分析
DOI: 10.12449/JCH260210
Efficacy and safety of Babaodan Capsule in patients with chronic hepatitis B virus infection comorbid with gallbladder polyps
-
摘要:
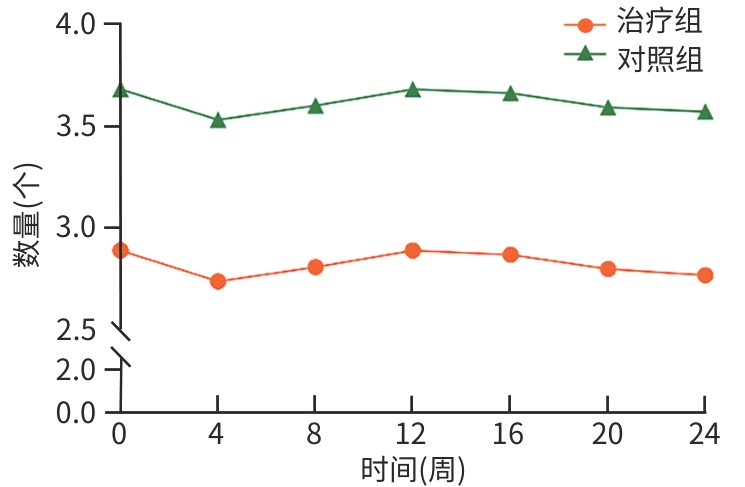
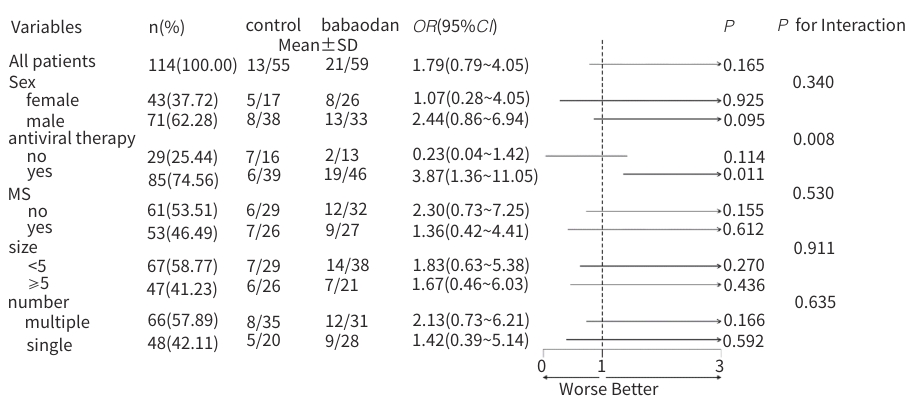
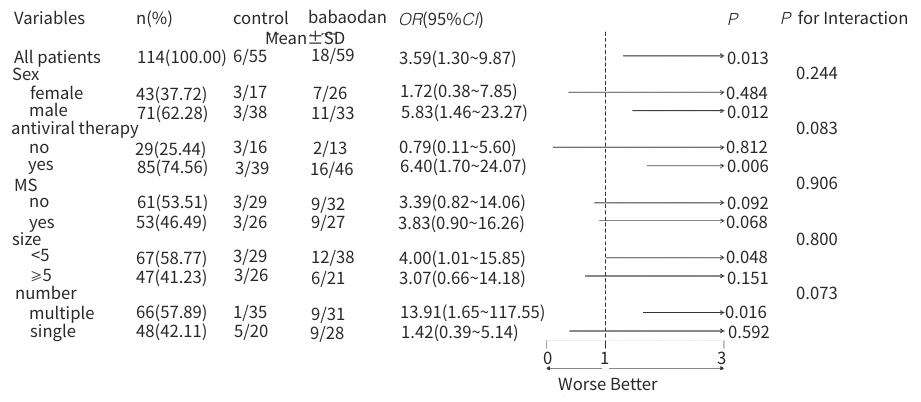
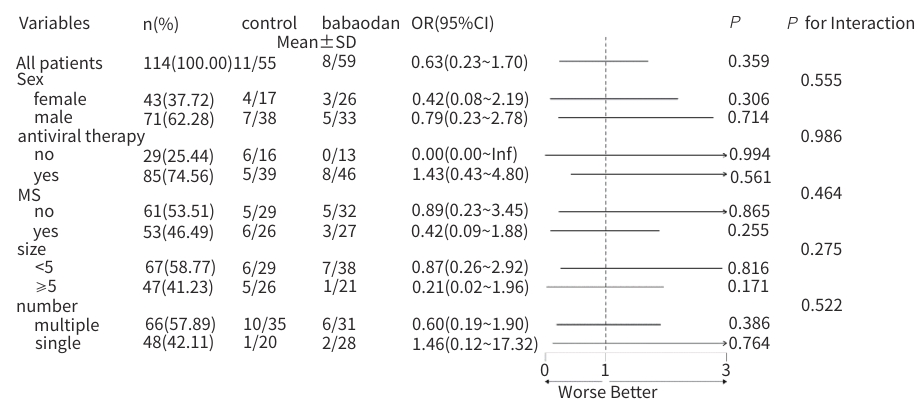
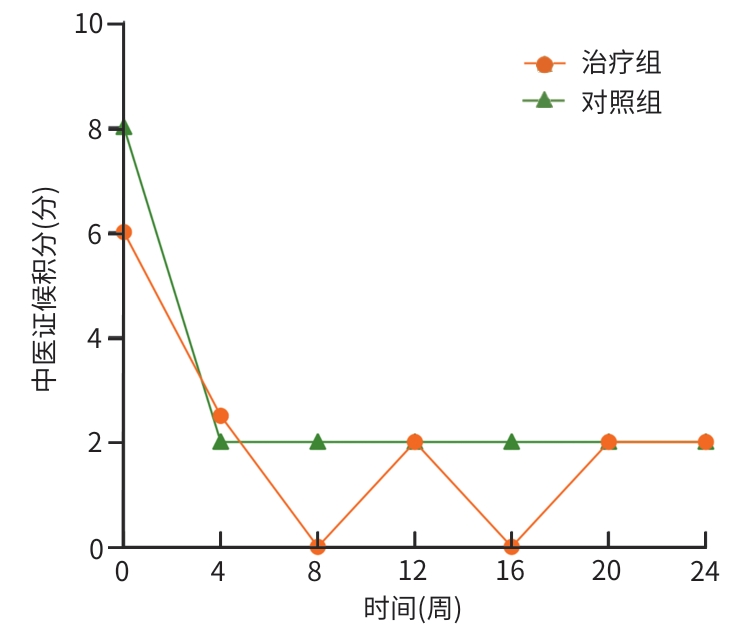
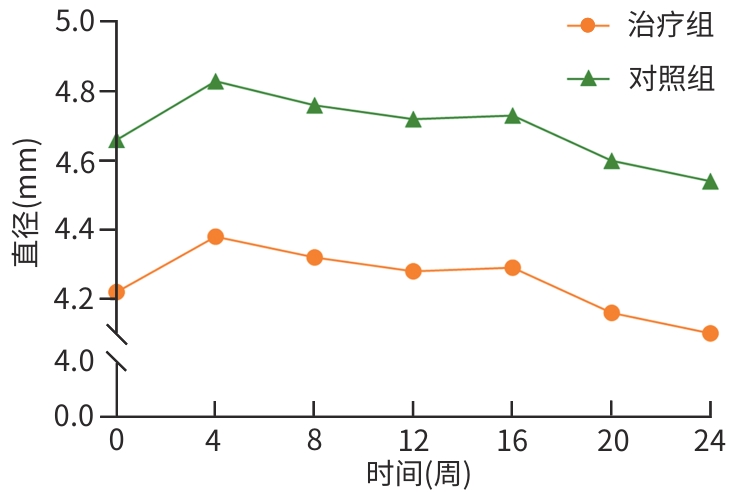
目的 评估八宝丹胶囊治疗肝胆湿热型慢性乙型肝炎病毒(HBV)感染合并胆囊息肉患者的效果及安全性。 方法 采用随机、双盲、安慰剂对照的单中心设计,纳入2020年8月—2023年4月首都医科大学附属北京佑安医院收治的慢性HBV感染者120例,分为治疗组(八宝丹胶囊)与对照组(安慰剂),各60例,疗程24周,每4周随访1次。主要疗效指标是胆囊息肉数量与最大直径(超声评估),次要指标是中医证候积分、血脂及肝功能指标等。计量资料2组间比较采用成组t检验或Wilcoxon秩和检验,计数资料2组间比较采用χ²检验或Fisher精确概率法;等级资料2组间比较采用Wilcoxon秩和检验;重复测量数据采用广义估计方程进行分析。 结果 治疗8周后,治疗组息肉直径及数量均显著低于对照组(Z值分别为-1.76、-1.80,P值均<0.05);治疗24周后,治疗组息肉缩小率高于对照组,差异有统计学意义(30.51% vs 10.91%,P<0.05)。亚组分析显示,联合抗病毒治疗者、男性、息肉<5 mm及多发息肉患者获益更为显著。治疗后第8周,治疗组中医证候积分优于对照组(Z=-2.35,P<0.05);治疗后,治疗组高密度脂蛋白胆固醇升高幅度更显著(Z=-1.85,P<0.05),丙氨酸氨基转移酶(Z=-2.06,P<0.05)、天冬氨酸氨基转移酶(Z=-2.13,P<0.05)、总胆红素(Z=-2.12,P<0.05)和直接胆红素(Z=-3.09,P<0.05)水平显著低于对照组。2组均未发生严重的不良事件。 结论 八宝丹胶囊可缩小肝胆湿热型慢性HBV感染患者的胆囊息肉,改善中医证候,降低胆红素水平,安全性良好,可能更适用于联合抗病毒治疗及特定亚群(男性、息肉<5 mm、多发息肉)患者。 Abstract:Objective To investigate the efficacy and safety of Babaodan Capsule (BBD) in the treatment of patients with chronic hepatitis B virus (HBV) infection with damp-heat in the liver and gallbladder comorbid with gallbladder polyps. Methods A randomized, double-blinded, placebo-controlled single-center trial was conducted among 120 patients with chronic HBV infection who were admitted to Beijing YouAn Hospital, Capital Medical University, from August 2020 to April 2023, and they were divided into treatment group (BBD) and control group (placebo), with 60 patients in each group. The course of treatment was 24 weeks, and follow-up assessments were conducted every 4 weeks. The primary outcome measures were the number and maximum diameter of gallbladder polyps (assessed by ultrasound), and the secondary outcome measures included traditional Chinese medicine (TCM) syndrome score, blood lipid levels, and liver function parameters. The independent-samples t test or the Wilcoxon rank-sum test was used for comparison of continuous data between two groups, and the chi-square test or the Fisher’s exact test was used for comparison of categorical data between two groups; the Wilcoxon rank-sum test was used for comparison of ranked data between two groups; the generalized estimating equation was used to analyze repeated measures data. Results After 8 weeks of treatment, the treatment group had a significantly smaller diameter of polyps and a significantly lower number of polyps than the control group (Z=-1.76 and -1.80, both P<0.05), and after 24 weeks of treatment, the treatment group had a significantly higher polyp reduction rate than the control group (30.51% vs 10.91%, P<0.05). The subgroup analysis showed that patients receiving combined antiviral therapy, male patients, patients with a diameter of polyps of <5 mm, and patients with multiple polyps tended to achieve significantly greater benefits. At week 8 of treatment, the treatment group had a significantly better TCM syndrome score than the control group (Z=-2.35, P<0.05); after treatment, compared with the control group, the treatment group had a significantly greater increase in high-density lipoprotein (Z=-1.85, P<0.05) and significantly lower levels of alanine aminotransferase (Z=-2.06, P <0.05), aspartate aminotransferase (Z=-2.13, P<0.05), total bilirubin (Z=-2.12, P<0.05), and direct bilirubin (Z=-3.09, P<0.05). No serious adverse events were reported in either group. Conclusion BBD can effectively reduce the size of gallbladder polyps, improve TCM syndrome score, and reduce the level of bilirubin in patients with chronic HBV infection with damp-heat in the liver and gallbladder, with a favorable safety profile, and it may be more suitable for patients receiving combined antiviral therapy and specific subgroups (male patients, patients with a diameter of polyps of <5 mm, and patients with multiple polyps. -
Key words:
- Hepatitis B Virus /
- Gallbladder Polyps /
- Babaodan Capsules /
- Therapeutics
-
表 1 基线资料对比
Table 1. Comparison of demographic data
指标 总计(n=114) 对照组(n=55) 治疗组(n=59) 统计值 P值 年龄(岁) 45.11±10.01 46.11±11.15 44.17±8.81 t=1.03 0.303 性别[例(%)] χ²=2.10 0.147 男 71(62.28) 38(69.09) 33(55.93) 女 43(37.72) 17(30.91) 26(44.07) 身高(cm) 169.03±8.12 170.22±8.64 167.92±7.50 t=1.52 0.131 体重(kg) 69.13±12.25 69.60±12.19 68.69±12.40 t=0.40 0.693 民族[例(%)] 0.361 汉族 111(97.37) 54(98.18) 57(96.61) 满族 2(1.75) 0(0.00) 2(3.39) 壮族 1(0.88) 1(1.82) 0(0.00) 婚姻状况[例(%)] χ²=0.00 >0.05 已婚 107(93.86) 52(94.55) 55(93.22) 未婚 7(6.14) 3(5.45) 4(6.78) 教育程度[例(%)] 0.087 初等教育 8(7.02) 5(9.09) 3(5.08) 中等教育 48(42.11) 28(50.91) 20(33.90) 高等教育 58(50.88) 22(40.00) 36(61.02) HBV感染史(年) 20.00(10.00~31.00) 20.00(13.75~33.25) 20.00(10.00~30.00) Z=-0.56 0.573 抗病毒治疗[例(%)] 85(74.56) 39 (70.91) 46(77.97) χ²=0.75 0.387 合并代谢综合征[例(%)] 53(46.49) 26(47.27) 27(45.76) χ²=0.03 0.872 肝功能指标 ALT(U/L) 22.00(15.50~31.00) 23.00(17.00~31.00) 20.00(14.25~31.00) Z=-0.98 0.325 AST(U/L) 24.00(20.00~27.00) 25.00(22.00~27.00) 24.00(19.00~27.75) Z=-1.52 0.128 TBil(μmol/L) 17.00(13.85~21.35) 17.20(14.00~22.50) 16.65(13.27~21.10) Z=-0.44 0.660 DBil(μmol/L) 5.10(4.10~6.55) 5.40(4.20~7.30) 4.85(4.10~6.07) Z=-1.17 0.243 GGT(U/L) 17.00(12.00~24.75) 18.00(12.00~25.00) 16.00(12.00~23.00) Z=-0.19 0.848 ALP(U/L) 76.00(64.50~88.00) 76.00(64.00~93.00) 76.00(65.25~83.75) Z=-0.35 0.730 血脂指标 TC(mmol/L) 0.98(0.71~1.27) 1.00(0.73~1.28) 0.96(0.71~1.24) Z=-0.19 0.849 TG(mmol/L) 4.60±0.80 4.60±0.75 4.60±0.86 t=0.01 0.990 HDL-C(mmol/L) 1.14(0.89~1.44) 1.08(0.89~1.48) 1.17(0.94~1.40) Z=-0.41 0.682 LDL-C(mmol/L) 2.86±0.72 2.81±0.64 2.90±0.79 t=-0.62 0.536 胆囊息肉特征 病程(年) 2.00(1.00~7.00) 3.00(1.00~8.00) 2.00(0.50~5.00) Z=-1.67 0.095 息肉直径(mm) 4.00(3.00~5.00) 4.00(4.00~5.50) 4.00(3.00~5.00) Z=-1.63 0.103 息肉大小[例(%)] χ²=1.60 0.206 直径<5 mm 67(58.77) 29(52.73) 38(64.41) 直径≥5 mm 47(41.23) 26(47.27) 21(35.59) 息肉数量(个) 2(1~5) 2(1~5) 2(1~4) Z=-1.53 0.127 息肉类别[例(%)] χ²=1.44 0.231 单发 48(42.11) 20(36.36) 28(47.46) 多发 66(57.89) 35(63.64) 31(52.54) 胆囊壁[例(%)] 毛糙 113(99.12) 55(100.00) 58(98.31) χ²=0.00 >0.05 增厚>3 mm 8(7.02) 3(5.45) 5(8.47) χ²=0.07 0.792 治疗前中医证候积分(分) 7.00(4.00~10.00) 8.00(5.00~10.50) 6.00(4.00~10.00) Z=-1.19 0.233 注:HBV,乙型肝炎病毒;ALT,丙氨酸氨基转移酶;AST,天冬氨酸氨基转移酶;TBil,总胆红素;DBil,直接胆红素;GGT,γ-谷氨酰转移酶;ALP,碱性磷酸酶;TC,总胆固醇;TG,甘油三酯;HDL-C,高密度脂蛋白胆固醇;LDL-C,低密度脂蛋白胆固醇。
表 2 治疗24周后总有效率对比
Table 2. Comparison of total effective rate after 24 weeks of treatment
指标 总计(n=114) 对照组(n=55) 治疗组(n=59) 统计值 P值 疗效[例(%)] Z=-1.44 0.086 无效 80(70.18) 42(76.36) 38(64.41) 有效 30(26.32) 12(21.82) 18(30.51) 治愈 4(3.51) 1(1.82) 3(5.08) 总有效率[例(%)] 34(29.82) 13(23.64) 21(35.59) χ²=1.94 0.117 注:总有效率=(治愈例数+有效例数)/总例数×100%。
表 3 治疗24周后息肉缩小率对比
Table 3. Comparison of polyp size reduction rate after 24 weeks of treatment
指标 总计(n=114) 对照组(n=55) 治疗组(n=59) 统计值 P值 疗效[例(%)] Z=-2.54 0.007 无效 90(78.95) 49(89.09) 41(69.49) 缩小 20(17.54) 5(9.09) 15(25.42) 消失 4(3.51) 1(1.82) 3(5.08) 缩小率[例(%)] 24(21.05) 6(10.91) 18(30.51) χ²=6.58 0.009 注:息肉缩小率=(消失例数+息肉缩小>25%例数)/总例数×100%。
表 4 治疗24周后息肉减少率对比
Table 4. Comparison of polyp number reduction rate after 24 weeks of treatment
指标 总计(n=114) 对照组(n=55) 治疗组(n=59) 统计值 P值 疗效[例(%)] Z=-0.81 0.249 无效 95(83.33) 44(80.00) 51(86.44) 减少 15(13.16) 10(18.18) 5(8.47) 消失 4(3.51) 1(1.82) 3(5.08) 减少率[例(%)] 19(16.67) 11(20.00) 8(13.56) χ²=0.85 0.251 注:息肉减少率=(消失例数+数量减少例数)/总例数×100%。
表 5 治疗24周后中医证候积分对比
Table 5. Comparison of TCM syndrome scores after 24 weeks of treatment
中医证候积分 总计(n=114) 对照组(n=55) 治疗组(n=59) Z值 P值 治疗前 7.00(4.00~10.00) 8.00(5.00~10.50) 6.00(4.00~10.00) -1.19 0.233 治疗后 2.00(0.00~4.00) 2.00(0.00~3.00) 2.00(0.00~4.75) -0.47 0.320 表 6 治疗24周后血脂指标变化对比
Table 6. Comparison of blood lipid after 24 weeks of treatment
指标 总计(n=114) 对照组(n=55) 治疗组(n=59) 统计值 P值 TG(mmol/L) 0.94(0.72~1.37) 1.06(0.75~1.45) 0.89(0.70~1.33) Z=-0.75 0.229 TC(mmol/L) 4.48(3.96~5.16) 4.42(3.99~5.14) 4.75(3.94~5.25) Z=-0.51 0.308 HDL-C(mmol/L) 1.20(0.96~1.45) 1.13(0.92~1.44) 1.27(0.99~1.45) Z=-0.89 0.189 LDL-C(mmol/L) 2.82±0.71 2.77±0.67 2.86±0.75 t=-0.62 0.269 注:TC,总胆固醇;TG,甘油三酯;HDL-C,高密度脂蛋白胆固醇;LDL-C,低密度脂蛋白胆固醇。
表 7 治疗24周后肝功能指标变化对比
Table 7. Comparison of liver function after 24 weeks of treatment
指标 总计(n=114) 对照组(n=55) 治疗组(n=59) 统计值 P值 ALT(U/L) 21.00(16.00~30.00) 24.00(16.00~35.00) 19.00(15.25~24.00) Z=-2.06 0.020 AST(U/L) 23.00(20.00~28.00) 23.00(21.00~30.50) 22.00(19.25~25.50) Z=-2.13 0.016 TBil(μmol/L) 16.90(13.60~21.80) 19.20(14.55~22.20) 15.80(12.83~18.80) Z=-2.12 0.017 DBil(μmol/L) 5.30(4.00~6.40) 5.90(4.70~7.60) 4.40(3.62~6.20) Z=-3.09 0.001 GGT(U/L) 18.00(13.00~25.00) 18.00(14.00~26.00) 17.50(12.00~23.75) Z=-0.34 0.367 ALP(U/L) 75.86±21.35 77.81±24.58 73.87±17.52 t=0.89 0.189 注:ALT,丙氨酸氨基转移酶;AST,天冬氨酸氨基转移酶;TBil,总胆红素;DBil,直接胆红素;GGT,γ-谷氨酰转移酶;ALP,碱性磷酸酶。
-
[1] FOLEY KG, LAHAYE MJ, THOENI RF, et al. Management and follow-up of gallbladder polyps: Updated joint guidelines between the ESGAR, EAES, EFISDS and ESGE[J]. Eur Radiol, 2022, 32( 5): 3358- 3368. DOI: 10.1007/s00330-021-08384-w. [2] ZHENG YM, BAI XS, YAO GB, et al. Risk factors of gallbladder polyps formation in East Asian population: A meta-analysis and systematic review[J]. Asian J Surg, 2020, 43( 1): 52- 59. DOI: 10.1016/j.asjsur.2019.03.015. [3] MAO YS, MAI YF, LI FJ, et al. Prevalence and risk factors of gallbladder polypoid lesions in Chinese petrochemical employees[J]. World J Gastroenterol, 2013, 19( 27): 4393- 4399. DOI: 10.3748/wjg.v19.i27.4393. [4] CHEN SP, WANG ZX, ZHANG XD, et al. Epidemiology of polypoid lesions of the gallbladder and related risk factors[J]. J Clin Hepatol, 2019, 35( 2): 441- 443. DOI: 10.3969/j.issn.1001-5256.2019.02.045.陈善鹏, 王智翔, 张小弟, 等. 胆囊息肉样病变的流行病学及危险因素[J]. 临床肝胆病杂志, 2019, 35( 2): 441- 443. DOI: 10.3969/j.issn.1001-5256.2019.02.045. [5] HUANG TY, ZHANG RH, LUO H. Study on the therapeutic effect of single-port and multi-port laparoscopic surgery on benign gallbladder polyps[J]. Trauma and Crit Medicine, 2024, 12( 4): 245- 247. DOI: 10.16048/j.issn.2095-5561.2024.04.11.黄太元, 张若涵, 罗恒. 单孔、多孔腹腔镜手术治疗良性胆囊息肉疗效研究[J]. 创伤与急危重病医学, 2024, 12( 4): 245- 247. DOI: 10.16048/j.issn.2095-5561.2024.04.11. [6] YANG HX, GOU J, HUANG P, et al. Babaodan Capsules for viral hepatitis: Systematic review of clinical efficacy and safety and Meta-analysis of randomized clinical trials[J]. China J Chin Mater Med, 2019, 44( 22): 4953- 4961. DOI: 10.19540/j.cnki.cjcmm.20190627.501.杨昊昕, 苟金, 黄坡, 等. 八宝丹胶囊治疗病毒性肝炎的疗效和安全性系统评价及Meta分析[J]. 中国中药杂志, 2019, 44( 22): 4953- 4961. DOI: 10.19540/j.cnki.cjcmm.20190627.501. [7] LIU PF. Clinical effect of Babaodan capsules in the treatment of jaundice type viral hepatitis[J]. Clin Res Pract, 2018, 3( 10): 138- 139. DOI: 10.19347/j.cnki.2096-1413.201810066.刘蒲芳. 八宝丹胶囊治疗黄疸型病毒性肝炎的临床效果[J]. 临床医学研究与实践, 2018, 3( 10): 138- 139. DOI: 10.19347/j.cnki.2096-1413.201810066. [8] CHEN DL, SHI JB. Study on therapeutic effect of Babaodan capsule on chronic viral hepatitis[J]. Strait Pharm J, 2016, 28( 7): 184- 185. DOI: 10.3969/j.issn.1006-3765.2016.07.099.陈冬玲, 施进宝. 八宝丹胶囊对慢性病毒性肝炎的治疗效果研究[J]. 海峡药学, 2016, 28( 7): 184- 185. DOI: 10.3969/j.issn.1006-3765.2016.07.099. [9] Hepatobiliary Disease Branch of Chinese Society of Traditional Chinese Medicine. Expert consensus on clinical application of Babaodan capsule in the treatment of viral hepatitis[J]. Chin J Integr Tradit West Med Liver Dis, 2020, 30( 6): 579- 580.中华中医药学会肝胆病学分会. 八宝丹胶囊治疗病毒性肝炎临床应用专家共识[J]. 中西医结合肝病杂志, 2020, 30( 6): 579- 580. [10] REN JB, SHEN YX, ZHANG YG, et al. Clinical trial of Babaodan capsules in the treatment of patients with acute cholecystitis[J]. Chin J Clin Pharmacol, 2023, 39( 12): 1689- 1693. DOI: 10.13699/j.cnki.1001-6821.2023.12.004.任军帮, 沈有秀, 张永刚, 等. 八宝丹胶囊治疗急性胆囊炎患者的临床研究[J]. 中国临床药理学杂志, 2023, 39( 12): 1689- 1693. DOI: 10.13699/j.cnki.1001-6821.2023.12.004. [11] Spleen and Stomach Disease Branch of Chinese Society of Traditional Chinese Medicine. Consensus opinion of TCM diagnosis and treatment experts on cholecystitis(2017)[J]. Chin J Integr Tradit West Med Dig, 2017, 25( 4): 241- 246. DOI: 10.3969/j.issn.1671-038X.2017.04.01.中华中医药学会脾胃病分会. 胆囊炎中医诊疗专家共识意见(2017)[J]. 中国中西医结合消化杂志, 2017, 25( 4): 241- 246. DOI: 10.3969/j.issn.1671-038X.2017.04.01. [12] Hepatobiliary Specialized Committee of China Association of Chinese Medicine, Liver Diseases Specialized Committee of China Medical Association of Minorities. The clinical guidelines of diagnosis and treatment of chronic hepatitis B with traditional Chinese medicine(2018)[J]. J Clin Hepatol, 2018, 34( 12): 2520- 2525. DOI: 10.3969/j.issn.1001-5256.2018.12.007.中华中医药学会肝胆病专业委员会, 中国民族医药学会肝病专业委员会. 慢性乙型肝炎中医诊疗指南(2018年版)[J]. 临床肝胆病杂志, 2018, 34( 12): 2520- 2525. DOI: 10.3969/j.issn.1001-5256.2018.12.007. [13] SHAO H. Clinical observation on 68 cases of polypoid lesions of gallbladder treated with self-made Huayu Lidan decoction[J]. Forum Tradit Chin Med, 2004, 19( 2): 33- 34. DOI: 10.3969/j.issn.1002-1078.2004.02.027.邵华. 自拟化瘀利胆汤治疗胆囊息肉样病变68例临床观察[J]. 国医论坛, 2004, 19( 2): 33- 34. DOI: 10.3969/j.issn.1002-1078.2004.02.027. [14] JIANG H, CHEN HQ. Clinical study on Xiaoyan Lidan Tang for gallbladder polyps[J]. J New Chin Med, 2019, 51( 6): 172- 174. DOI: 10.13457/j.cnki.jncm.2019.06.051.江浩, 陈华群. 消炎利胆汤治疗胆囊息肉临床研究[J]. 新中医, 2019, 51( 6): 172- 174. DOI: 10.13457/j.cnki.jncm.2019.06.051. [15] YUAN SQ. Study on the clinical efficacy of Shiwuwei Gandanshu Pill in the treatment of polypoid lesions of gallbladder(damp-heat of liver and gallbladder with blood stasis syndrome)[J]. Zhengzhou: Henan University of Chinese Medicine, 2018.袁帅强. 十五味肝胆舒丸治疗胆囊息肉样病变(肝胆湿热夹血瘀证)临床疗效研究[D]. 郑州: 河南中医药大学, 2018. [16] LI RJ, LI ZQ, DANG ZQ, et al. Self-made Lidan Hewei Granules combined with Danshi Tongli Tablets in the treatment of 40 cases of gallbladder polyps[J]. Chin J Ethnomed Ethnopharmacy, 2016, 25( 17): 97- 98, 100. DOI: 10.3969/j.issn.1007-8517.2016.17.zgmzmjyyzz201617035.李瑞娇, 李自强, 党中勤, 等. 自拟利胆和胃颗粒联合胆石通利片治疗胆囊息肉40例[J]. 中国民族民间医药, 2016, 25( 17): 97- 98, 100. DOI: 10.3969/j.issn.1007-8517.2016.17.zgmzmjyyzz201617035. [17] LI YF, SHENG HD, QIAN J, et al. The Chinese medicine babaodan suppresses LPS-induced sepsis by inhibiting NLRP3-mediated inflammasome activation[J]. J Ethnopharmacol, 2022, 292: 115205. DOI: 10.1016/j.jep.2022.115205. [18] QIAN J, XU HD, LV DQ, et al. Babaodan controls excessive immune responses and may represent a cytokine-targeted agent suitable for COVID-19 treatment[J]. Biomed Pharmacother, 2021, 139: 111586. DOI: 10.1016/j.biopha.2021.111586. [19] LI GD, LI XP, FENG CY, et al. Comparative study on the effects of in vitro cultured bezoar and natural bezoar on the metabolic profiles of serum and tissue bile acids in mice[J]. Lishizhen Med Mater Med Res, 2021, 32( 6): 1328- 1331. DOI: 10.3969/j.issn.1008-0805.2021.06.13.李国栋, 李喜平, 冯承阳, 等. 体外培育牛黄和天然牛黄对小鼠血清和组织胆汁酸代谢轮廓影响的对比研究[J]. 时珍国医国药, 2021, 32( 6): 1328- 1331. DOI: 10.3969/j.issn.1008-0805.2021.06.13. [20] SONG J, LI J, GUO SJ, et al. An initial observation about the therapeutic effect of Danweishu& yunjingyi treatment on gallbladder polyps[J]. Chin Arch Tradit Chin Med, 2010, 28( 1): 168- 170. DOI: 10.13193/j.archtcm.2010.01.170.songj.021.宋杰, 李健, 郭绍举, 等. 胆胃舒联合运经仪治疗胆囊息肉的初步观察[J]. 中华中医药学刊, 2010, 28( 1): 168- 170. DOI: 10.13193/j.archtcm.2010.01.170.songj.021. [21] SHENG DD, ZHAO SM, GAO L, et al. BabaoDan attenuates high-fat diet-induced non-alcoholic fatty liver disease via activation of AMPK signaling[J]. Cell Biosci, 2019, 9: 77. DOI: 10.1186/s13578-019-0339-2. -



 PDF下载 ( 1255 KB)
PDF下载 ( 1255 KB)

 下载:
下载: