丁型肝炎病毒感染病原学检测技术的临床应用及研究进展
DOI: 10.12449/JCH260203
Etiological detection techniques for hepatitis D virus infection: Clinical application and research advances
-
摘要: 丁型肝炎是由丁型肝炎病毒(HDV)感染引起的严重感染性疾病,其临床表现与转归因感染方式(同时感染和重叠感染)的不同存在差异。本文系统阐述了HDV病原学标志物、HDV感染的筛查策略、临床诊断及治疗管理原则;同时,从病毒结构的特点、基因型及检测技术等角度,探讨了HDV感染病原学检测所面临的挑战,并对该领域的新技术进行了综述,以期为HDV感染者的临床诊疗实践提供参考,并为推动病原学检测技术的标准化与国产化研发提供思路。Abstract: Hepatitis D is a severe infectious disease caused by hepatitis D virus (HDV), and its clinical manifestation and outcome vary depending on the mode of infection (co-infection and super-infection). This article systematically elaborates on the etiological markers for HDV, screening strategies for HDV infection, clinical diagnosis, and principles for treatment and management. In addition, it also discusses the challenges in etiological detection of HDV infection from the perspectives of the unique structure of the virus, genotypes, and detection techniques and reviews the new techniques in this field, in order to provide a reference for the clinical diagnosis and treatment of patients with HDV and offer new ideas for the standardization and domestication of etiological detection techniques.
-
Key words:
- Hepatitis Delta Virus /
- Etiological /
- Detection Technologies
-
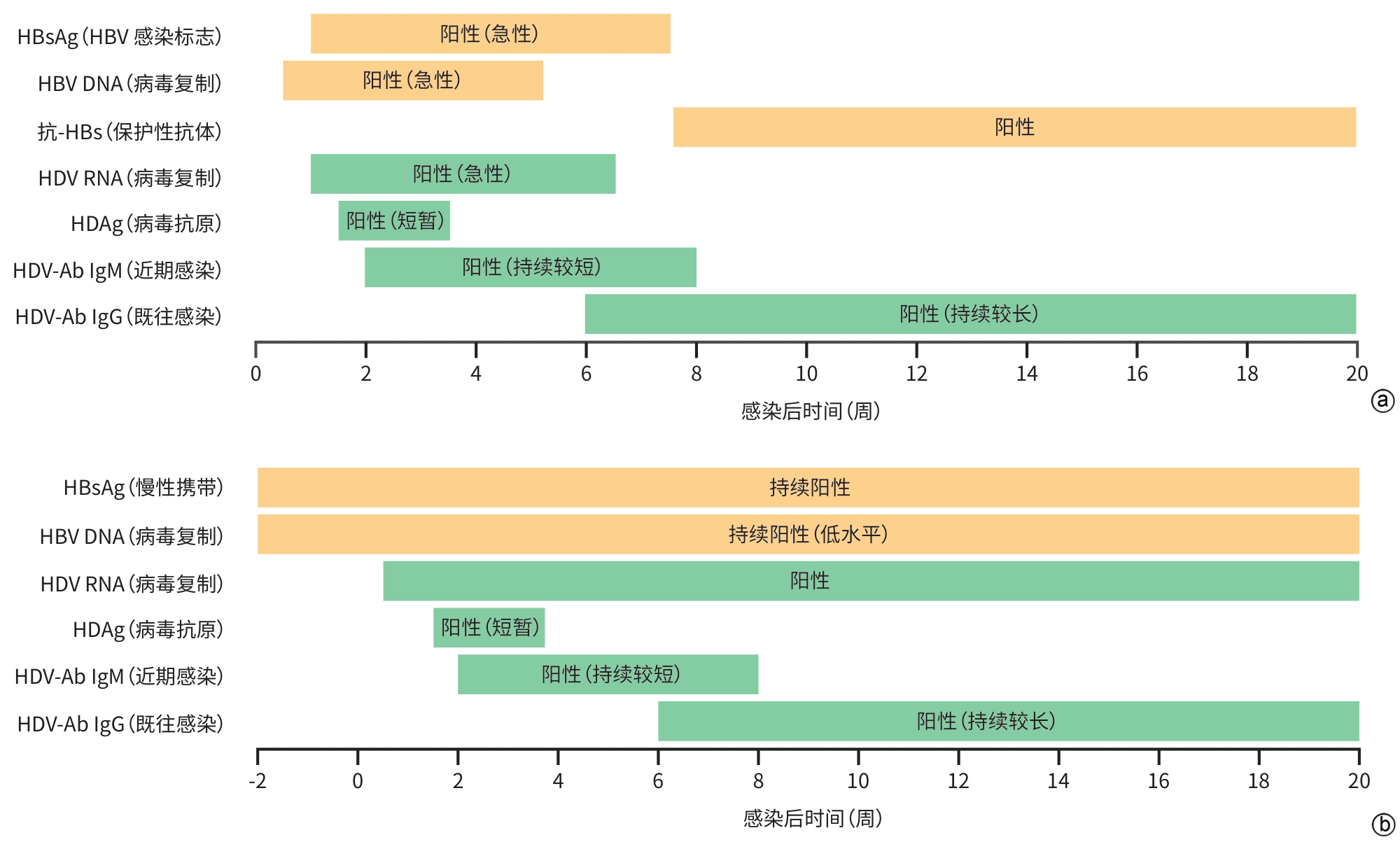
注: a,HDV/HBV同时感染(co-infection)通常表现为急性肝炎。血清中HDAg可能出现较早,但持续时间短且难以检测;HDV-Ab出现时间较晚;在无法进行HDV RNA检测时,HDV-Ab可能是HDV急性感染的唯一标志物,需通过后续检测记录血清中抗体转换情况。b,HDV的重叠感染(super-infection)可能表现为慢性HBV感染急性恶化。HBsAg阳性且HDV RNA及HDV-Ab检测阳性,可判断为HDV重叠感染,通常伴随HBsAg及HBV DNA水平下降,但HBV和HDV感染持续存在,因此,患者体内仍保持HBsAg、HBV DNA、HDV-Ab以及HDV RNA持续阳性状态[ 8]。HBsAg,乙型肝炎表面抗原;HBV,乙型肝炎病毒;抗-HBs,乙型肝炎表面抗体;HDV,丁型肝炎病毒;HDAg,丁型肝炎病毒抗原;HDV-Ab,丁型肝炎病毒抗体;IgM,免疫球蛋白M;IgG,免疫球蛋白G。
图 1 HDV感染病原学标志物的临床变化
Figure 1. Clinical changes of etiological markers in HDV infection
-
[1] DA BL, HELLER T, KOH C. Hepatitis D infection: From initial discovery to current investigational therapies[J]. Gastroenterol Rep, 2019, 7( 4): 231- 245. DOI: 10.1093/gastro/goz023. [2] LIU HM, FAN Y, CHEN WT, et al. Hepatitis D virus and HDV-like virus[J]. Chin J Exp Clin Virol, 2023, 37( 5): 564- 570. DOI: 10.3760/cma.j.cn112866-20230404-00035.刘慧敏, 范熠, 陈文婷, 等. 丁型肝炎病毒与HDV样病毒[J]. 中华实验和临床病毒学杂志, 2023, 37( 5): 564- 570. DOI: 10.3760/cma.j.cn112866-20230404-00035. [3] LI QF, ZHENG H, WU CQ, et al. Application of synthetic 27 oligopeptide of HDV antigen for detecting serum anti-HD in HBV infected subjects in Chongqing[J]. Chin J Intern Med, 1994, 33( 4): 237- 240.李奇芬, 郑红, 吴纯清, 等. 应用丁型肝炎病毒27肽抗原检测丁型肝炎病毒抗体[J]. 中华内科杂志, 1994, 33( 4): 237- 240. [4] LU XB, WANG XZ. Treatment and challenges of bulevirtide in chronic hepatitis D[J]. Chin J Hepatol, 2022, 30( 10): 1026- 1028. DOI: 10.3760/cma.j.cn501113-20221019-00502.鲁晓擘, 王晓忠. Bulevirtide治疗慢性丁型肝炎研究现状和面临的挑战[J]. 中华肝脏病杂志, 2022, 30( 10): 1026- 1028. DOI: 10.3760/cma.j.cn501113-20221019-00502. [5] ROMEO R, FOGLIENI B, CASAZZA G, et al. High serum levels of HDV RNA are predictors of cirrhosis and liver cancer in patients with chronic hepatitis delta[J]. PLoS One, 2014, 9( 3): e92062. DOI: 10.1371/journal.pone.0092062. [6] KAMAL H, WESTMAN G, FALCONER K, et al. Long-term study of hepatitis delta virus infection at secondary care centers: The impact of viremia on liver-related outcomes[J]. Hepatology, 2020, 72( 4): 1177- 1190. DOI: 10.1002/hep.31214. [7] ROULOT D, BRICHLER S, LAYESE R, et al. Origin, HDV genotype and persistent viremia determine outcome and treatment response in patients with chronic hepatitis delta[J]. J Hepatol, 2020, 73( 5): 1046- 1062. DOI: 10.1016/j.jhep.2020.06.038. [8] NEGRO F, LOK AS. Hepatitis D: A review[J]. Jama, 2023, 330( 24): 2376. DOI: 10.1001/jama.2023.23242. [9] World Health Organization. Guidelines for the prevention, diagnosis, care and treatment for people with chronic hepatitis B infection[EB/OL].( 2024-03-29)[ 2025-12-01]. https://www.who.int/publications/i/item/9789240090903. https://www.who.int/publications/i/item/9789240090903 [10] LEFKOWITCH JH. Scheuer’s liver biopsy interpretation(9th Edition.)[M]. YUAN N, DONG L, SUN MY, et al. Translation. Beijing: People’s Medical Publishing House, 2016: 92.杰伊·勒夫科维奇. 朔伊尔肝脏活检病理解读(第9版)[M]. 袁农, 董蕾, 孙明瑜, 等译. 北京: 人民卫生出版社, 2016: 92. [11] HAO LJ. Hepatitis D virus infection[J]. Jiangsu Med J, 1989, 15( 5): 265- 266.郝连杰. 丁型肝炎病毒感染[J]. 江苏医药, 1989, 15( 5): 265- 266. [12] ZHENG RJ, TANGNUER TLF, DENG ZR, et al. Sero-epidemiological characteristics of the hepatitis D virus infection among hepatitis B virus infected-patients at a single center in Xinjiang region[J]. Chin J Hepatol, 2022, 30( 10): 1044- 1049. DOI: 10.3760/cma.j.cn501113-20220406-00169.郑嵘炅, 唐努尔·塔拉甫, 邓泽润, 等. 新疆地区单中心乙型肝炎病毒感染者中丁型肝炎病毒感染的血清流行病学特点[J]. 中华肝脏病杂志, 2022, 30( 10): 1044- 1049. DOI: 10.3760/cma.j.cn501113-20220406-00169. [13] LIU BY, RAO HY. Research progress on the detection method of hepatitis D[J]. Chin J Hepatol, 2022, 30( 10): 1022- 1025. DOI: 10.3760/cma.j.cn501113-20221019-00504.刘百怡, 饶慧瑛. 丁型肝炎检测方法研究进展[J]. 中华肝脏病杂志, 2022, 30( 10): 1022- 1025. DOI: 10.3760/cma.j.cn501113-20221019-00504. [14] MAO Q, WANG SQ, LI QF, et al. Antiviral effect of an antisense phosphorothioate oligodeoxynucleotides complementary to the nucleotides seqience of genomic HDV ribozyme[J]. Immunol J, 1996, 12( 1): 5- 8.毛青, 王升启, 李奇芬, 等. 反义寡脱氧核苷酸抗丁型肝炎病毒的作用[J]. 免疫学杂志, 1996, 12( 1): 5- 8. [15] ROCCO C, BONAVOLTA R, VALLEFUOCO L, et al. Comparison of anti-hepatitis D virus(HDV) ETI-AB-DELTAK-2 assay and the novel LIAISON® XL MUREX anti-HDV assay in the diagnosis of HDV infection[J]. Diagn Microbiol Infect Dis, 2019, 95( 4): 114873. DOI: 10.1016/j.diagmicrobio.2019.114873. [16] GAO Y, REN F, DUAN ZP. Current research status of hepatitis D virus RNA detection[J]. J Clin Hepatol, 2023, 39( 4): 758- 765. DOI: 10.3969/j.issn.1001-5256.2023.04.004.高耀, 任锋, 段钟平. HDV RNA检测的研究现状[J]. 临床肝胆病杂志, 2023, 39( 4): 758- 765. DOI: 10.3969/j.issn.1001-5256.2023.04.004. [17] MAO Q, WANG SQ, LI QF, et al. Detection of hepatitis D virus nucleic acid in serum by polymerase chain reaction[J]. Chin J Lab Med, 1996, 19( 2): 83- 85.毛青, 王升启, 李奇芬, 等. 聚合酶链反应检测血清丁型肝炎病毒核酸[J]. 中华医学检验杂志, 1996, 19( 2): 83- 85. [18] KARATAYLı E, ALTUNOĞLU YÇ, KARATAYLı SC, et al. A one step real time PCR method for the quantification of hepatitis delta virus RNA using an external armored RNA standard and intrinsic internal control[J]. J Clin Virol, 2014, 60( 1): 11- 15. DOI: 10.1016/j.jcv.2014.01.021. [19] WANG C, SHEN X, LU J, et al. Development of a reverse transcription-loop-mediated isothermal amplification(RT-LAMP) system for rapid detection of HDV genotype 1[J]. Lett Appl Microbiol, 2013, 56( 3): 229- 235. DOI: 10.1111/lam.12039. [20] XU L, ZHANG XY, CAO YL, et al. Digital droplet PCR for detection and quantitation of hepatitis delta virus[J]. Clin Transl Gastroenterol, 2022, 13( 7): e00509. DOI: 10.14309/ctg.0000000000000509. [21] TERRAULT NA, LOK ASF, MCMAHON BJ, et al. Update on prevention, diagnosis, and treatment of chronic hepatitis B: AASLD 2018 hepatitis B guidance[J]. Hepatology, 2018, 67( 4): 1560- 1599. DOI: 10.1002/hep.29800. [22] PALOM A, RANDO-SEGURA A, VICO J, et al. Implementation of anti-HDV reflex testing among HBsAg-positive individuals increases testing for hepatitis D[J]. JHEP Rep, 2022, 4( 10): 100547. DOI: 10.1016/j.jhepr.2022.100547. [23] LIU HM, CHEN WT, LI SL, et al. Key points of EASL Clinical Practice Guidelines on hepatitis delta virus(2023)[J]. J Clin Hepatol, 2023, 39( 11): 2558- 2563. DOI: 10.3969/j.issn.1001-5256.2023.11.007.刘慧敏, 陈文婷, 李世炼, 等.《2023年欧洲肝病学会临床实践指南: 丁型肝炎病毒》意见要点[J]. 临床肝胆病杂志, 2023, 39( 11): 2558- 2563. DOI: 10.3969/j.issn.1001-5256.2023.11.007. [24] GHANY MG, PAN CQ, LOK AS, et al. AASLD/IDSA practice guideline on treatment of chronic hepatitis B[J]. Hepatology, 2025. DOI: 10.1097/HEP.0000000000001549. [25] ROGGENBACH I, CHI XM, LEMPP FA, et al. HDV seroprevalence in HBsAg-positive patients in China occurs in hotspots and is not associated with HCV mono-infection[J]. Viruses, 2021, 13( 9): 1799. DOI: 10.3390/v13091799. [26] LIU YM, GUO XP, ZHANG HM, et al. An investigation of hepatitis D virus infection among patients with chronic hepatitis B virus infection in some regions of China[J]. J Clin Hepatol, 2023, 39( 4): 795- 803. DOI: 10.3969/j.issn.1001-5256.2023.04.009.刘玉梅, 郭小平, 张慧敏, 等. 我国部分地区慢性HBV感染者HDV感染情况调查[J]. 临床肝胆病杂志, 2023, 39( 4): 795- 803. DOI: 10.3969/j.issn.1001-5256.2023.04.009. [27] LIU HM, TAN ZX, YANG J, et al. Prevalence and clinical characteristics of hepatitis D in Southwest of China[J]. Chin J Exp Clin Virol, 2022, 36( 1): 85- 91. DOI: 10.3760/cma.j.cn112866-20210611-00096.刘慧敏, 谭朝霞, 杨京, 等. 中国西南地区丁型肝炎的流行状况与临床特征分析[J]. 中华实验和临床病毒学杂志, 2022, 36( 1): 85- 91. DOI: 10.3760/cma.j.cn112866-20210611-00096. [28] ZHUANG H. Neglected hepatitis D[J]. Chin J Lab Med, 2024, 47( 3): 205- 209. DOI: 10.3760/cma.j.cn114452-20240122-00040.庄辉. 被忽视的丁型肝炎[J]. 中华检验医学杂志, 2024, 47( 3): 205- 209. DOI: 10.3760/cma.j.cn114452-20240122-00040. [29] CAVIGLIA GP, CIANCIO A, RIZZETTO M. A review of HDV infection[J]. Viruses, 2022, 14( 8): 1749. DOI: 10.3390/v14081749. [30] ZHANG YY, HAO LJ. Recent research on experimental infection of hepatitis D virus and animal model[J]. Int J Epidemiol Infect Dis, 1989, 16( 2): 71- 73.张永源, 郝连杰. 丁型肝炎病毒实验感染与动物模型研究近况[J]. 国外医学(流行病学传染病学分册), 1989, 16( 2): 71- 73. [31] XUE F, WEI L. Summary for hepatitis D research reports from EASL 2022[J]. Chin J Hepatol, 2022, 30( 10): 1120- 1123. DOI: 10.3760/cma.j.cn501113-20221019-00500.薛峰, 魏来. 2022年欧洲肝病学会年会关于慢性丁型肝炎研究的进展[J]. 中华肝脏病杂志, 2022, 30( 10): 1120- 1123. DOI: 10.3760/cma.j.cn501113-20221019-00500. [32] LUCIFORA J, ALFAIATE D, PONS C, et al. Hepatitis D virus interferes with hepatitis B virus RNA production via interferon-dependent and-independent mechanisms[J]. J Hepatol, 2023, 78( 5): 958- 970. DOI: 10.1016/j.jhep.2023.01.005. [33] HOMS M, RODRIGUEZ-FRIAS F, GREGORI J, et al. Evidence of an exponential decay pattern of the hepatitis delta virus evolution rate and fluctuations in quasispecies complexity in long-term studies of chronic delta infection[J]. PLoS One, 2016, 11( 6): e0158557. DOI: 10.1371/journal.pone.0158557. [34] LE GAL F, GAULT E, RIPAULT MP, et al. Eighth major clade for hepatitis delta virus[J]. Emerg Infect Dis, 2006, 12( 7): 1447- 1450. DOI: 10.3201/eid1209.060112. [35] CASEY JL, BROWN TL, COLAN EJ, et al. A genotype of hepatitis D virus that occurs in northern South America[J]. Proc Natl Acad Sci U S A, 1993, 90( 19): 9016- 9020. DOI: 10.1073/pnas.90.19.9016. [36] IVANIUSHINA V, RADJEF N, ALEXEEVA M, et al. Hepatitis delta virus genotypes I and II cocirculate in an endemic area of Yakutia, Russia[J]. J Gen Virol, 2001, 82( 11): 2709- 2718. DOI: 10.1099/0022-1317-82-11-2709. [37] LE GAL F, BRICHLER S, DRUGAN T, et al. Genetic diversity and worldwide distribution of the Deltavirus genus: A study of 2, 152 clinical strains[J]. Hepatology, 2017, 66( 6): 1826- 1841. DOI: 10.1002/hep.29574. [38] WATANABE H, NAGAYAMA K, ENOMOTO N, et al. Chronic hepatitis delta virus infection with genotype IIb variant is correlated with progressive liver disease[J]. J Gen Virol, 2003, 84( 12): 3275- 3289. DOI: 10.1099/vir.0.19499-0. [39] WANG Y, GLENN JS, WINTERS MA, et al. A new dual-targeting real-time RT-PCR assay for hepatitis D virus RNA detection[J]. Diagn Microbiol Infect Dis, 2018, 92( 2): 112- 117. DOI: 10.1016/j.diagmicrobio.2018.05.016. [40] BRICHLER S, LE GAL F, BUTT A, et al. Commercial real-time reverse transcriptase PCR assays can underestimate or fail to quantify hepatitis delta virus viremia[J]. Clin Gastroenterol Hepatol, 2013, 11( 6): 734- 740. DOI: 10.1016/j.cgh.2013.01.025. [41] ANOLLI MP, UCEDA RENTERIA S, DEGASPERI E, et al. Comparing methods for plasma HDV RNA quantification in bulevirtide-treated and untreated patients with HDV[J]. JHEP Rep, 2025, 7( 3): 101299. DOI: 10.1016/j.jhepr.2024.101299. [42] YAMASHIRO T, NAGAYAMA K, ENOMOTO N, et al. Quantitation of the level of hepatitis delta virus RNA in serum, by real-time polymerase chain reaction: And its possible correlation with the clinical stage of liver disease[J]. J Infect Dis, 2004, 189( 7): 1151- 1157. DOI: 10.1086/382133. [43] WEDEMEYER H, LEUS M, BATTERSBY TR, et al. HDV RNA assays: Performance characteristics, clinical utility, and challenges[J]. Hepatology, 2025, 81( 2): 637- 650. DOI: 10.1097/HEP.0000000000000584. [44] STELZL E, CIESEK S, CORNBERG M, et al. Reliable quantification of plasma HDV RNA is of paramount importance for treatment monitoring: A European multicenter study[J]. J Clin Virol, 2021, 142: 104932. DOI: 10.1016/j.jcv.2021.104932. [45] STELZL E, BERGER A, CIESEK S, et al. Accurate quantification using the new RoboGene HDV RNA Quantification Kit 3.0: A European multicenter study[J]. J Clin Virol, 2025, 179: 105828. DOI: 10.1016/j.jcv.2025.105828. [46] Longhua Hospital Shanghai University of Traditional Chinese Medicine. Rapid detection system and method for hepatitis D virus nucleic acid by one-step digital PCR: CN202311286029.6[P]. 2024-11-01.上海中医药大学附属龙华医院. 丁型肝炎病毒核酸一步法数字PCR快速检测体系及检测方法: CN202311286029.6[P]. 2024-11-01. [47] Kunming University of Science and Technology. Dual fluorescence quantitative PCR detection reagent for all genotypes of hepatitis D virus: CN202411010160.4[P]. 2024-11-12.昆明理工大学. 丁型肝炎病毒全基因型双重荧光定量PCR检测试剂: CN202411010160.4[P]. 2024-11-12. [48] Shenzhen Yahuilong Biotechnology Co., Ltd. Nucleic acid combination, kit and application for detecting hepatitis D virus: CN202310590474.5[P]. 2023-11-24.深圳市亚辉龙生物科技股份有限公司. 用于检测丁型肝炎病毒的核酸组合及试剂盒和应用: CN202310590474.5[P]. 2023-11-24. [49] Beijing Capital Medical University Science Park Technology Development Co., Ltd. Fluorescent quantitative PCR method hepatitis D virus nucleic acid detection kit: CN202210184262.2[P]. 2023-09-01.北京首医科园科技发展有限公司. 荧光定量PCR方法丁型肝炎病毒核酸检测试剂盒: CN202210184262.2[P]. 2023-09-01. [50] Beijing Capital Medical University Science Park Technology Development Co., Ltd. Hepatitis D virus nucleic acid detection kit based on digital PCR technology: CN202110900513.8[P]. 2023-02-17.北京首医科园科技发展有限公司. 基于数字PCR技术的丁型肝炎病毒核酸检测试剂盒: CN202110900513.8[P]. 2023-02-17. [51] CAO YL, TIAN Y, FAN ZH, et al. Establishment and evaluation of a RAA-CRISPR-Cas13a method for detecting KPC carbapenemase genes[J]. Chin J Lab Med, 2024, 47( 2): 159- 164. DOI: 10.3760/cma.j.cn114452-20231009-00195.曹亚玲, 田原, 范子豪, 等. 基于RAA-CRISPR-Cas13a检测KPC型碳青霉烯酶基因方法的建立及评价[J]. 中华检验医学杂志, 2024, 47( 2): 159- 164. DOI: 10.3760/cma.j.cn114452-20231009-00195. -



 PDF下载 ( 764 KB)
PDF下载 ( 764 KB)

 下载:
下载:


