聚乙二醇干扰素-α-2b对HBeAg阴性慢性HBV感染者HBV特异性CD8+T淋巴细胞杀伤功能的影响
DOI: 10.12449/JCH250406
Effect of pegylated interferon-α-2b therapy on cytotoxicity of virus-specific CD8+ T cells in HBeAg-negative patients with chronic hepatitis B virus infection
-
摘要:
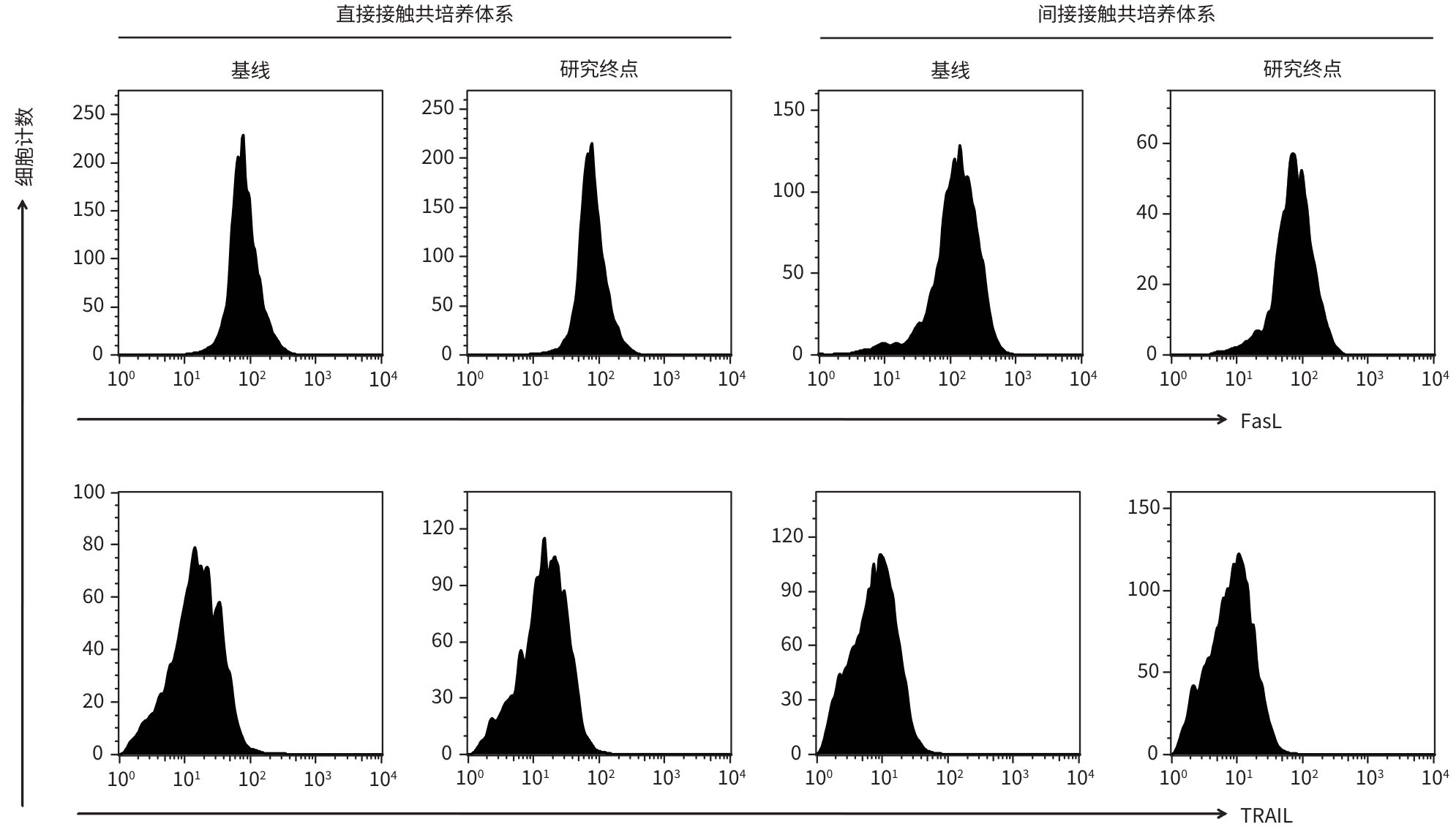
目的 观察经聚乙二醇干扰素-α-2b(PEG-IFN-α-2b)治疗后,HBeAg阴性慢性HBV感染者HBV特异性CD8+T淋巴细胞活性的变化。 方法 纳入2020年4月—2022年6月于新乡医学院第一附属医院、空军军医大学唐都医院就诊的HBeAg阴性慢性HBV感染者53例,予以PEG-IFN-α-2b(180 μg/周,皮下注射)抗病毒治疗,研究终点为HBsAg阴转(疗程<48周)或48周(疗程≥48周),分别选取基线和研究终点外周血单个核细胞,检测外周血T淋巴细胞计数,酶联免疫斑点试验检测分泌穿孔素、颗粒酶B、IFN-γ的HBV特异性CD8+T淋巴细胞频数。选择HLA-A*02限制性患者17例,纯化CD8+T淋巴细胞,建立HBV特异性CD8+T淋巴细胞与HepG2.2.15细胞直接接触和间接接触共培养系统,通过检测上清中乳酸脱氢酶水平计算HepG2.2.15细胞死亡率,检测上清中HBV DNA水平、毒性分子和细胞因子分泌,流式细胞术检测凋亡配体表达,评估HBV特异性CD8+T淋巴细胞的杀伤功能。符合正态分布的计量资料2组间比较采用成组t检验或配对t检验,不符合正态分布的计量资料2组间比较采用Mann-Whitney U检验或Wilcoxon秩和检验。 结果 研究终点时,HBsAg阴转率为30.19%(16/53)。外周血T淋巴细胞计数(CD3+、CD4+、CD8+T淋巴细胞)在基线和研究终点时的差异均无统计学意义(P值均>0.05)。研究终点时,患者分泌穿孔素、颗粒酶B、IFN-γ的HBV特异性CD8+T淋巴细胞频数较基线显著升高(U=177.50,t=11.90,U=186.50,P值均<0.001),发生HBsAg阴转的患者分泌穿孔素、颗粒酶B、IFN-γ的HBV特异性CD8+T淋巴细胞频数亦显著高于未发生HBsAg阴转的患者(U=120.50,t=2.73,U=121.50,P值均<0.01)。在直接接触和间接接触共培养系统中,研究终点时HBV特异性CD8+T淋巴细胞均可诱导HepG2.2.15细胞上清中HBV DNA显著下降(P值均<0.001),IFN-γ和TNF-α分泌水平显著升高(P值均<0.05),但仅在直接接触共培养系统中HBV特异性CD8+T淋巴细胞诱导HepG2.2.15细胞死亡比例升高(13.62%± 3.27% vs 11.39%±2.40%,t=2.27,P=0.030),穿孔素和颗粒酶B分泌水平亦升高(t=72.50,U=52.50,P值均<0.05)。在直接接触和间接接触共培养系统中,发生HBsAg阴转的患者HBV特异性CD8+T淋巴细胞较未发生HBsAg阴转的患者诱导HBV DNA下降水平均更显著(P值均<0.05),IFN-γ和TNF-α分泌水平均升高(P值均<0.05)。 结论 PEG-IFN-α-2b治疗HBeAg阴性慢性HBV感染者可获得较高的HBsAg阴转率,HBV特异性CD8+T淋巴细胞活性显著增强,且与HBsAg阴转密切相关。 -
关键词:
- 聚乙二醇干扰素 /
- 乙型肝炎e抗原 /
- 乙型肝炎病毒 /
- CD8阳性T淋巴细胞
Abstract:Objective To investigate the change in the activity of hepatitis B virus (HBV)-specific CD8+ T cells after pegylated interferon-α-2b (PEG-IFN-α-2b) therapy in HBeAg-negative patients with chronic HBV infection. Methods A total of 53 HBeAg-negative patients with chronic HBV infection who attended The First Affiliated Hospital of Xinxiang Medical University and Tangdu Hospital of Air Force Mdical University from April 2020 to June 2022 were enrolled and treated with PEG-IFN-α-2b (180 μg/week, subcutaneous injection) antiviral therapy. The study endpoint was HBsAg clearance (course of treatment<48 weeks) or 48 weeks (course of treatment≥48 weeks). Peripheral blood mononuclear cells were isolated at baseline and study endpoint, and peripheral blood T cell counts were measured. Enzyme-linked immunospot assay was used to measure the frequency of HBV-specific CD8+ T cells secreting perforin, granzyme B, and interferon-γ. A total of 17 HLA-A*02-restricted patients were selected, and CD8+ T cells were purified to establish direct- and indirect-contact co-culture systems for HBV-specific CD8+ T cells and HepG2.2.15 cells. The level of lactate dehydrogenase in supernatant was measured to calculate the mortality rate of HepG2.2.15 cells, and the levels of HBV DNA, cytotoxic molecules, and cytokines in supernatant were also measured. Flow cytometry was used to measure the expression of apoptosis ligands, and the cytotoxicity of HBV-specific CD8+ T cells was evaluated. The independent samples t-test or the paired t-test was used for comparison of normally distributed continuous data between two groups, and the Mann-Whitney U test or the Wilcoxon rank-sum test was used for comparison of non-normally distributed continuous data between two groups. Results The HBsAg clearance rate at study endpoint was 30.19% (16/53). There were no significant differences in peripheral blood T cell counts (CD3+, CD4+, and CD8+ T cells) between baseline and study endpoint (P>0.05). At study endpoint, there was a significant increase in the frequency of HBV-specific CD8+ T cells secreting perforin, granzyme B, and interferon-γ (U=177.50, t=11.90, U=186.50, all P<0.001), and the patients with HBsAg clearance had a significantly higher frequency of such HBV-specific CD8+ T cells than those without HBsAg clearance (U=120.50, t=2.73, U=121.50, all P<0.01). In the direct- and indirect-contact co-culture systems at study endpoint, HBV-specific CD8+ T cells induced a significant reduction in HBV DNA in the supernatant of HepG2.2.15 cells (all P<0.001) and significant increases in the secretion of interferon-γ and tumor necrosis factor-α (all P<0.05); in the direct-contact co-culture system, HBV-specific CD8+ T cells induced significant increases in the mortality rate of HepG2.2.15 cells (13.62%±3.27% vs 11.39%±2.40%, t=2.27, P=0.030) and the secretion of perforin and granzyme B (t=72.50, U=52.50, both P<0.05). In the direct- and indirect-contact co-culture systems, compared with HBV-specific CD8+ T cells from the patients without HBsAg clearance, the HBV-specific CD8+ T cells from patients with HBsAg clearance had a significantly greater reduction in HBV DNA (P<0.05) and significant increases in the secretion of interferon-γ and tumor necrosis factor-α (P<0.05). Conclusion PEG-IFN-α-2b therapy can help to achieve a relatively high HBsAg clearance rate in HBeAg-negative patients with chronic HBV infection, and the activity of HBV-specific CD8+ T cells is significantly enhanced, which is closely associated with HBsAg clearance. -
表 1 PEG-IFN-α-2b治疗前后患者外周血T淋巴细胞数量的比较
Table 1. Comparison of the absolute peripheral T cell counts in patients before and after PEG-IFN-α-2b therapy
T淋巴细胞类型 基线(n=53) 研究终点(n=53) t值 P值 CD3+(个/μL) 1 771±416 1 750±432 0.55 0.587 CD4+(个/μL) 714±183 717±189 0.13 0.898 CD8+(个/μL) 1 057±287 1 033±346 0.56 0.581 表 2 发生和未发生HBsAg阴转的患者外周血T淋巴细胞数量的比较
Table 2. Comparison of the absolute peripheral T cell counts in patients who achieved HBsAg loss and did not achieve HBsAg loss
T淋巴细胞类型 发生HBsAg阴转(n=16) 未发生HBsAg阴转(n=37) t值 P值 CD3+(个/μL) 1 674±264 1 783±488 0.84 0.407 CD4+(个/μL) 766±161 696±198 1.25 0.219 CD8+(个/μL) 908±274 1 087±363 1.76 0.085 表 3 PEG-IFN-α-2b治疗前后HBV特异性CD8+T淋巴细胞穿孔素、颗粒酶B、IFN-γ分泌水平比较
Table 3. Comparison of the secretion of perforin, granzyme B, and interferon-γ by viral specific CD8+ T cells before and after PEG-IFN-α-2b therapy
指标 基线(n=53) 研究终点(n=53) 统计值 P值 穿孔素(SFC/106个PBMC) 30.00(25.00~34.50) 84.00(65.00~125.00) U=177.50 <0.001 颗粒酶B(SFC/106个PBMC) 12.28±5.41 38.13±13.57 t=11.90 <0.001 IFN-γ(SFC/106个PBMC) 49.00(44.00~56.00) 116.00(87.00~155.00) U=186.50 <0.001 表 4 发生和未发生HBsAg阴转的患者HBV特异性CD8+T淋巴细胞穿孔素、颗粒酶B、IFN-γ分泌水平比较
Table 4. Comparison of the secretion of perforin, granzyme B, and interferon-γ by viral specific CD8+ T cells in patients who achieved and did not achieve HBsAg loss
指标 发生HBsAg阴转(n=16) 未发生HBsAg阴转(n=37) 统计值 P值 穿孔素(SFC/106个PBMC) 125.00(98.25~159.80) 72.00(61.50~106.50) U=120.50 <0.001 颗粒酶B(SFC/106个PBMC) 45.44±15.88 34.97±11.27 t=2.73 0.009 IFN-γ(SFC/106个PBMC) 141.50(116.00~242.30) 97.00(80.00~128.00) U=121.50 <0.001 表 5 PEG-IFN-α-2b治疗前后HBV特异性CD8+T淋巴细胞在共培养系统中的功能变化比较
Table 5. The functional comparison of HBV- specific CD8+ T cells in co-culture systems between patients before and after PEG-IFN-α-2b therapy
指标 基线(n=17) 研究终点(n=17) 统计值 P值 直接接触共培养系统 靶细胞死亡比例(%) 11.39±2.40 1) 13.62±3.27 1) t=2.27 0.030 HBV DNA(IU/mL) 6.47±1.67 4.37±1.35 t=4.04 <0.001 穿孔素(pg/mL) 106.61(70.06~177.29)1) 173.64(127.68~225.12)1) t=72.50 0.014 颗粒酶B(pg/mL) 53.07(42.63~89.61)1) 107.92(80.04~151.37)1) U=52.50 0.002 IFN-γ(pg/mL) 48.99(30.53~86.27) 131.41(85.91~178.94) U=44.50 <0.001 TNF-α(pg/mL) 653.47(511.80~963.86) 1 263.93(707.94~2 488.78) U=68.00 0.009 FasL MFI 171.79±92.40 190.24±62.64 t=0.68 0.503 TRAIL MFI 26.47±8.38 24.80±8.35 t=0.58 0.564 间接接触共培养系统 靶细胞死亡比例(%) 5.59±1.07 6.09±1.72 t=1.01 0.318 HBV DNA(IU/mL) 7.18±1.49 4.82±1.32 t=4.90 <0.001 穿孔素(pg/mL) 85.56(61.38~136.40) 104.23(73.16~150.03) U=127.50 0.570 颗粒酶B(pg/mL) 46.11(34.37~77.00) 61.77(42.20~94.40) U=106.00 0.190 IFN-γ(pg/mL) 34.08(25.56~72.77) 140.54(88.58~220.20) U=38.00 <0.001 TNF-α(pg/mL) 707.92(511.81~887.59) 1 100.72(637.10~1 748.82) U=76.00 0.019 FasL MFI 177.50±86.15 199.41±71.01 t=0.81 0.425 TRAIL MFI 28.70±5.70 25.26±7.51 t=1.50 0.143 注:1)与间接接触共培养系统中同一指标比较,P<0.05。
表 6 发生和未发生HBsAg阴转患者HBV特异性CD8+T淋巴细胞在共培养系统中的功能变化比较
Table 6. The functional comparison of HBV- specific CD8+ T cells in co-culture systems between patients who achieved and did not achieve HBsAg loss
指标 发生HBsAg阴转(n=7) 未发生HBsAg阴转(n=10) 统计值 P值 直接接触共培养系统 靶细胞死亡比例(%) 16.23±3.48 11.79±1.41 t=3.66 0.002 HBV DNA(IU/mL) 3.61±0.91 4.91±1.38 t=2.18 0.046 穿孔素(pg/mL) 212.01(168.58~357.09) 148.19(104.84~194.37) U=12.50 0.032 颗粒酶B(pg/mL) 154.03(127.91~255.78) 84.39(75.47~112.91) U=5.50 0.005 IFN-γ(pg/mL) 181.11(155.50~210.17) 90.88(73.84~121.21) U=0.00 <0.001 TNF-α(pg/mL) 2 690.28(1 263.82~3 311.53) 882.14(612.55~1 345.37) U=7.00 0.005 FasL MFI 209.04±66.87 176.89±59.36 t=1.04 0.314 TRAIL MFI 21.20±6.65 27.31±8.80 t=1.55 0.143 间接接触共培养系统 靶细胞死亡比例(%) 6.10±1.65 6.08±1.87 t=0.02 0.983 HBV DNA(IU/mL) 3.96±0.60 5.42±1.38 t=2.61 0.020 穿孔素(pg/mL) 119.02(91.76~160.03) 76.88(71.30~148.84) U=25.00 0.364 颗粒酶B(pg/mL) 72.21(47.85~87.87) 58.73(37.19~103.08) U=28.50 0.558 IFN-γ(pg/mL) 217.03(142.57~236.89) 104.62(68.87~142.61) U=9.00 0.010 TNF-α(pg/mL) 2 091.67(1 198.20~2 483.93) 746.00(560.76~1 062.33) U=4.00 0.001 FasL MFI 207.43±51.82 193.79±84.19 t=0.38 0.709 TRAIL MFI 21.70±7.05 27.75±7.09 t=1.74 0.103 -
[1] STASI C, SILVESTRI C, VOLLER F. Hepatitis B vaccination and immunotherapies: an update[J]. Clin Exp Vaccine Res, 2020, 9( 1): 1- 7. DOI: 10.7774/cevr.2020.9.1.1. [2] SHAN S, ZHAO XY, JIA JD. Comprehensive approach to controlling chronic hepatitis B in China[J]. Clin Mol Hepatol, 2024, 30( 2): 135- 143. DOI: 10.3350/cmh.2023.0412. [3] CAO GY, LIU J, LIU M. Trends in mortality of liver disease due to hepatitis B in China from 1990 to 2019: Findings from the global burden of disease study[J]. Chin Med J(Engl), 2022, 135( 17): 2049- 2055. DOI: 10.1097/CM9.0000000000002331. [4] POLLICINO T, CAMINITI G. HBV-integration studies in the clinic: Role in the natural history of infection[J]. Viruses, 2021, 13( 3): 368. DOI: 10.3390/v13030368. [5] ZHUANG H. Debates on the natural history of chronic hepatitis B virus infection during the update of Guidelines for the Prevention and Treatment of Chronic Hepatitis B(2022 Version)[J]. J Clin Hepatol, 2023, 39( 6): 1295- 1298. DOI: 10.3969/j.issn.1001-5256.2023.06.006.庄辉.《慢性乙型肝炎防治指南(2022年版)》修订过程中关于慢性HBV感染自然史的讨论[J]. 临床肝胆病杂志, 2023, 39( 6): 1295- 1298. DOI: 10.3969/j.issn.1001-5256.2023.06.006. [6] CHU CM, LIAW YF. HBsAg seroclearance in asymptomatic carriers of high endemic areas: Appreciably high rates during a long-term follow-up[J]. Hepatology, 2007, 45( 5): 1187- 1192. DOI: 10.1002/hep.21612. [7] HUANG Y, QI M, LIAO CJ, et al. Analysis of the efficacy and safety of PEGylated interferon-α2b treatment in inactive hepatitis B surface antigen carriers[J]. Infect Dis Ther, 2021, 10( 4): 2323- 2331. DOI: 10.1007/s40121-021-00511-w. [8] DUAN SP, ZHU LH, HOU LJ, et al. Effect of tenofovir disoproxil fumarate antiviral therapy on virus-specific CD8+T Cells function in patients with chronic hepatitis B[J]. Chin J Hepatol, 2021, 29( 5): 421- 426. DOI: 10.3760/cma.j.cn501113-20191113-00420.段树鹏, 朱利红, 侯丽娟, 等. 替诺福韦酯抗病毒治疗对慢性乙型肝炎患者病毒特异性CD8+T细胞功能的影响[J]. 中华肝脏病杂志, 2021, 29( 5): 421- 426. DOI: 10.3760/cma.j.cn501113-20191113-00420. [9] WAN LH, ZHANG J, CHEN S, et al. Influnces of tenofovir dipivoxil combined with compound Yiganling tablets on immune function, liver and kidney function in HBV carrier complicated with pulmonary tuberculosis[J]. J Clin Exp Med, 2023, 22( 18): 1938- 1942. DOI: 10.3969/j.issn.1671-4695.2023.18.010.万丽红, 张静, 陈爽, 等. 替诺福韦酯联合复方益肝灵片对肺结核合并HBV携带患者免疫功能及肝肾功能的影响[J]. 临床和实验医学杂志, 2023, 22( 18): 1938- 1942. DOI: 10.3969/j.issn.1671-4695.2023.18.010. [10] DONG J, YANG XF, WANG LX, et al. Modulation of tim-3 expression by antigen-dependent and-independent factors on T cells from patients with chronic hepatitis B virus infection[J]. Front Cell Infect Microbiol, 2017, 7: 98. DOI: 10.3389/fcimb.2017.00098. [11] McLANE LM, ABDEL-HAKEEM MS, WHERRY EJ. CD8 T cell exhaustion during chronic viral infection and cancer[J]. Annu Rev Immunol, 2019, 37: 457- 495. DOI: 10.1146/annurev-immunol-041015-055318. [12] WONG GLH, GANE E, LOK ASF. How to achieve functional cure of HBV: Stopping NUCs, adding interferon or new drug development?[J]. J Hepatol, 2022, 76( 6): 1249- 1262. DOI: 10.1016/j.jhep.2021.11.024. [13] DENG W, JIANG TT, BI XY, et al. Progress on the treatment of chronic hepatitis B with interferons[J/CD]. Chin J Liver Dis(Electronic Version), 2023, 15( 2): 1- 6. DOI: 10.3969/j.issn.1674-7380.2023.02.001.邓雯, 蒋婷婷, 毕潇月, 等. 干扰素治疗慢性乙型肝炎研究进展[J/CD]. 中国肝脏病杂志(电子版), 2023, 15( 2): 1- 6. DOI: 10.3969/j.issn.1674-7380.2023.02.001. [14] ISLAM M, KUMAR K, SEVAK JK, et al. Immune drivers of HBsAg loss in HBeAg-negative CHB patients after stopping nucleotide analog and administration of Peg-IFN[J]. Hepatol Commun, 2023, 7( 5): e0098. DOI: 10.1097/HC9.0000000000000098. [15] PANG XQ, LI X, ZHU WH, et al. LAG3+ erythroid progenitor cells inhibit HBsAg seroclearance during finite pegylated interferon treatment through LAG3 and TGF-β[J]. Antiviral Res, 2023, 213: 105592. DOI: 10.1016/j.antiviral.2023.105592. [16] HUANG D, YAN WM, HAN MF, et al. Insufficient immunity led to virologic breakthrough in NAs-treated chronic hepatitis B patients switching to Peg-IFN-α[J]. Antiviral Res, 2022, 197: 105220. DOI: 10.1016/j.antiviral.2021.105220. [17] WANG DY, FU BQ, SHEN XK, et al. Restoration of HBV-specific CD8+ T-cell responses by sequential low-dose IL-2 treatment in non-responder patients after IFN-α therapy[J]. Signal Transduct Target Ther, 2021, 6( 1): 376. DOI: 10.1038/s41392-021-00776-0. [18] MONTANARI NR, CONCEIÇÃO-NETO N, van den WYNGAERT I, et al. Differential gene expression, irrespective of circulating hepatitis B surface antigen levels, between inactive carrier and nucleos(t)ide analogue-treated hepatitis B virus patients[J]. J Infect Dis, 2022, 225( 8): 1471- 1476. DOI: 10.1093/infdis/jiaa614. [19] SONG AX, LIN X, LU JF, et al. Pegylated interferon treatment for the effective clearance of hepatitis B surface antigen in inactive HBsAg carriers: A meta-analysis[J]. Front Immunol, 2021, 12: 779347. DOI: 10.3389/fimmu.2021.779347. [20] CHEN XB, LIU FF, SHU FL, et al. Peginterferon Alfa-2b combined with tenofovir disoproxil fumarate induced high clinical cure rate in inactive chronic hepatitis B virus carriers[J]. Clin Res Hepatol Gastroenterol, 2021, 45( 5): 101723. DOI: 10.1016/j.clinre.2021.101723. [21] CAO ZH, LIU YL, MA LN, et al. A potent hepatitis B surface antigen response in subjects with inactive hepatitis B surface antigen carrier treated with pegylated-interferon alpha[J]. Hepatology, 2017, 66( 4): 1058- 1066. DOI: 10.1002/hep.29213. [22] ZHU L, LI J, XU JC, et al. Significance of T-cell subsets for clinical response to peginterferon Alfa-2a therapy in HBeAg-positive chronic hepatitis B patients[J]. Int J Gen Med, 2022, 15: 4441- 4451. DOI: 10.2147/IJGM.S356696. [23] PENG MJ, GUO XQ, ZHANG WL, et al. Effect of pegylated interferon-α2b add-on therapy on renal function in chronic hepatitis B patients: A real-world experience[J]. Front Microbiol, 2022, 13: 980250. DOI: 10.3389/fmicb.2022.980250. [24] RASKOV H, ORHAN A, CHRISTENSEN JP, et al. Cytotoxic CD8+ T cells in cancer and cancer immunotherapy[J]. Br J Cancer, 2021, 124( 2): 359- 367. DOI: 10.1038/s41416-020-01048-4. [25] VERDON DJ, MULAZZANI M, JENKINS MR. Cellular and molecular mechanisms of CD8+ T cell differentiation, dysfunction and exhaustion[J]. Int J Mol Sci, 2020, 21( 19): 7357. DOI: 10.3390/ijms21197357. [26] PHILLIPS S, CHOKSHI S, RIVA A, et al. CD8+ T cell control of hepatitis B virus replication: Direct comparison between cytolytic and noncytolytic functions[J]. J Immunol, 2010, 184( 1): 287- 295. DOI: 10.4049/jimmunol.0902761. [27] SHAO X, MA JT, JIA SN, et al. Interleukin-35 suppresses antiviral immune response in chronic hepatitis B virus infection[J]. Front Cell Infect Microbiol, 2017, 7: 472. DOI: 10.3389/fcimb.2017.00472. [28] Chinese Society of Hepatology, Chinese Society of Infectious Diseases. Guidelines for the prevention and treatment of chronic hepatitis B(2022 version)[J]. Chin J Infect Dis, 2023, 41( 1): 3- 28. DOI: 10.3760/cma.j.cn311365-20230220-00050.中华医学会肝病学分会, 中华医学会感染病学分会. 慢性乙型肝炎防治指南(2022年版)[J]. 中华传染病杂志, 2023, 41( 1): 3- 28. DOI: 10.3760/cma.j.cn311365-20230220-00050. -



 PDF下载 ( 990 KB)
PDF下载 ( 990 KB)

 下载:
下载:


