| [1] |
WU J, YIN F, ZHOU X. Efficacy of nucleoside analogues for hepatitis B virus-related liver failure: A network meta-analysis[J]. Acta Pharm, 2018, 68(1): 19-30. DOI: 10.2478/acph-2018-0010. |
| [2] |
LEE CW, CHEN YF, WU HH, et al. Historical perspectives and advances in mesenchymal stem cell research for the treatment of liver diseases[J]. Gastroenterology, 2018, 154(1): 46-56. DOI: 10.1053/j.gastro.2017.09.049. |
| [3] |
KOK B, KARVELLAS CJ. Management of cerebral edema in acute liver failure[J]. Semin Respir Crit Care Med, 2017, 38(6): 821-829. DOI: 10.1055/s-0037-1608772. |
| [4] |
TORRES S, BAULIES A, INSAUSTI-URKIA N, et al. Endoplasmic reticulum stress-induced upregulation of STARD1 promotes acetaminophen-induced acute liver failure[J]. Gastroenterology, 2019, 157(2): 552-568. DOI: 10.1053/j.gastro.2019.04.023. |
| [5] |
YAN N, YAN T, XIA Y, et al. The pathophysiological function of non-gastrointestinal farnesoid X receptor[J]. Pharmacol Ther, 2021, 226: 107867. DOI: 10.1016/j.pharmthera.2021.107867. |
| [6] |
WILLIAMS CM, HARPER CALDERON J, E H, et al. Monomeric/dimeric forms of Fgf15/FGF19 show differential activity in hepatocyte proliferation and metabolic function[J]. FASEB J, 2021, 35(2): e21286. DOI: 10.1096/fj.202002203R. |
| [7] |
GULFO J, ROTONDO F, ÁVALOS de LEÓN CG, et al. FGF15 improves outcomes after brain dead donor liver transplantation with steatotic and non-steatotic grafts in rats[J]. J Hepatol, 2020, 73(5): 1131-1143. DOI: 10.1016/j.jhep.2020.05.007. |
| [8] |
BAI Q, ZHANG X, XU L, et al. Oxysterol sulfation by cytosolic sulfotransferase suppresses liver X receptor/sterol regulatory element binding protein-1c signaling pathway and reduces serum and hepatic lipids in mouse models of nonalcoholic fatty liver disease[J]. Metabolism, 2012, 61(6): 836-845. DOI: 10.1016/j.metabol.2011.11.014. |
| [9] |
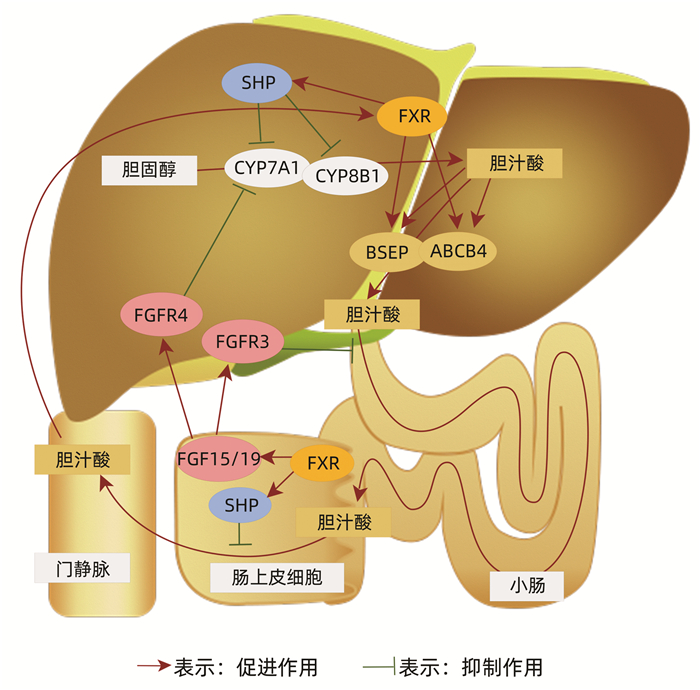
CHEN YY, LAN YM, WANG MG, et al. Mechanism of action of bile acid-farnesoid X receptor-intestinal microecological axis in the development of liver failure and liver regeneration[J]. J Clin Hepatol, 2021, 37(2): 480-484. DOI: 10.3969/j.issn.1001-5256.2021.02.049. |
| [10] |
|
| [11] |
van MIERLO KM, SCHAAP FG, DEJONG CH, et al. Liver resection for cancer: New developments in prediction, prevention and management of postresectional liver failure[J]. J Hepatol, 2016, 65(6): 1217-1231. DOI: 10.1016/j.jhep.2016.06.006. |
| [12] |
HASHEMI GORADEL N, DARABI M, SHAMSASENJAN K, et al. Methods of liver stem cell therapy in rodents as models of human liver regeneration in hepatic failure[J]. Adv Pharm Bull, 2015, 5(3): 293-298. DOI: 10.5681/apb.2015.041. |
| [13] |
MICHALOPOULOS GK. Hepatostat: Liver regeneration and normal liver tissue maintenance[J]. Hepatology, 2017, 65(4): 1384-1392. DOI: 10.1002/hep.28988. |
| [14] |
KHOLODENKO IV, YARYGIN KN. Cellular mechanisms of liver regeneration and cell-based therapies of liver diseases[J]. Biomed Res Int, 2017, 2017: 8910821. DOI: 10.1155/2017/8910821. |
| [15] |
ADAMEK B, ZALEWSKA-ZIOB M, STRZELCZYK JK, et al. Hepatocyte growth factor and epidermal growth factor activity during later stages of rat liver regeneration upon interferon α-2b influence[J]. Clin Exp Hepatol, 2017, 3(1): 9-15. DOI: 10.5114/ceh.2017.65499. |
| [16] |
MURTHA-LEMEKHOVA A, FUCHS J, GHAMARNEJAD O, et al. Influence of cytokines, circulating markers and growth factors on liver regeneration and post-hepatectomy liver failure: A systematic review and meta-analysis[J]. Sci Rep, 2021, 11(1): 13739. DOI: 10.1038/s41598-021-92888-4. |
| [17] |
DOIGNON I, JULIEN B, SERRIÈRE-LANNEAU V, et al. Immediate neuroendocrine signaling after partial hepatectomy through acute portal hyperpressure and cholestasis[J]. J Hepatol, 2011, 54(3): 481-488. DOI: 10.1016/j.jhep.2010.07.012. |
| [18] |
SOUSA IP Jr, CARVALHO C, GOMES A. Current understanding of the role of cholesterol in the life cycle of alphaviruses[J]. Viruses, 2020, 13(1): 35. DOI: 10.3390/v13010035. |
| [19] |
GUZIOR DV, QUINN RA. Review: Microbial transformations of human bile acids[J]. Microbiome, 2021, 9(1): 140. DOI: 10.1186/s40168-021-01101-1. |
| [20] |
MATHUR B, SHAJAHAN A, ARIF W, et al. Nuclear receptors FXR and SHP regulate protein N-glycan modifications in the liver[J]. Sci Adv, 2021, 7(17): eabf4865. DOI: 10.1126/sciadv.abf4865. |
| [21] |
TRAUNER M, BOYER JL. Bile salt transporters: Molecular characterization, function, and regulation[J]. Physiol Rev, 2003, 83(2): 633-671. DOI: 10.1152/physrev.00027.2002. |
| [22] |
HOFMANN AF. The enterohepatic circulation of bile acids in mammals: Form and functions[J]. Front Biosci (Landmark Ed), 2009, 14: 2584-2598. DOI: 10.2741/3399. |
| [23] |
PEREZ MJ, BRIZ O. Bile-acid-induced cell injury and protection[J]. World J Gastroenterol, 2009, 15(14): 1677-1689. DOI: 10.3748/wjg.15.1677. |
| [24] |
ALVAREZ-SOLA G, URIARTE I, LATASA MU, et al. Bile acids, FGF15/19 and liver regeneration: From mechanisms to clinical applications[J]. Biochim Biophys Acta Mol Basis Dis, 2018, 1864(4 Pt B): 1326-1334. DOI: 10.1016/j.bbadis.2017.06.025. |
| [25] |
PANZITT K, WAGNER M. FXR in liver physiology: Multiple faces to regulate liver metabolism[J]. Biochim Biophys Acta Mol Basis Dis, 2021, 1867(7): 166133. DOI: 10.1016/j.bbadis.2021.166133. |
| [26] |
MEYER K, MORALES-NAVARRETE H, SEIFERT S, et al. Bile canaliculi remodeling activates YAP via the actin cytoskeleton during liver regeneration[J]. Mol Syst Biol, 2020, 16(2): e8985. DOI: 10.15252/msb.20198985. |
| [27] |
PENG J, YU J, XU H, et al. Enhanced liver regeneration after partial hepatectomy in sterol regulatory element-binding protein (SREBP)-1c-null mice is associated with increased hepatocellular cholesterol availability[J]. Cell Physiol Biochem, 2018, 47(2): 784-799. DOI: 10.1159/000490030. |
| [28] |
DE GIORGI M, JARRETT KE, BURTON JC, et al. Depletion of essential isoprenoids and ER stress induction following acute liver-specific deletion of HMG-CoA reductase[J]. J Lipid Res, 2020, 61(12): 1675-1686. DOI: 10.1194/jlr.RA120001006. |
| [29] |
NUÑEZ-GARCIA M, GOMEZ-SANTOS B, SAENZ de URTURI D, et al. Atorvastatin provides a new lipidome improving early regeneration after partial hepatectomy in osteopontin deficient mice[J]. Sci Rep, 2018, 8(1): 14626. DOI: 10.1038/s41598-018-32919-9. |
| [30] |
JANG YO, KIM SH, CHO MY, et al. Synergistic effects of simvastatin and bone marrow-derived mesenchymal stem cells on hepatic fibrosis[J]. Biochem Biophys Res Commun, 2018, 497(1): 264-271. DOI: 10.1016/j.bbrc.2018.02.067. |
| [31] |
TOKUNAGA T, IKEGAMI T, YOSHIZUMI T, et al. Beneficial effects of fluvastatin on liver microcirculation and regeneration after massive hepatectomy in rats[J]. Dig Dis Sci, 2008, 53(11): 2989-2994. DOI: 10.1007/s10620-008-0241-y. |
| [32] |
ZHANG L, HUANG X, MENG Z, et al. Significance and mechanism of CYP7a1 gene regulation during the acute phase of liver regeneration[J]. Mol Endocrinol, 2009, 23(2): 137-145. DOI: 10.1210/me.2008-0198. |
| [33] |
IBRAHIM S, DAYOUB R, SABERI V, et al. Augmenter of Liver Regeneration (ALR) regulates bile acid synthesis and attenuates bile acid-induced apoptosis via glycogen synthase kinase-3β (GSK-3β) inhibition[J]. Exp Cell Res, 2020, 397(1): 112343. DOI: 10.1016/j.yexcr.2020.112343. |
| [34] |
HAKIM A, MOLL M, BRANCALE J, et al. Genetic variation in the mitochondrial glycerol-3-phosphate acyltransferase is associated with liver injury[J]. Hepatology, 2021, 74(6): 3394-3408. DOI: 10.1002/hep.32038. |
| [35] |
ZHANG J, LI J, CHEN Y, et al. Prognostic factors related to the mortality rate of acute-on-chronic liver failure patients[J]. Diabetes Metab Syndr Obes, 2021, 14: 2573-2580. DOI: 10.2147/DMSO.S309641. |
| [36] |
DELGADO-COELLO B, BRIONES-ORTA MA, MACíAS-SILVA M, et al. Cholesterol: Recapitulation of its active role during liver regeneration[J]. Liver Int, 2011, 31(9): 1271-1284. DOI: 10.1111/j.1478-3231.2011.02542.x. |
| [37] |
DAL K, BULUR O, ATA N, et al. The role of insulin - like growth factor - 1 on steatohepatitis[J]. Acta Gastroenterol Belg, 2017, 80(1): 21-24.
|
| [38] |
DING BS, LIU CH, SUN Y, et al. HDL activation of endothelial sphingosine-1-phosphate receptor-1 (S1P1) promotes regeneration and suppresses fibrosis in the liver[J]. JCI Insight, 2016, 1(21): e87058. DOI: 10.1172/jci.insight.87058. |
| [39] |
SYDOR S, GU Y, SCHLATTJAN M, et al. Steatosis does not impair liver regeneration after partial hepatectomy[J]. Lab Invest, 2013, 93(1): 20-30. DOI: 10.1038/labinvest.2012.142. |
| [40] |
WANG G, CHEN QM, MINUK GY, et al. Enhanced expression of cytosolic fatty acid binding protein and fatty acid uptake during liver regeneration in rats[J]. Mol Cell Biochem, 2004, 262(1-2): 41-49. DOI: 10.1023/b:mcbi.0000038214.52184.82. |
| [41] |
CICOGNANI C, MALAVOLTI M, MORSELLI-LABATE AM, et al. Serum lipid and lipoprotein patterns in patients with liver cirrhosis and chronic active hepatitis[J]. Arch Intern Med, 1997, 157(7): 792-796.
|
| [42] |
BAUMBERGER C, ULEVITCH RJ, DAYER JM. Modulation of endotoxic activity of lipopolysaccharide by high-density lipoprotein[J]. Pathobiology, 1991, 59(6): 378-383. DOI: 10.1159/000163681. |
| [43] |
GALBOIS A, THABUT D, TAZI KA, et al. Ex vivo effects of high-density lipoprotein exposure on the lipopolysaccharide-induced inflammatory response in patients with severe cirrhosis[J]. Hepatology, 2009, 49(1): 175-184. DOI: 10.1002/hep.22582. |
| [44] |
GUO L, AI J, ZHENG Z, et al. High density lipoprotein protects against polymicrobe-induced sepsis in mice[J]. J Biol Chem, 2013, 288(25): 17947-17953. DOI: 10.1074/jbc.M112.442699. |
| [45] |
ETOGO-ASSE FE, VINCENT RP, HUGHES SA, et al. High density lipoprotein in patients with liver failure; relation to sepsis, adrenal function and outcome of illness[J]. Liver Int, 2012, 32(1): 128-136. DOI: 10.1111/j.1478-3231.2011.02657.x. |
| [46] |
MANKA P, OLLIGES V, BECHMANN LP, et al. Low levels of blood lipids are associated with etiology and lethal outcome in acute liver failure[J]. PLoS One, 2014, 9(7): e102351. DOI: 10.1371/journal.pone.0102351. |
| [47] |
ŽALOUDKOVÁ L, TICHÁ A, NEKVINDOVÁ J, et al. Different forms of ursolic acid and their effect on liver regeneration[J]. Evid Based Complement Alternat Med, 2020, 2020: 4074068. DOI: 10.1155/2020/4074068. |
| [48] |
XU L, KIM JK, BAI Q, et al. 5-cholesten-3β, 25-diol 3-sulfate decreases lipid accumulation in diet-induced nonalcoholic fatty liver disease mouse model[J]. Mol Pharmacol, 2013, 83(3): 648-658. DOI: 10.1124/mol.112.081505. |
| [49] |
ZHANG X, BAI Q, XU L, et al. Cytosolic sulfotransferase 2B1b promotes hepatocyte proliferation gene expression in vivo and in vitro[J]. Am J Physiol Gastrointest Liver Physiol, 2012, 303(3): g344-g355. DOI: 10.1152/ajpgi.00403.2011. |
| [50] |
XU L, SHEN S, MA Y, et al. 25-Hydroxycholesterol-3-sulfate attenuates inflammatory response via PPARγ signaling in human THP-1 macrophages[J]. Am J Physiol Endocrinol Metab, 2012, 302(7): e788-e799. DOI: 10.1152/ajpendo.00337.2011. |
| [51] |
SALADIN R, FAJAS L, DANA S, et al. Differential regulation of peroxisome proliferator activated receptor gamma1 (PPARgamma1) and PPARgamma2 messenger RNA expression in the early stages of adipogenesis[J]. Cell Growth Differ, 1999, 10(1): 43-48.
|
| [52] |
NING Y, KIM JK, MIN HK, et al. Cholesterol metabolites alleviate injured liver function and decrease mortality in an LPS-induced mouse model[J]. Metabolism, 2017, 71: 83-93. DOI: 10.1016/j.metabol.2016.12.007. |
| [53] |
INOUE M, SHINOHARA ML. The role of interferon-β in the treatment of multiple sclerosis and experimental autoimmune encephalomyelitis-in the perspective of inflammasomes[J]. Immunology, 2013, 139(1): 11-18. DOI: 10.1111/imm.12081. |
| [54] |
|
| [55] |
REBOLDI A, DANG EV, MCDONALD JG, et al. Inflammation. 25-Hydroxycholesterol suppresses interleukin-1-driven inflammation downstream of type I interferon[J]. Science, 2014, 345(6197): 679-684. DOI: 10.1126/science.1254790. |







 DownLoad:
DownLoad: