| [1] |
MARRERO JA, KULIK LM, SIRLIN C, et al. Diagnosis, staging, and management of hepatocellular carcinoma: 2018 practice guidance by the american association for the study of liver diseases[J]. Hepatology, 2018, 68(2): 723-50. DOI: 10.1002/hep.29913 |
| [2] |
European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma[J]. J Hepatol, 2018, 69(1): 182-236. DOI: 10.1016/j.jhep.2018.03.019 |
| [3] |
Bureau of Medical Administration, National Health Commission of the People's Republic of China. Guidelines for diagnosis and treatment of primary liver cancer in China (2019 edition)[J]. J Clin Hepatol, 2020, 36(2): 277-292. (in Chinese) DOI: 10.3969/j.issn.1001-5256.2020.02.007 |
| [4] |
SIEGHART W, HUCKE F, PECK-RADOSAVLJEVIC M. Transarterial chemoembolization: Modalities, indication, and patient selection[J]. J Hepatol, 2015, 62(5): 1187-1195. DOI: 10.1016/j.jhep.2015.02.010 |
| [5] |
LENCIONI R, LLOVET JM. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma[J]. Semin Liver Dis, 2010, 30(1): 52-60. DOI: 10.1055/s-0030-1247132 |
| [6] |
GILLMORE R, STUART S, KIRKWOOD A, et al. EASL and mRECIST responses are independent prognostic factors for survival in hepatocellular cancer patients treated with transarterial embolization[J]. J Hepatol, 2011, 55(6): 1309-1316. DOI: 10.1016/j.jhep.2011.03.007 |
| [7] |
JUNG ES, KIM JH, YOON EL, et al. Comparison of the methods for tumor response assessment in patients with hepatocellular carcinoma undergoing transarterial chemoembolization[J]. J Hepatol, 2013, 58(6): 1181-1187. DOI: 10.1016/j.jhep.2013.01.039 |
| [8] |
KIM BK, KIM KA, PARK JY, et al. Prospective comparison of prognostic values of modified Response Evaluation Criteria in Solid Tumours with European Association for the Study of the Liver criteria in hepatocellular carcinoma following chemoembolisation[J]. Eur J Cancer, 2013, 49(4): 826-834. DOI: 10.1016/j.ejca.2012.08.022 |
| [9] |
PRAJAPATI HJ, SPIVEY JR, HANISH SI, et al. mRECIST and EASL responses at early time point by contrast-enhanced dynamic MRI predict survival in patients with unresectable hepatocellular carcinoma (HCC) treated by doxorubicin drug-eluting beads transarterial chemoembolization (DEB TACE)[J]. Ann Oncol, 2013, 24(4): 965-973. DOI: 10.1093/annonc/mds605 |
| [10] |
KIM CJ, KIM HJ, PARK JH, et al. Radiologic response to transcatheter hepatic arterial chemoembolization and clinical outcomes in patients with hepatocellular carcinoma[J]. Liver Int, 2014, 34(2): 305-312. DOI: 10.1111/liv.12270 |
| [11] |
KIM BK, KIM SU, KIM KA, et al. Complete response at first chemoembolization is still the most robust predictor for favorable outcome in hepatocellular carcinoma[J]. J Hepatol, 2015, 62(6): 1304-1310. DOI: 10.1016/j.jhep.2015.01.022 |
| [12] |
VINCENZI B, MAIO MD, SILLETTA M, et al. Prognostic relevance of objective response according to EASL Criteria and mRECIST Criteria in hepatocellular carcinoma patients treated with loco-regional therapies: A literature-based Meta-analysis[J]. Plos One, 2015, 10(7): 1-12. DOI: 10.1371/journal.pone.0133488 |
| [13] |
TACHER V, LIN M, DURAN R, et al. Comparison of existing response criteria in patients with hepatocellular carcinoma treated with transarterial chemoembolization using a 3D quantitative approach[J]. Radiology, 2016, 278(1): 275-284. DOI: 10.1148/radiol.2015142951 |
| [14] |
GEORGIADES C, GESCHWIND JF, HARRISON N, et al. Lack of response after initial chemoembolization for hepatocellular carcinoma: Does it predict failure of subsequent treatment?[J]. Radiology, 2012, 265(1): 115-123. DOI: 10.1148/radiol.12112264 |
| [15] |
LIU L, WANG W, CHEN H, et al. EASL- and mRECIST-evaluated responses to combination therapy of sorafenib with transarterial chemoembolization predict survival in patients with hepatocellular carcinoma[J]. Clin Cancer Res, 2014, 20(6): 1623-1631. DOI: 10.1158/1078-0432.CCR-13-1716 |
| [16] |
SHIM JH, LEE HC, KIM SO, et al. Which response criteria best help predict survival of patients with hepatocellular carcinoma following chemoembolization? A validation study of old and new models[J]. Radiology, 2012, 262(2): 708-718. DOI: 10.1148/radiol.11110282 |
| [17] |
JUNG YK, JUNG CH, SEO YS, et al. BCLC stage B is a better designation for single large hepatocellular carcinoma than BCLC stage A[J]. J Gastroenterol Hepatol, 2016, 31(2): 467-474. DOI: 10.1111/jgh.13152 |
| [18] |
ZHAO Y, WANG WJ, GUAN S, et al. Sorafenib combined with transarterial chemoembolization for the treatment of advanced hepatocellular carcinoma: A large-scale multicenter study of 222 patients[J]. Ann Oncol, 2013, 24(7): 1786-1792. DOI: 10.1093/annonc/mdt072 |
| [19] |
WANG YY, ZHONG JH, XU HF, et al. A modified staging of early and intermediate hepatocellular carcinoma based on single tumour > 7 cm and multiple tumours beyond up-to-seven criteria[J]. Aliment Pharmacol Ther, 2019, 49(2): 202-210. DOI: 10.1111/apt.15074 |



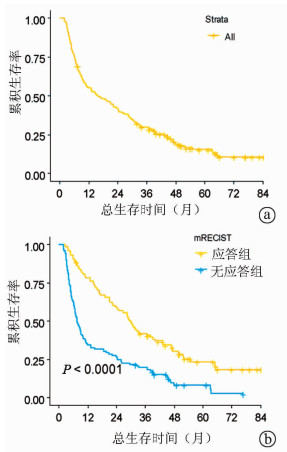




 DownLoad:
DownLoad: