二巯丁二酸联合硫酸锌与青霉胺治疗儿童肝豆状核变性的效果比较
DOI: 10.3969/j.issn.1001-5256.2023.09.017
Effect of dimercaptosuccinic acid combined with zinc sulfate versus D-penicillamine in treatment of children with Wilson’s disease
-
摘要:
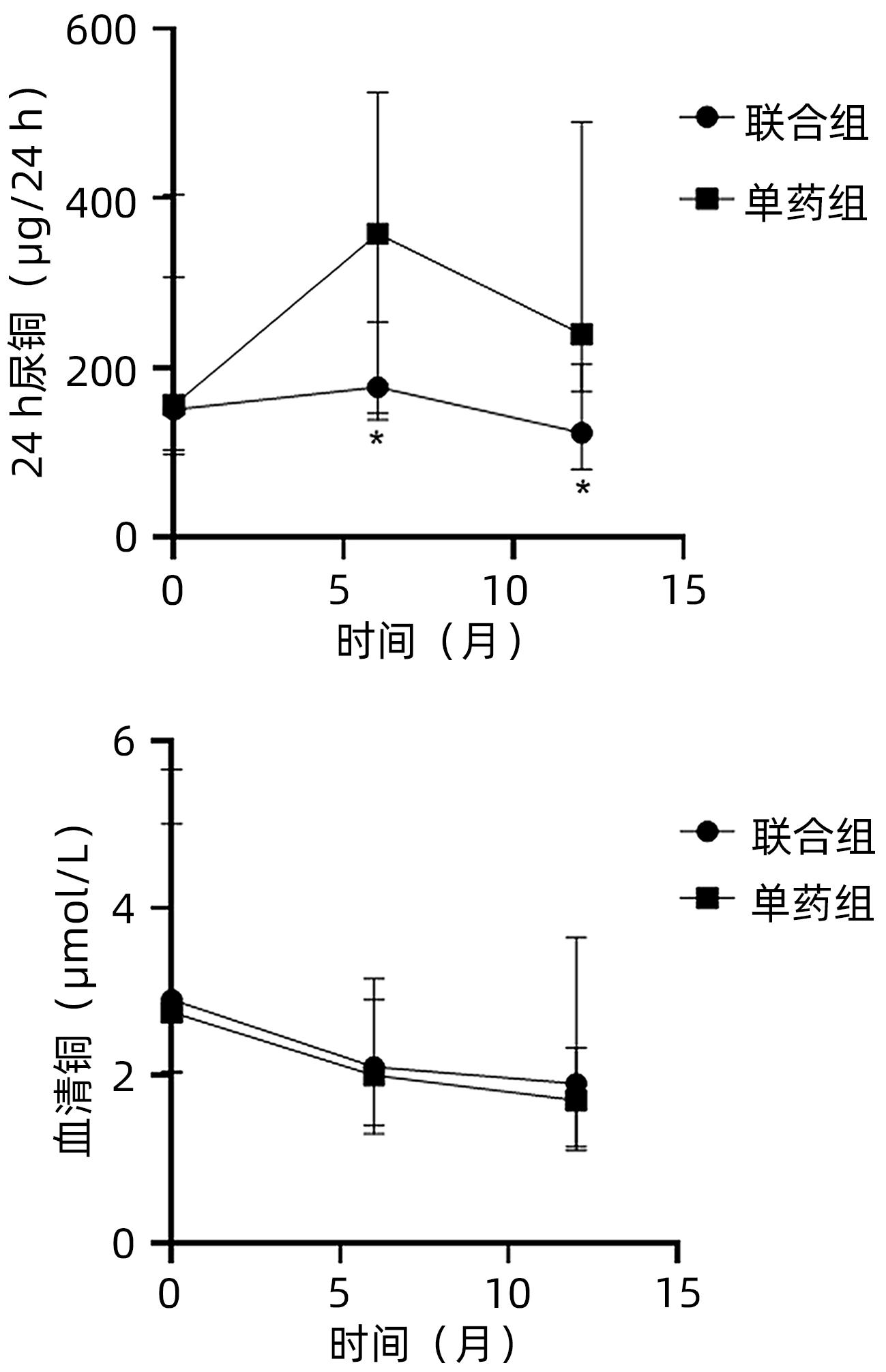
目的 比较二巯丁二酸联合硫酸锌与青霉胺单药治疗儿童肝豆状核变性12个月的疗效,为临床用药提供参考。 方法 纳入解放军总医院第五医学中心2018年5月—2021年5月住院治疗的111例2~18岁肝豆状核变性患儿,随机分配至二巯丁二酸联合硫酸锌治疗组(联合组,n=52)和青霉胺单药治疗组(单药组,n=59),比较两组患儿基线、治疗6个月及12个月时的肝功能实验室指标和24 h尿铜。计量资料两组间比较采用Mann-Whitney U检验,组内比较采用Wilcoxon符号秩和检验。计数资料两组间比较采用χ2检验或Fisher检验。 结果 治疗12个月时,联合组因药物不良反应脱落率为3.85%(2/52),低于单药组脱落率20.34%(12/59),差异有统计学意义(χ2=6.821,P=0.009)。治疗6个月和12个月时,联合组及单药组患儿肝功能ALT、AST水平均改善,与治疗前相比差异均有统计学意义(P值均<0.001),而治疗后两组间比较差异均无统计学意义(P值均>0.05)。治疗6个月时两组患儿24 h尿铜水平上升,12个月时下降,两组患儿分别下降至123.00(80.25~204.04) μg/24 h、239.50(171.50~490.25) μg/24 h,单药组24 h尿铜水平显著高于联合组(Z=-3.090,P=0.002)。治疗12个月时两组肝硬度值(LSM)≥7.3 kPa的患儿LSM均有下降,联合组患儿(n=22)LSM下降了1.8(0.3~7.2) kPa,单药组(n=13)患儿LSM下降了2.2(0.9~7.0) kPa,两组的LSM下降人数占比分别为81.82%、84.62%,差异无统计学意义(χ2=0.127,P=0.721)。 结论 二巯丁二酸联合硫酸锌方案对比青霉胺单药方案治疗儿童肝豆状核变性,尽管尿排铜量低,但二者在临床疗效方面无显著性差异,且前者安全性优于后者。 Abstract:Objective To investigate the effect of dimercaptosuccinic acid (DMSA) combined with zinc sulfate versus D-penicillamine (DPA) monotherapy in the treatment of children with Wilson’s Disease (WD), and to provide a reference for clinical medication. Methods A total of 111 children with WD, aged 2-18 years, who were admitted to the Fifth Medical Center of Chinese PLA General Hospital from May 2018 to May 2021 were enrolled in this study and were randomly divided into DMSA+zinc sulfate treatment group (combination group with 52 children) and DPA monotherapy group (monotherapy group with 59 children), and the two groups were compared in terms of liver function parameters and 24-hour urinary copper at baseline and 6 and 12 months of treatment. The Mann-Whitney U test was used for comparison of continuous data between two groups, and the Wilson rank sum test was used for comparison within each group; the chi-square test or the Fisher’s test was used for comparison of categorical data between two groups. Results At 12 months of treatment, the combination group had a significantly lower loss to follow-up rate due to adverse drug reactions than the monotherapy group [3.85% (2/52) vs 20.34% (12/59), χ2=6.821, P=0.009]. At 6 and 12 months of treatment, both the combination group and the monotherapy group had significant improvements in alanine aminotransferase and aspartate aminotransferase (all P<0.001), while there were no significant differences between the two groups after treatment (all P>0.05). Both groups had an increase in 24-hour urinary copper at 6 months of treatment and a reduction at 12 months of treatment, with a level of 123.00 (80.25-204.04) μg/24 h in the combination group and 239.50 (171.50-490.25) μg/24 h in the monotherapy group, and the monotherapy group had a significantly higher level of 24-hour urinary copper than the combination group (Z=-3.090, P=0.002). For both groups at 12 months of treatment, there was a reduction in LSM in the children with a liver stiffness measurement (LSM) of ≥7.3 kPa; LSM was reduced by 1.8 (0.3-7.2) kPa in the 22 children in the combination group and was reduced by 2.2 (0.9-7.0) kPa in the 13 children in the monotherapy group; the children with a reduction in LSM accounted for 81.82% and 84.62%, respectively, in the two groups, with no significant difference between the two groups (χ2=0.127, P=0.721). Conclusion In the treatment of WD, although DMSA combined with zinc sulfate has lower urinary copper excretion than DPA monotherapy, there is no significant difference in clinical efficacy between the two regimens, and DMSA combined with zinc sulfate has better safety than DPA monotherapy. -
Key words:
- Hepatolenticular Degeneration /
- Succimer /
- Zinc Sulfate /
- Penicillamine /
- Treatment Outcome
-
表 1 纳入病例基线特征
Table 1. Baseline characteristics of cases enrolled
项目 联合组(n=52) 单药组(n=59) 统计值 P值 年龄(岁) 9(6~13) 6(5~12) Z=-1.664 0.096 男/女(例) 26/26 37/22 χ2=1.820 0.177 ALT(U/L) 92(44~171) 98(42~203) Z=-0.485 0.628 AST(U/L) 89(52~131) 94(51~131) Z=-0.180 0.857 ChE(U/L) 4 858(2 853~6 432) 5 839(4 002~7 072) Z=-1.958 0.050 24 h尿铜(μg/24 h) 154.50(103.00~390.75) 159.00(98.70~437.60) Z=-0.206 0.837 血清铜(μmol/L) 2.90(2.05~5.65) 2.75(2.03~5.00) Z=-0.624 0.533 LSM(kPa) 8.10(5.20~17.90) 6.25(4.90~10.78) Z=-1.590 0.112 LSM≥7.3 kPa[例(%)] 13(25.00) 22(37.29) χ2=1.933 0.164 肝硬化[例(%)] 20(38.46) 19(32.20) χ2=0.475 0.491 铜蓝蛋白(g/L) 0.03(0.02~0.07) 0.03(0.02~0.03) Z=-0.956 0.339 角膜K-F环阳性[例(%)] 17(32.69) 15(25.42) χ2=0.986 0.611 表 2 两组治疗的不良反应
Table 2. Side effects of treatment in two groups
项目 联合组 (n=52) 单药组 (n=59) P值 尿微球蛋白(例) 1 3 0.703 粒细胞减少(例) 4 1 0.288 尿蛋白(例) 1 2 >0.05 发热(例) 0 1 >0.05 过敏、皮疹、丘疹(例) 0 3 0.288 尿潜血(例) 0 4 0.161 血小板减少(例) 0 1 >0.05 关节痛(例) 0 1 >0.05 口腔天疱疮(例) 0 1 >0.05 表 3 两组患儿治疗6个月及12个月时生化指标变化
Table 3. Changes of biochemical parameters at 6 and 12 months of treatment in two groups of children
项目 联合组(n=50) 单药组(n=47) Z值 P值 ALT(U/L) 基线 94(49~173) 100(62~212) -0.729 0.466 6个月 33(23~53) 33(20~47) -0.062 0.951 12个月 34(23~53) 30(21~53) -0.775 0.438 AST(U/L) 基线 85(51~132) 101(52~134) -0.567 0.571 6个月 45(31~61) 43(32~64) -0.070 0.944 12个月 39(29~50) 38(27~49) -0.142 0.887 ChE(U/L) 基线 4 858(2 853~6 432) 5 839(4 002~7 072) -1.854 0.064 6个月 5 286(3 927~6 607) 5 843(4 836~7 255) -1.745 0.081 12个月 5 694(4 344~7 396) 6 600(5 476~7 337) -1.363 0.173 注:排除退出试验患儿。 表 4 两组治疗后LSM变化
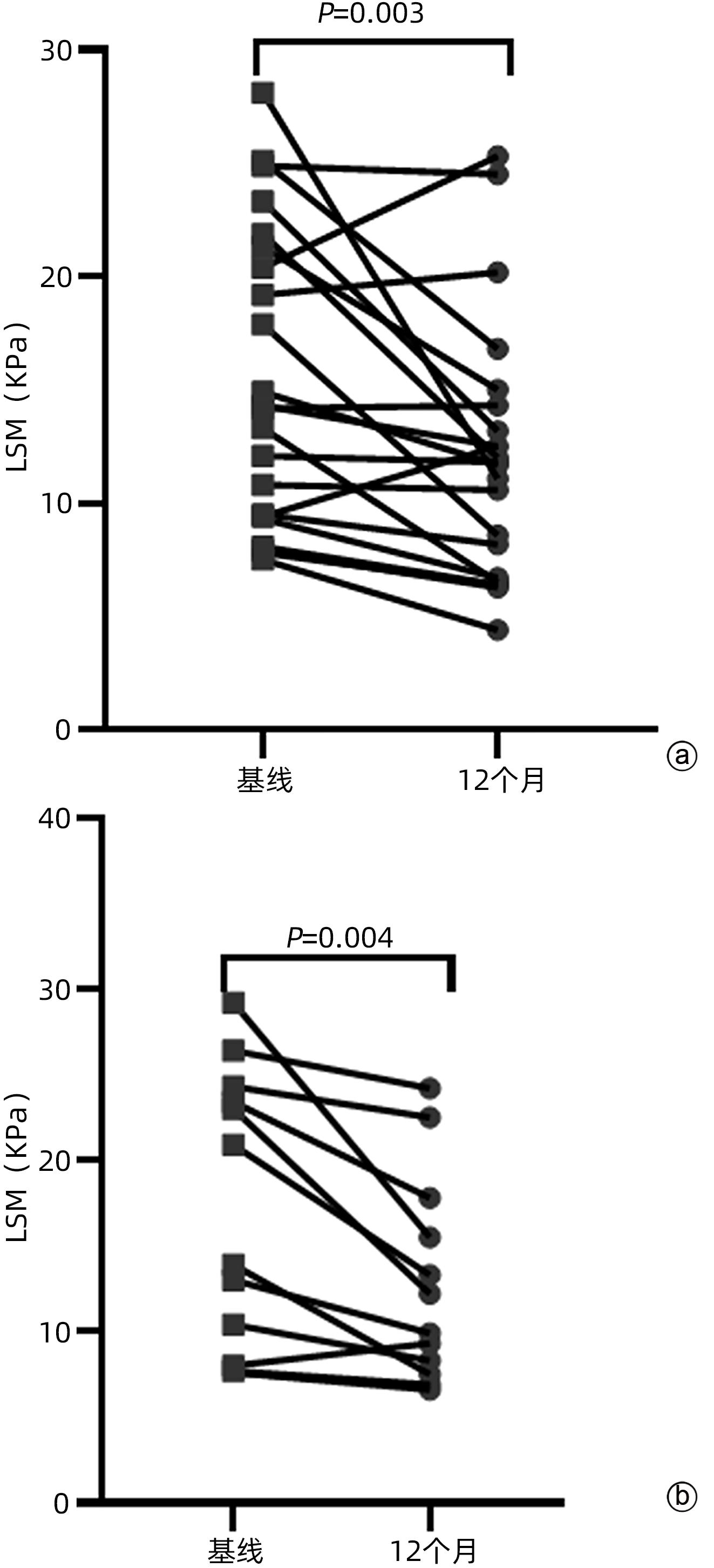
Table 4. The changes of LSM in two groups after treatment
项目 联合组(n=22) 单药组(n=13) 统计值 P值 基线LSM(kPa) 14.3(9.4~21.5) 13.9(7.9~23.9) Z=-0.137 0.891 治疗12个月LSM(kPa) 11.8(6.7~14.5) 9.9(7.2~16.7) Z=-0.307 0.759 下降差值(kPa) 1.8(0.3~7.2) 2.2(0.9~7.0) Z=-0.649 0.516 下降率(%) 81.82(18/22) 84.62(11/13) χ2=0.127 0.721 注:排除LSM<7.3 kPa及退出试验的患儿。 -
[1] Inherited Metabolic Liver Disease Collaboration Group, Chinese Society of Hepatology, Chinese Medical Association. Guidelines for the diagnosis and treatment of hepatolenticular degeneration(2022 edition)[J]. Chin J Hepatol, 2022, 30( 1): 9- 20. DOI: 10.3760/cma.j.cn501113-20211217-00603.中华医学会肝病学分会遗传代谢性肝病协作组. 肝豆状核变性诊疗指南(2022年版)[J]. 中华肝脏病杂志, 2022, 30( 1): 9- 20. DOI: 10.3760/cma.j.cn501113-20211217-00603. [2] REN MS, ZHANG Z, CAI YL, et al. Comparison of long-term therapeutic effects between succimer and penicillamine in hepatolenticular degeneration[J]. Chin J New Drugs Clin Remed, 2000, 19( 3): 166- 169. DOI: 10.3969/j.issn.1007-7669.2000.03.003.任明山, 张志, 蔡永亮, 等. 二巯丁二酸与青霉胺长期维持治疗肝豆状核变性疗效比较[J]. 中国新药与临床杂志, 2000, 19( 3): 166- 169. DOI: 10.3969/j.issn.1007-7669.2000.03.003. [3] KALITA J, KUMAR V, RANJAN A, et al. Role of oxidative stress in the worsening of neurologic Wilson disease following chelating therapy[J]. Neuromolecular Med, 2015, 17( 4): 364- 372. DOI: 10.1007/s12017-015-8364-8. [4] BREWER GJ, ASKARI F, DICK RB, et al. Treatment of Wilson’s disease with tetrathiomolybdate: V. Control of free copper by tetrathiomolybdate and a comparison with trientine[J]. Transl Res, 2009, 154( 2): 70- 77. DOI: 10.1016/j.trsl.2009.05.002. [5] LI WJ, WANG XP. The therapeutic drug for Wilson’s disease, dimercaptosuccinic acid(sodium)[J]. World Clin Drugs, 2012, 33( 9): 574- 576.李文杰, 王晓平. 肝豆状核变性治疗药物二巯丁二酸(钠)[J]. 世界临床药物, 2012, 33( 9): 574- 576. [6] KEMP W, LEVY M, WELTMAN M, et al. Australian Liver Association(ALA) expert consensus recommendations for the use of transient elastography in chronic viral hepatitis[J]. J Gastroenterol Hepatol, 2015, 30( 3): 453- 462. DOI: 10.1111/jgh.12865. [7] ZHOU SM, GUO LP, CAI WF, et al. Latest advances in the treatment of hepatolenticular degeneration[J]. J Clin Hepatol, 2020, 36( 1): 218- 221. DOI: 10.3969/j.issn.1001-5256.2020.01.052.周思敏, 郭丽萍, 蔡王锋, 等. 肝豆状核变性的治疗现状[J]. 临床肝胆病杂志, 2020, 36( 1): 218- 221. DOI: 10.3969/j.issn.1001-5256.2020.01.052. [8] XIE JJ, WU ZY. Wilson’s disease in China[J]. Neurosci Bull, 2017, 33( 3): 323- 330. DOI: 10.1007/s12264-017-0107-4. [9] LI XH. Medical treatment of Wilson’s disease[J]. J Intern Med Concepts Pract, 2021, 16( 5): 289- 293. DOI: 10.16138/j.1673-6087.2021.05.001.李洵桦. 肝豆状核变性的内科治疗[J]. 内科理论与实践, 2021, 16( 5): 289- 293. DOI: 10.16138/j.1673-6087.2021.05.001. [10] CHEN DW, ZHANG M. Advance in diagnostic and treatment of Wilson’s disease[J]. Infect Dis Info, 2019, 32( 2): 158- 161. DOI: 10.3969/j.issn.1007-8134.2019.02.016.陈大为, 张敏. 肝豆状核变性诊疗新进展[J]. 传染病信息, 2019, 32( 2): 158- 161. DOI: 10.3969/j.issn.1007-8134.2019.02.016. [11] BREWER GJ, DICK RD, JOHNSON VD, et al. Treatment of Wilson’s disease with zinc: XV long-term follow-up studies[J]. J Lab Clin Med, 1998, 132( 4): 264- 278. DOI: 10.1016/s0022-2143(98)90039-7. [12] Chinese Foundation for Hepatitis Prevention and Control; Chinese Society of Infectious Disease and Chinese Society of Hepatology, Chinese Medical Association; Liver Disease Committee of Chinese Research Hospital Association. Consensus on clinical application of transient elastography detecting liver fibrosis: a 2018 update[J]. Chin J Hepatol, 2019, 27( 3): 182- 191. DOI: 10.3760/cma.j.issn.1007-3418.2019.03.004.中国肝炎防治基金会, 中华医学会感染病学分会, 中华医学会肝病学分会和中国研究型医院学会肝病专业委员会. 瞬时弹性成像技术诊断肝纤维化专家共识(2018年更新版)[J]. 中华肝脏病杂志, 2019, 27( 3): 182- 191. DOI: 10.3760/cma.j.issn.1007-3418.2019.03.004. [13] ZHOU XX, PU XY, XIAO X, et al. Observation on the changes of clinical symptoms, blood and brain copper deposition in Wilson disease patients treated with dimercaptosuccinic acid for 2 years[J]. J Clin Neurosci, 2020, 81: 448- 454. DOI: 10.1016/j.jocn.2020.09.017. -



 PDF下载 ( 761 KB)
PDF下载 ( 761 KB)

 下载:
下载: