三黄泻心汤对肝性脑病大鼠脑组织炎性损伤的改善作用及机制
DOI: 10.3969/j.issn.1001-5256.2023.09.015
Role and mechanism of action of Sanhuang Xiexin Decoction in improving inflammatory injury of brain tissue in rats with hepatic encephalopathy
-
摘要:
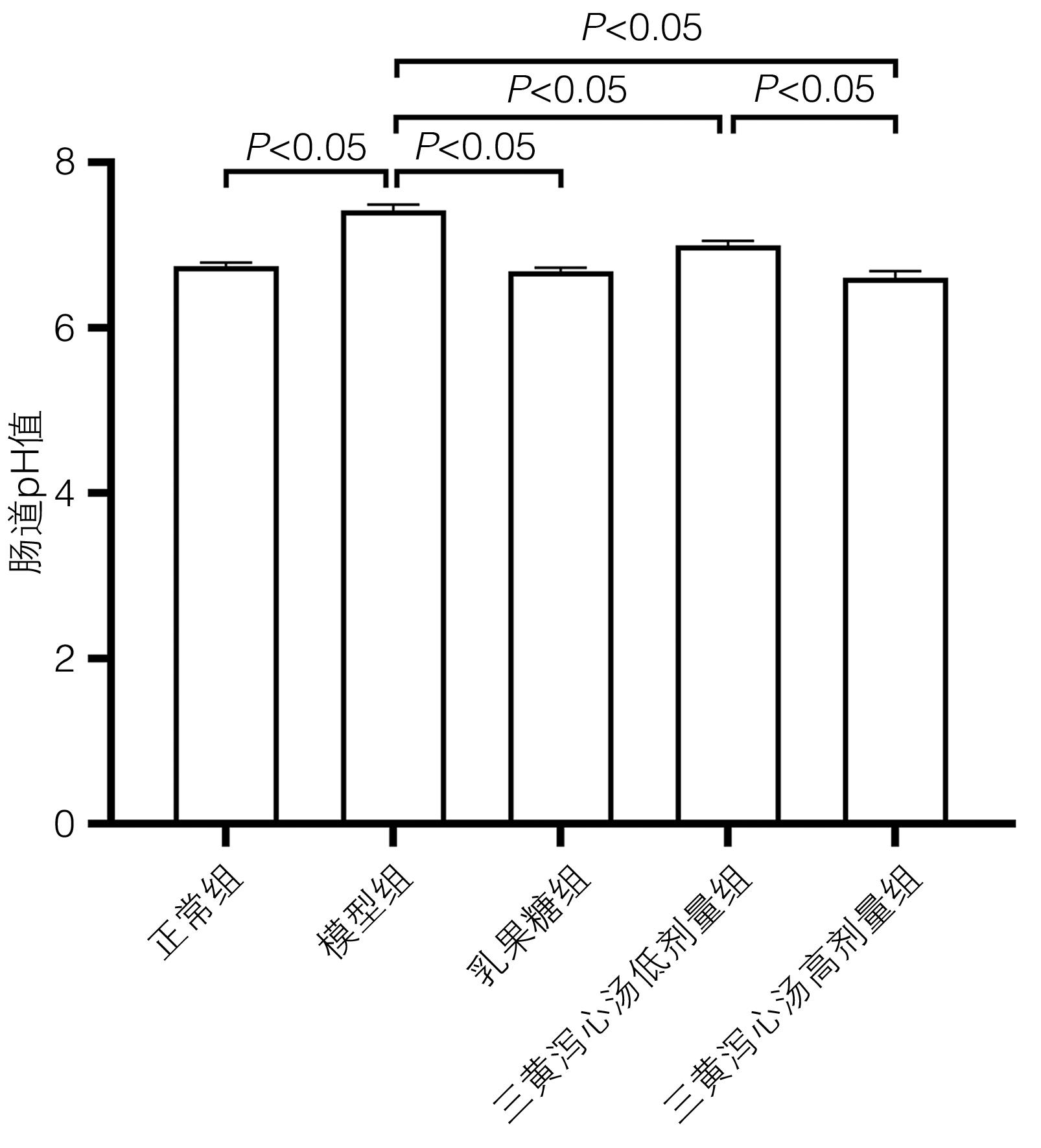
目的 通过建立肝性脑病大鼠模型,研究三黄泻心汤对肝性脑病大鼠脑组织炎性损伤的改善作用及机制。 方法 将30只SD雄性大鼠随机分为正常组、模型组、乳果糖组(0.167 g/kg)、三黄泻心汤低剂量组(生药4.37 g/kg)和三黄泻心汤高剂量组(生药8.74 g/kg),每组各6只。通过40%四氯化碳油溶液皮下注射构建肝硬化模型(持续9周,每周2次,首周每次注射5 mL/kg,而后每次注射3 mL/kg),之后腹腔注射硫代乙酰胺(连续3 d,1次/d,每次注射300 mg/kg)构建肝性脑病模型。造模后各治疗组给予对应剂量药物灌胃1周。在造模及给药后分别进行Morris水迷宫测试大鼠学习记忆能力。之后处死大鼠,取血液、肝脏、结肠,于冰上取脑组织。HE染色后观察各组大鼠肝脏、脑组织病理切片,检测各组大鼠肝功能、肠道pH值、血氨水平;酶联免疫吸附测定法检测各组大鼠血清中TNF-α、IL-1β、IL-6水平;计算脑组织含水量;实时荧光定量聚合酶链式反应检测TNF-α、IL-1β、IL-6 mRNA表达,蛋白免疫印迹法检测NF-κB p65蛋白亚基及其磷酸化表达。计量资料多组间比较采用单因素方差分析,进一步两两比较采用LSD-t检验或Dunnett’s T3法。 结果 与模型组相比,三黄泻心汤低、高剂量组肝性脑病大鼠肝细胞、脑细胞变性坏死情况均减轻,肝功能改善(P值均<0.05),血氨及血清中TNF-α、IL-1β、IL-6水平均明显降低(P值均<0.05),肠道pH值趋于正常(P值均<0.05),Morris水迷宫测试寻台潜伏期、穿台次数结果均显著改善(P值均<0.05),脑组织含水量降低(P值均<0.05),TNF-α、IL-1β、IL-6 mRNA表达均降低(P值均<0.05),NF-κB p65及其磷酸化蛋白表达受抑制(P值均<0.05),且高剂量组治疗效果均优于低剂量组(P值均<0.05)。 结论 三黄泻心汤可通过有效抑制肝性脑病大鼠脑部炎性因子转录水平及NF-κB p65磷酸化来改善大鼠的脑组织炎性损伤。 -
关键词:
- 肝性脑病 /
- 泻心汤 /
- 治疗结果 /
- 大鼠, Sprague-Dawley
Abstract:Objective To investigate the role and mechanism of action of Sanhuang Xiexin Decoction in improving inflammatory injury of brain tissue in rats with hepatic encephalopathy (HE) by establishing a rat model of HE. Methods A total of 30 Sprague-Dawley rats were randomly divided into normal group, model group, lactulose group (0.167 g/kg), low-dose Sanhuang Xiexin Decoction group (4.37 g/kg crude drug), and high-dose Sanhuang Xiexin Decoction group (8.74 g/kg crude drug), with 6 rats in each group. The rats were given subcutaneous injection of 40% carbon tetrachloride oil solution (twice a week for 9 weeks, at a dose of 5 mL/kg each time in the first week, followed by 3 mL/kg each time) to establish a model of liver cirrhosis, and then they were given intraperitoneal injection of thioacetamide (300 mg/kg, once a day for 3 consecutive days) to establish a model of HE. After modeling, each treatment group was given the corresponding drug by gavage for 1 week. Morris water maze was used to observe the learning and memory abilities of rats after modeling and administration, and then the rats were sacrificed to collect blood, liver, and colon samples, with brain tissue collected on ice. HE staining was used to observe the pathological sections of liver and brain tissue; liver function, intestinal pH value, and blood ammonia were measured for all rats; ELISA was used to measure the serum levels of tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), and interleukin-6 (IL-6); the water content of brain tissue was calculated; quantitative real-time PCR was used to measure the mRNA expression levels of TNF-α, IL-1β, and IL-6, and Western blotting was used to measure the expression levels of nuclear factor-kappa B (NF-κB) p65 and phosphorylated NF-κB p65. A one-way analysis of variance was used for comparison of continuous data between multiple groups, and the least significant difference t-test or the Dunnett’s T3 method was used for further comparison between two groups. Results Compared with the model group, the low-and high-dose Sanhuang Xiexin Decoction groups had significant alleviation of the degeneration and necrosis of hepatocytes and brain cells and improvement in liver function in rats with HE (all P<0.05), significant reductions in blood ammonia and the serum levels of TNF-α, IL-1β, and IL-6 (all P<0.05), a relatively normal intestinal pH value (all P<0.05), significant improvements in searching platform latency and number of platform crossings in the Morris water maze test (all P<0.05), significant reductions in the water content of brain tissue and the mRNA expression levels of TNF-α, IL-1β, and IL-6 (all P<0.05), and significantly inhibited expression levels of NF-κB p65 and its phosphorylated protein (all P<0.05), and high-dose Sanhuang Xiexin Decoction tended to have a significantly better effect than low-dose Sanhuang Xiexin Decoction (all P<0.05). Conclusion Sanhuang Xiexin Decoction can improve inflammatory injury of brain tissue in HE rats by effectively inhibiting the transcriptional level of inflammatory factors and NF-κB p65 phosphorylation in the brain of HE rats. -
Key words:
- Hepatic Encephalopathy /
- Xie Xin Tang /
- Treatment Outcome /
- Rats, Sprague-Dawley
-
表 1 RT-qPCR引物序列
Table 1. RT-qPCR primer sequences
基因 上游(5′-3′) 下游(5′-3′) TNF-α GTCCGGGCAGGTCTACTTTG GGCTGGGTAGAGAACGGATG IL-1β CCTATGTCTTGCCCGTGGAG CACACACTAGCAGGTCGTCA IL-6 GCCCACCAGGAACGAAAGTC TGGCTGGAAGTCTCTTGCG GADPH CTCTCTGCTCCTCCCTGTTC TCACACCGACCTTCACCATC 表 2 三黄泻心汤对肝性脑病大鼠血氨及肝功能的影响
Table 2. Effect of Sanhuang Xiexin Decoction on blood ammonia and liver function in rats with hepatic encephalopathy
组别 动物数(只) 血氨(mmol/L) ALT(U/L) AST(U/L) 正常组 6 58.25±6.34 56.27±9.04 144.43±13.72 模型组 6 228.20±8.741) 1 787.02±86.091) 2 452.20±104.221) 乳果糖组 6 133.93±8.071)2) 539.37±38.741)2) 660.52±34.821)2) 三黄泻心汤低剂量组 6 169.53±7.771)2) 826.92±34.151)2) 850.82±32.171)2) 三黄泻心汤高剂量组 6 129.55±6.871)2)3) 541.65±35.771)2)3) 667.57±40.781)2)3) F值 399.12 1 082.11 1 543.34 P值 <0.001 <0.001 <0.001 注:与正常组比较,1)P<0.05;与模型组比较,2)P<0.05;与三黄泻心汤低剂量组比较,3)P<0.05。 Table . Effect of Sanhuang Xiexin Decoction on serum TNF-α、IL-1β、IL-6 content in rats with hepatic encephalopathy
组别 动物数(只) TNF-α(pg/mL) IL-1β(pg/mL) IL-6(pg/mL) 正常组 6 35.21±1.43 38.30±5.41 49.89±9.34 模型组 6 113.57±5.001) 231.97±16.751) 204.46±22.261) 乳果糖组 6 67.02±5.511)2) 113.59±11.481)2) 100.60±11.241)2) 三黄泻心汤低剂量组 6 85.36±4.341)2) 154.37±13.161)2) 144.71±15.121)2) 三黄泻心汤高剂量组 6 65.90±4.451)2)3) 113.79±11.901)2)3) 101.23±12.811)2)3) F值 258.70 197.69 91.14 P值 <0.001 <0.001 <0.001 注:与正常组比较,1)P<0.05;与模型组比较,2)P<0.05;与三黄泻心汤低剂量组比较,3)P<0.05。 表 4 三黄泻心汤对肝性脑病大鼠学习记忆能力的影响
Table 4. Effect of Sanhuang Xiexin Decoction on learning and memory ability of rats with hepatic encephalopathy
组别 动物数(只) 造模后寻台潜伏期(s) 用药后寻台潜伏期(s) 穿台次数(次) 正常组 6 16.33±0.50 15.83±0.93 10.17±1.72 模型组 6 53.58±5.511) 58.28±1.601) 2.83±1.171) 乳果糖组 6 56.48±4.231)2) 31.12±1.331)2) 8.00±0.891)2) 三黄泻心汤低剂量组 6 56.07±3.531)2) 45.48±6.151)2) 5.83±1.171)2) 三黄泻心汤高剂量组 6 56.33±2.481)2)3) 31.02±0.771)2)3) 7.83±0.751)2)3) F值 138.61 179.34 32.29 P值 <0.001 <0.001 <0.001 注:与正常组比较,1)P<0.05;与模型组比较,2)P<0.05;与三黄泻心汤低剂量组比较,3)P<0.05。 Table . Effect of Sanhuang Xiexin Decoction on brain mass and brain tissue water content in rats
组别 动物数(只) 脑质量(g) 脑组织含水量(%) 正常组 6 2.29±0.05 75.96±0.64 模型组 6 2.40±0.011) 80.78±0.501) 乳果糖组 6 2.32±0.011)2) 77.07±0.471)2) 三黄泻心汤低剂量组 6 2.38±0.011)2) 79.12±0.461)2) 三黄泻心汤高剂量组 6 2.32±0.011)2)3) 77.14±0.601)2)3) F值 17.30 75.42 P值 <0.001 <0.001 注:与正常组比较,1)P<0.05;与模型组比较,2)P<0.05;与三黄泻心汤低剂量组比较,3)P<0.05。 表 6 三黄泻心汤对肝性脑病大鼠脑组织中TNF-α、IL-1β、IL-6 mRNA表达的影响
Table 6. Effect of Sanhuang Xiexin Decoction on mRNA expression of TNF-α、IL-1 β、 IL-6 on brain tissue of rats with hepatic encephalopathy
组别 动物数(只) TNF-α mRNA IL-1 mRNA IL-6 mRNA 正常组 6 1.01±0.16 1.02±0.21 1.01±0.13 模型组 6 2.29±0.241) 2.18±0.231) 2.00±0.201) 乳果糖组 6 1.38±0.171)2) 1.38±0.171)2) 1.27±0.151)2) 三黄泻心汤低剂量组 6 1.79±0.201)2) 1.80±0.211)2) 1.69±0.191)2) 三黄泻心汤高剂量组 6 1.54±0.171)2)3) 1.50±0.171)2)3) 1.46±0.171)2)3) F值 37.72 29.11 29.88 P值 <0.001 <0.001 <0.001 注:与正常组比较,1)P<0.05;与模型组比较,2)P<0.05;与三黄泻心汤低剂量组比较,3)P<0.05。 -
[1] ROSE CF, AMODIO P, BAJAJ JS, et al. Hepatic encephalopathy: Novel insights into classification, pathophysiology and therapy[J]. J Hepatol, 2020, 73( 6): 1526- 1547. DOI: 10.1016/j.jhep.2020.07.013. [2] SAID VJ, GARCIA-TRUJILLO E. Beyond lactulose: Treatment options for hepatic encephalopathy[J]. Gastroenterol Nurs, 2019, 42( 3): 277- 285. DOI: 10.1097/SGA.0000000000000376. [3] YAO JQ, XIAO YQ, LIU Y, et al. Anti-inflammatory effect comparison of water and ethanol extracts of Sanhuang Xiexin Tang and component analysis of its active extracts[J]. Chin J Exp Med Formul, 2015, 21( 13): 31- 35. DOI: 10.13422/j.cnki.syfjx.2015130031.姚佳琪, 肖永庆, 刘颖, 等. 三黄泻心汤水、醇提取物抗炎作用比较及有效提取物的成分分析[J]. 中国实验方剂学杂志, 2015, 21( 13): 31- 35. DOI: 10.13422/j.cnki.syfjx.2015130031. [4] WANG YX, LIU WW, LI XP, et al. Determination of activity of Sanhuang Xiexin decoction in inhibiting drug-resistant Staphylococcus aureus in vitro[J]. J Pract Tradit Chin Med, 2018, 34( 4): 500- 501. DOI: 10.3969/j.issn.1004-2814.2018.04.096.王钰雄, 刘伟伟, 李雪萍, 等. 三黄泻心汤体外抑制耐药金黄色葡萄球菌活性测定[J]. 实用中医药杂志, 2018, 34( 4): 500- 501. DOI: 10.3969/j.issn.1004-2814.2018.04.096. [5] YUE LJ, LIU C, ZHANG HJ, et al. Clinical effect of Sanhuang Xiexin decoction on patients with upper gastrointestinal hemorrhage and its influence on hemoglobin, urea nitrogen and Rockall score[J]. Shaanxi J Tradit Chin Med, 2021, 42( 5): 594- 596, 600. DOI: 10.3969/j.issn.1000-7369.2021.05.012.岳丽军, 刘畅, 张华军, 等. 三黄泻心汤治疗上消化道出血疗效及对患者血红蛋白、尿素氮、Rockall评分的影响[J]. 陕西中医, 2021, 42( 5): 594- 596, 600. DOI: 10.3969/j.issn.1000-7369.2021.05.012. [6] ZHOU XX, ZENG SL, YAN HP, et al. Retention enema therapy with rhubarb decoction in the adjuvant treatment of hepatic encephalopathy[J]. J Guangxi Univ Chin Med, 2022, 25( 1): 12- 16. DOI: 10.3969/j.issn.2095-4441.2022.01.003.周学寻, 曾胜澜, 严惠萍, 等. 大黄煎剂保留灌肠辅助治疗肝性脑病的临床观察[J]. 广西中医药大学学报, 2022, 25( 1): 12- 16. DOI: 10.3969/j.issn.2095-4441.2022.01.003. [7] LIN WJ, LI J, GUO FF, et al. Establishment of rat liver cirrhosis model[J]. Lingnan J Emerg Med, 2017, 22( 4): 310- 312. DOI: 10.3969/j.issn.1671-301X.2017.04.002.林文健, 李婕, 郭芬芬, 等. 大鼠肝硬化模型建立探讨[J]. 岭南急诊医学杂志, 2017, 22( 4): 310- 312. DOI: 10.3969/j.issn.1671-301X.2017.04.002. [8] LIU MH, LI K, ZHENG HJ. Effect of Angong Niuhuang Pill on TNF-α/NF-κB signal pathway and neurological function in rats with hepatic encephalopathy[J]. Drugs& Clinic, 2021, 36( 11): 2217- 2223. DOI: 10.7501/j.issn.1674-5515.2021.11.001.刘敏环, 李剀, 郑慧军. 安宫牛黄丸对肝性脑病大鼠TNF-α/NF-κB信号通路及神经功能的影响[J]. 现代药物与临床, 2021, 36( 11): 2217- 2223. DOI: 10.7501/j.issn.1674-5515.2021.11.001. [9] Chinese Society of Hepatology, Chinese Medical Association. Chinese guidelines on the management of liver cirrhosis[J]. J Clin Hepatol, 2019, 35( 11): 2408- 2425. DOI: 10.3969/j.issn.1001-5256.2019.11.006.中华医学会肝病学分会. 肝硬化诊治指南[J]. 临床肝胆病杂志, 2019, 35( 11): 2408- 2425. DOI: 10.3969/j.issn.1001-5256.2019.11.006. [10] LE YY, ZHANG RZ, XIAO WS, et al. Research progress in pathogenesis of hepatic encephalopathy[J]. Chin J Integr Tradit West Med Liver Dis, 2022, 32( 5): 468- 472. DOI: 10.3969/j.issn.1005-0264.2022.05.023.乐滢玉, 张荣臻, 肖伟松, 等. 肝性脑病发病机制的研究进展[J]. 中西医结合肝病杂志, 2022, 32( 5): 468- 472. DOI: 10.3969/j.issn.1005-0264.2022.05.023. [11] WALKER V. Ammonia metabolism and hyperammonemic disorders[J]. Adv Clin Chem, 2014, 67: 73- 150. DOI: 10.1016/bs.acc.2014.09.002. [12] SEPEHRINEZHAD A, ZARIFKAR A, NAMVAR G, et al. Astrocyte swelling in hepatic encephalopathy: molecular perspective of cytotoxic edema[J]. Metab Brain Dis, 2020, 35( 4): 559- 578. DOI: 10.1007/s11011-020-00549-8. [13] RAMA RAO KV, JAYAKUMAR AR, TONG X, et al. Marked potentiation of cell swelling by cytokines in ammonia-sensitized cultured astrocytes[J]. J Neuroinflammation, 2010, 7: 66. DOI: 10.1186/1742-2094-7-66. [14] JAYAKUMAR AR, RAMA RAO KV, NORENBERG MD. Neuroinflammation in hepatic encephalopathy: mechanistic aspects[J]. J Clin Exp Hepatol, 2015, 5( Suppl 1): S21-S28. DOI: 10.1016/j.jceh.2014.07.006. [15] LU L, WU C, LU BJ, et al. BabaoDan cures hepatic encephalopathy by decreasing ammonia levels and alleviating inflammation in rats[J]. J Ethnopharmacol, 2020, 249: 112301. DOI: 10.1016/j.jep.2019.112301. [16] YANG C, TAN Y, LI F, et al. Intestinal microecology of mice exposed to TiO2 nanoparticles and bisphenol A[J]. Foods, 2022, 11( 12): 1696. DOI: 10.3390/foods11121696. [17] RAI R, SARASWAT VA, DHIMAN RK. Gut microbiota: its role in hepatic encephalopathy[J]. J Clin Exp Hepatol, 2015, 5( Suppl 1): S29-S36. DOI: 10.1016/j.jceh.2014.12.003. [18] AHLUWALIA V, BETRAPALLY NS, HYLEMON PB, et al. Impaired gut-liver-brain axis in patients with cirrhosis[J]. Sci Rep, 2016, 6: 26800. DOI: 10.1038/srep26800. [19] ZHOU Z, LI K, GUO J, et al. Green tea catechin EGCG ameliorates thioacetamide-induced hepatic encephalopathy in rats via modulation of the microbiota-gut-liver axis[J]. Mol Nutr Food Res, 2022: e2200821. DOI: 10.1002/mnfr.202200821. [20] JIANG HN, MAO DW, YE QL, et al. Research progress in treatment of hepatic encephalopathy based on syndrome differentiation[J]. Shaanxi J Tradit Chin Med, 2020, 41( 11): 1678- 1680. DOI: 10.3969/j.issn.1000-7369.2020.11.042.蒋海南, 毛德文, 叶倩伶, 等. 中医辨证论治肝性脑病研究进展[J]. 陕西中医, 2020, 41( 11): 1678- 1680. DOI: 10.3969/j.issn.1000-7369.2020.11.042. [21] FU DQ, XU L, GE EN, et al. Research of effective component dissolution rules of Sanhuang Xiexin Decoction[J]. Chin Arch Tradit Chin Med, 2020, 38( 10): 16- 19. DOI: 10.13193/j.issn.1673-7717.2020.10.004.傅丹青, 徐莉, 葛尔宁, 等. 三黄泻心汤主要有效成分溶出规律研究[J]. 中华中医药学刊, 2020, 38( 10): 16- 19. DOI: 10.13193/j.issn.1673-7717.2020.10.004. [22] LI L, WANG Y, ZHAO L, et al. Sanhuang xiexin decoction ameliorates secondary liver injury in DSS-induced colitis involve regulating inflammation and bile acid metabolism[J]. J Ethnopharmacol, 2022, 299: 115682. DOI: 10.1016/j.jep.2022.115682. [23] MA BL, MA YM, YAN DM, et al. Effective constituents in Xiexin Decoction for anti-inflammation[J]. J Ethnopharmacol, 2009, 125( 1): 151- 156. DOI: 10.1016/j.jep.2009.05.035. [24] MANZHALII E, VIRCHENKO O, FALALYEYEVA T, et al. Hepatic encephalopathy aggravated by systemic inflammation[J]. Dig Dis, 2019, 37( 6): 509- 517. DOI: 10.1159/000500717. [25] ZHANG Y, CHEN W, MENG XL. The protective effect of Sanhuangxiexin Decoction against oxidative stress and inflammation injury of global cerebral ischemia reperfusion rats[J]. Pharmocol Clin Chin Mater Med, 2014, 30( 4): 1- 5. DOI: 10.13412/j.cnki.zyyl.2014.04.001.张祎, 陈文, 孟宪丽. 三黄泻心汤对全脑缺血再灌注大鼠氧化应激及炎性损伤的保护作用[J]. 中药药理与临床, 2014, 30( 4): 1- 5. DOI: 10.13412/j.cnki.zyyl.2014.04.001. -



 PDF下载 ( 1723 KB)
PDF下载 ( 1723 KB)

 下载:
下载:





