干扰素基因刺激蛋白(STING)通路对乙型肝炎肝硬化患者外周血单核细胞炎症因子分泌及吞噬功能的影响
DOI: 10.3969/j.issn.1001-5256.2023.09.013
Effect of the STING pathway on inflammatory cytokine secretion and phagocytosis by peripheral blood monocytes in patients with hepatitis B cirrhosis
-
摘要:
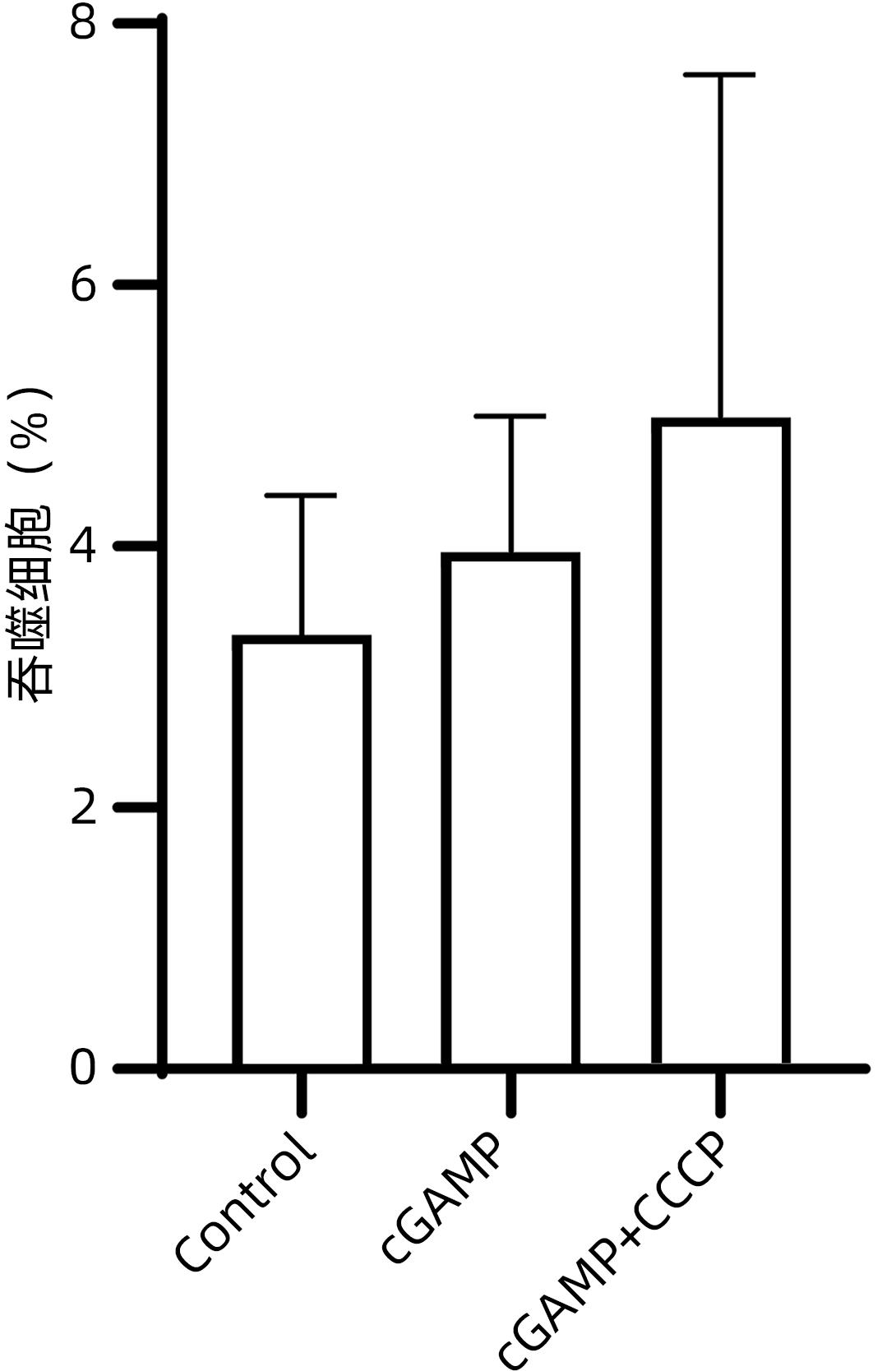
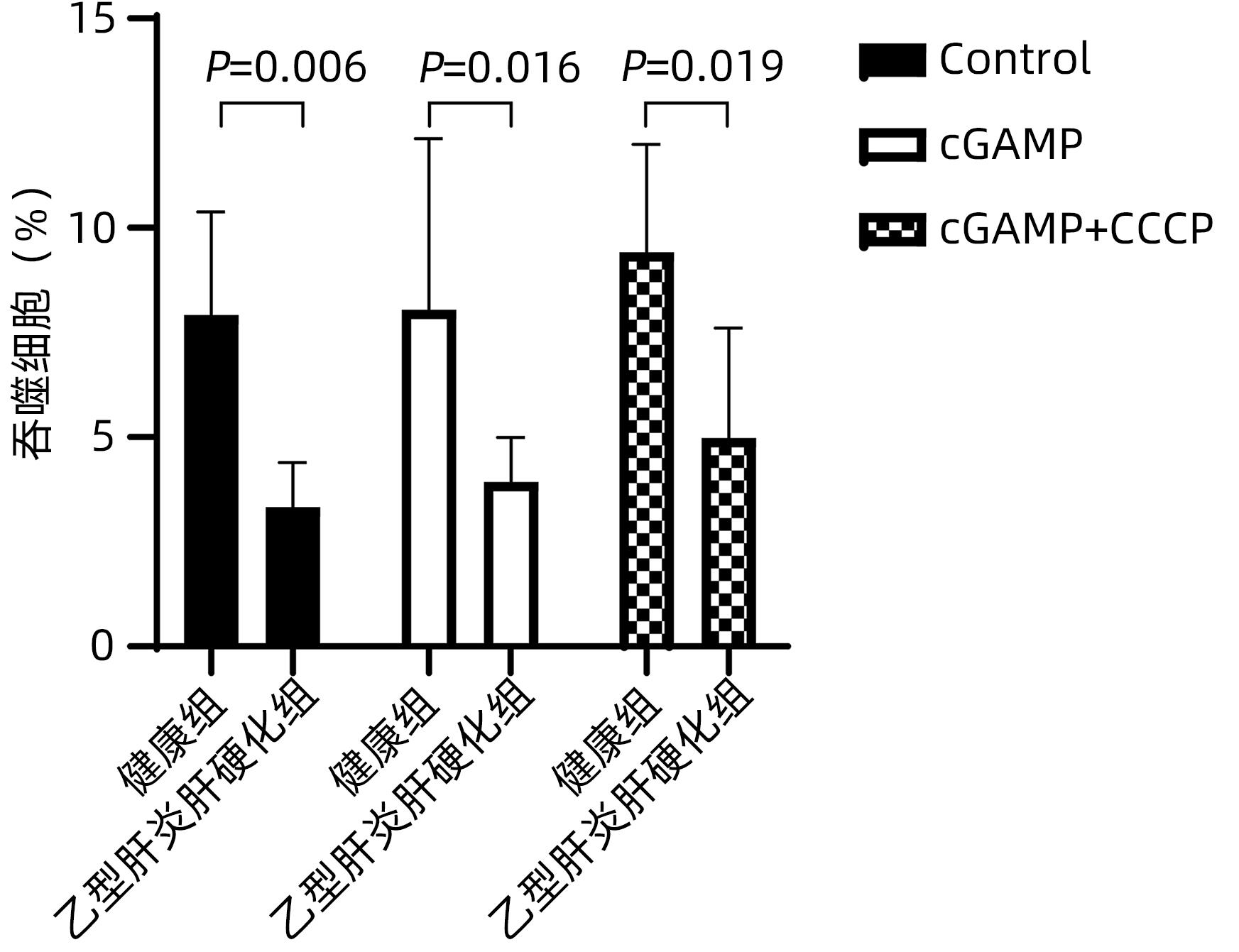
目的 研究干扰素基因刺激蛋白(STING)通路对乙型肝炎肝硬化患者外周血单核细胞生成炎症细胞因子及吞噬功能的影响。 方法 收集2020年5月—10月于徐州医科大学附属医院感染性疾病科住院的35例乙型肝炎肝硬化患者和10例体检中心健康体检者的外周静脉全血,分离并提取外周血单核细胞;另将乙型肝炎肝硬化患者分为环鸟苷酸-腺苷酸(cGAMP)(n=12)、cGAMP+CCCP(n=12)和Control(n=11)三组。体外以STING激活剂cGAMP和/或STING抑制剂CCCP加入单核细胞培养液中,ELISA法检测上清液IFN-α、IFN-β、IL-6和TNF-α水平。此外,将含cGAMP和/或CCCP的单核细胞培养体系与荧光标记的E.coli共同孵育后,采用流式细胞仪检测单核细胞吞噬功能的改变。计量资料两组间比较采用成组t检验;多组间比较采用单因素方差分析,进一步两两比较采用LSD-t检验。 结果 与加入抑制剂CCCP组相比,乙型肝炎肝硬化患者外周血单核细胞经cGAMP刺激后分泌IFN-α、IFN-β、IL-6和TNF-α水平显著升高(P值均<0.05)。乙型肝炎肝硬化患者外周血单核细胞在有或无cGAMP/cGAMP+CCCP处理下,吞噬E.coli能力均较健康组明显降低(t值分别为4.647、2.790、2.504,P值均<0.05),且有或无cGAMP与cGAMP+CCCP刺激,对乙型肝炎肝硬化患者单核细胞吞噬E.coli的能力没有显著影响(P>0.05)。 结论 外周血单核细胞STING通路活化参与了乙型肝炎肝硬化状态下全身性炎症反应的发生,STING通路的激活与否不影响乙型肝炎肝硬化外周血单核细胞的吞噬细菌能力。 Abstract:Objective To investigate the effect of the STING pathway on the inflammatory cytokine secretion and phagocytosis by peripheral blood monocytes in patients with hepatitis B cirrhosis. Methods Peripheral venous whole blood samples were collected from 35 patients with hepatitis B cirrhosis who were hospitalized in department of infectious diseases and 10 individuals who underwent physical examination in physical examination center in the Affiliated Hospital of Xuzhou Medical University from May to October 2020, and peripheral blood monocytes were isolated and extracted. Patients with hepatitis B cirrhosis were divided into three groups: cyclic GMP-AMP (cGAMP) (n=12), cGAMP+CCCP (n=12) and Control (n=11). In vitro, the STING activator cGAMP and/or the STING inhibitor CCCP were added to monocyte culture medium, and ELISA was used to measure the levels of interferon-α (IFN-α), interferon-β (IFN-β), interleukin-6 (IL-6), and tumor necrosis factor-α (TNF-α) in supernatant. In addition, the monocyte culture system containing cGAMP and/or CCCP was co-cultured with fluorescein-labeled Escherichia coli, and then flow cytometry was used to observe changes in the phagocytosis of monocytes. The independent-samples t test was used for comparison of continuous data between two groups; a one-way analysis of variance was used for comparison between multiple groups, and the least significant difference t-test was used for further comparison between two groups. Results Compared with the peripheral blood monocytes treated by the inhibitor CCCP, the peripheral blood monocytes stimulated by cGAMP secreted significantly higher levels of IFN-α, IFN-β, IL-6, and TNF-α (all P<0.05). Compared with the healthy group, the hepatitis B cirrhosis group had a significant reduction in the phagocytosis of E. coli by peripheral blood monocytes, with or without cGAMP/cGAMP+CCCP treatment (t=4.647, 2.790, and 2.504, all P<0.05), and cGAMP or cGAMP+CCCP stimulation had no significant effect on the phagocytosis of E. coli by peripheral blood monocytes in the patients with hepatitis B cirrhosis (P>0.05). Conclusion The activation of the STING pathway in peripheral blood monocytes is involved in the development of systemic inflammatory response in hepatitis B cirrhosis, and the activation of the STING pathway has no significant effect on the phagocytosis of bacteria by peripheral blood monocytes in hepatitis B cirrhosis. -
Key words:
- Liver Cirrhosis /
- Cytokines /
- Stimulator of Interferon Genes
-
表 1 cGAMP作用下乙型肝炎肝硬化患者和健康成人外周血单核细胞炎症因子分泌水平比较
Table 1. Comparison of inflammatory factor secretion levels in peripheral blood mononuclear cells of patients with hepatitis B cirrhosis and healthy adults under the effect of cGAMP
组别 例数 IFN-α (pg/mL) IFN-β (pg/mL) IL-6 (pg/mL) TNF-α (pg/mL) 乙型肝炎肝硬化组 35 31.31 ± 3.02 908.03 ± 109.98 47.02 ± 3.89 56.32 ± 4.92 健康组 10 21.69 ± 1.41 690.35 ± 52.90 41.07 ± 2.04 44.44 ± 4.11 t值 7.315 4.475 3.467 5.072 P值 <0.001 <0.001 0.003 <0.001 表 2 cGAMP作用下乙型肝炎肝硬化患者外周血单核细胞炎症因子分泌水平
Table 2. Inflammatory factor secretion levels in peripheral blood mononuclear cells of patients with hepatitis B cirrhosis under the influence of cGAMP
组别 例数 IFN-α (pg/mL) IFN-β (pg/mL) IL-6 (pg/mL) TNF-α (pg/mL) cGAMP组 12 31.31 ± 3.02 908.03 ± 109.98 47.02 ± 3.89 56.32 ± 4.92 cGAMP+CCCP组 12 21.13 ± 2.471) 714.60 ± 151.261) 35.29 ± 4.121) 30.47 ± 6.891) Control组 11 24.46 ± 2.551) 597.50 ± 99.561) 37.72 ± 4.281) 32.69 ± 7.941) F值 46.006 14.823 25.138 54.784 P值 <0.001 <0.001 <0.001 <0.001 注:与cGAMP组比较,1)P<0.05。 -
[1] ALBILLOS A, LARIO M, ÁLVAREZ-MON M. Cirrhosis-associated immune dysfunction: distinctive features and clinical relevance[J]. J Hepatol, 2014, 61( 6): 1385- 1396. DOI: 10.1016/j.jhep.2014.08.010. [2] RACANELLI V, REHERMANN B. The liver as an immunological organ[J]. Hepatology, 2006, 43(2 Suppl 1): S54-S62. DOI: 10.1002/hep.21060. [3] TILG H, VOGEL W, WIEDERMANN CJ, et al. Circulating interleukin-1 and tumor necrosis factor antagonists in liver disease[J]. Hepatology, 1993, 18( 5): 1132- 1138. [4] ERIKSSON AS, GRETZER C, WALLERSTEDT S. Elevation of cytokines in peritoneal fluid and blood in patients with liver cirrhosis[J]. Hepatogastroenterology, 2004, 51( 56): 505- 509. [5] SIEGAL FP, KADOWAKI N, SHODELL M, et al. The nature of the principal type 1 interferon-producing cells in human blood[J]. Science, 1999, 284( 5421): 1835- 1837. DOI: 10.1126/science.284.5421.1835. [6] PAIJO J, DÖRING M, SPANIER J, et al. cGAS senses human cytomegalovirus and induces type I interferon responses in human monocyte-derived cells[J]. PLoS Pathog, 2016, 12( 4): e1005546. DOI: 10.1371/journal.ppat.1005546. [7] ZHANG C, SHANG G, GUI X, et al. Structural basis of STING binding with and phosphorylation by TBK1[J]. Nature, 2019, 567( 7748): 394- 398. DOI: 10.1038/s41586-019-1000-2. [8] ISHIKAWA H, MA Z, BARBER GN. STING regulates intracellular DNA-mediated, type I interferon-dependent innate immunity[J]. Nature, 2009, 461( 7265): 788- 792. DOI: 10.1038/nature08476. [9] ABE T, BARBER GN. Cytosolic-DNA-mediated, STING-dependent proinflammatory gene induction necessitates canonical NF-κB activation through TBK1[J]. J Virol, 2014, 88( 10): 5328- 5341. DOI: 10.1128/JVI.00037-14. [10] FITZGERALD KA, MCWHIRTER SM, FAIA KL, et al. IKKepsilon and TBK1 are essential components of the IRF3 signaling pathway[J]. Nat Immunol, 2003, 4( 5): 491- 496. DOI: 10.1038/ni921. [11] SMEDSRØD B, PERTOFT H, GUSTAFSON S, et al. Scavenger functions of the liver endothelial cell[J]. Biochem J, 1990, 266( 2): 313- 327. DOI: 10.1042/bj2660313. [12] WILLEKENS FL, WERRE JM, KRUIJT JK, et al. Liver Kupffer cells rapidly remove red blood cell-derived vesicles from the circulation by scavenger receptors[J]. Blood, 2005, 105( 5): 2141- 2145. DOI: 10.1182/blood-2004-04-1578. [13] GREGORY SH, SAGNIMENI AJ, WING EJ. Bacteria in the bloodstream are trapped in the liver and killed by immigrating neutrophils[J]. J Immunol, 1996, 157( 6): 2514- 2520. [14] MERRIMAN RB, TRAN TT. AASLD practice guidelines: The past, the present, and the future[J]. Hepatology, 2016, 63( 1): 31- 34. DOI: 10.1002/hep.28345. [15] CHRYSAVGIS L, PAPATHEODORIDI A, CHOLONGITAS E, et al. Significance of circulating cell-free DNA species in non-alcoholic fatty liver disease[J]. Int J Mol Sci, 2021, 22( 16): 8849. DOI: 10.3390/ijms22168849. [16] BLASI A, PATEL VC, ADELMEIJER J, et al. Plasma levels of circulating DNA are associated with outcome, but not with activation of coagulation in decompensated cirrhosis and ACLF[J]. JHEP Rep, 2019, 1( 3): 179- 187. DOI: 10.1016/j.jhepr.2019.06.002. [17] KHANAM A, CHUA JV, KOTTILIL S. Immunopathology of chronic hepatitis B infection: Role of innate and adaptive immune response in disease progression[J]. Int J Mol Sci, 2021, 22( 11): 5497. DOI: 10.3390/ijms22115497. [18] RAZAVI H. Global epidemiology of viral hepatitis[J]. Gastroenterol Clin North Am, 2020, 49( 2): 179- 189. DOI: 10.1016/j.gtc.2020.01.001. [19] TSENG TC, HUANG LR. Immunopathogenesis of hepatitis B virus[J]. J Infect Dis, 2017, 216( suppl_8): S765-S770. DOI: 10.1093/infdis/jix356. [20] ASHARE A, STANFORD C, HANCOCK P, et al. Chronic liver disease impairs bacterial clearance in a human model of induced bacteremia[J]. Clin Transl Sci, 2009, 2( 3): 199- 205. DOI: 10.1111/j.1752-8062.2009.00122.x. [21] BERNSMEIER C, TRIANTAFYLLOU E, BRENIG R, et al. CD14+ CD15- HLA-DR- myeloid-derived suppressor cells impair antimicrobial responses in patients with acute-on-chronic liver failure[J]. Gut, 2018, 67( 6): 1155- 1167. DOI: 10.1136/gutjnl-2017-314184. -



 PDF下载 ( 711 KB)
PDF下载 ( 711 KB)

 下载:
下载: