血氨及胆碱酯酶对肝硬化伴轻微肝性脑病的早期诊断价值
DOI: 10.3969/j.issn.1001-5256.2023.02.013
Value of blood ammonia and cholinesterase in the early diagnosis of liver cirrhosis with minimal hepatic encephalopathy
-
摘要:
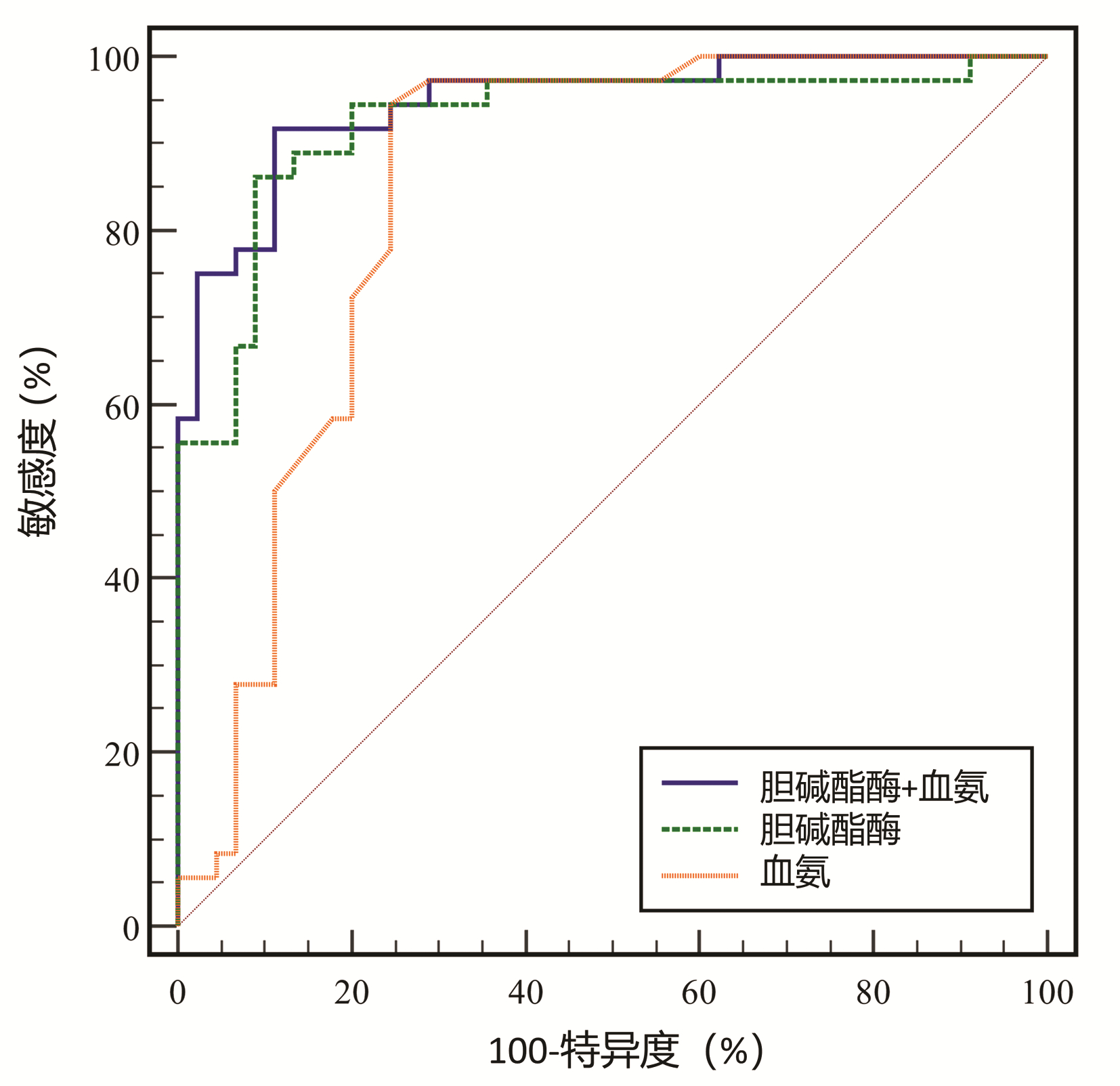
目的 探讨血清标志物对肝硬化合并轻微型肝性脑病(MHE)的早期诊断价值。 方法 前瞻性纳入2020年4月—2022年2月于宁夏医科大学总医院住院治疗,并经临床表现、实验室检查、影像学检查或肝穿刺活检确诊为乙型肝炎肝硬化患者81例。根据数字连接试验A(NCT-A)和数字符号试验(DST)分为单纯肝硬化组45例、MHE组36例。检测肝功能(ALT、AST、GGT、ALP和TBil)、白蛋白、血氨、胆碱酯酶和凝血酶原时间。计数资料2组间比较采用χ2检验;正态分布的计量资料2组间比较采用独立样本t检验;非正态分布的计量资料2组间比较采用Mann-Whitney U检验,多组间比较采用Kruskal-Wallis H检验。使用Logistic回归和受试者工作特征曲线下面积(AUC)分析MHE的预测因素。 结果 与单纯肝硬化组相比,MHE组NCT-A量表评分升高(Z=-7.110, P<0.001),DST量表评分降低(t=12.223, P<0.001),差异均有统计学意义。单因素分析显示,AST、白蛋白、凝血酶原时间、胆碱酯酶和血氨在MHE患者中显著改变(Z值分别为-2.319、-2.643、-1.982、-6.594、-5.331,P值均<0.05);但在多变量分析中,只有胆碱酯酶和血氨水平是显著的预测因子(P值均<0.05),并且它们与Child-Pugh评分分级相关(P值均<0.05)。胆碱酯酶、血氨以及两者联合诊断MHE的AUC值分别为0.925、0.845和0.941,最佳截断值分别为2966、60和0.513。 结论 血氨、胆碱酯酶及两者联合检测对肝硬化合并MHE的早期诊断具有潜在的临床价值。 Abstract:Objective To investigate the value of serum markers in the early diagnosis of liver cirrhosis with minimal hepatic encephalopathy (MHE). Methods A prospective analysis was performed for 81 patients who were hospitalized and treated in General Hospital of Ningxia Medical University from April 2020 to February 2022, and all these patients were diagnosed with hepatitis B cirrhosis based on clinical manifestation, laboratory examination, and radiological examination or liver biopsy. According to digital connection test A (NCT-A) and digital symbol test (DST), these patients were divided into simple cirrhosis group with 45 patients and MHE group with 36 patients. Related indices were measured, including liver function [alanine aminotransferase (ALT), aspartate aminotransferase (AST), gamma-glutamyl transpeptidase (GGT), alkaline phosphatase (ALP), and total bilirubin (TBil)], albumin, blood ammonia, cholinesterase, and prothrombin time. The independent samples t-test was used for comparison of normally distributed continuous data between two groups; the Mann-Whitney U test was used for comparison of non-normally distributed continuous data between two groups, and the Kruskal-Wallis H test was used for comparison between multiple groups; the chi-square test was used for comparison of categorical data between groups. The logistic regression analysis and the area under the ROC curve (AUC) were used to investigate the predictive factors for MHE. Results Compared with the simple cirrhosis group, the MHE group had a significant increase in NCT-A score (Z=-7.110, P < 0.001) and a significant reduction in DST score (t=12.223, P < 0.001). The univariate analysis showed that there were significant changes in AST, albumin, prothrombin time, cholinesterase, and blood ammonia in the patients with MHE (Z=-2.319, -2.643, -1.982, -6.594, and -5.331, all P < 0.05), while the multivariate analysis showed that only cholinesterase and blood ammonia were significant predictive factors (all P < 0.05) and were correlated with Child-Pugh score (all P < 0.05). Cholinesterase, blood ammonia, and their combination had an AUC of 0.925, 0.845, and 0.941, respectively, in the diagnosis of MHE, with an optimal cut-off value of 2966, 60, and 0.513, respectively. Conclusion Blood ammonia, cholinesterase, and their combined measurement have a potential clinical value in the early diagnosis of liver cirrhosis with MHE. -
Key words:
- Liver Cirrhosis /
- Hepatic Encephalopathy /
- Hyperammonemia /
- Cholinesterases /
- Early Diagnosis
-
表 1 肝硬化组及MHE组患者的一般资料比较
Table 1. General information of liver cirrhosis and MHE group
项目 肝硬化组(n=45) MHE组(n=36) 统计值 P值 男/女(例) 30/15 23/13 χ2=0.068 0.749 年龄(年) 48.5±8.5 50.3±9.7 t=-0.856 0.395 教育水平(年) 7.8±1.8 7.7±1.9 t=0.335 0.738 NCT-A(s) 39.5(32.8~44.8) 70.7(60.2~84.6) Z=-7.110 0.001 DST(分) 43.0±9.8 21.2±4.7 t=12.223 0.001 ALT(U/L) 89.5(41.9~126.0) 50.5(78.9~130.1) Z=-0.033 0.973 AST(U/L) 64.6(43.0~87.8) 90.9(52.1~125.1) Z=-2.319 0.020 GGT(U/L) 67.8(60.4~111.0) 66.4(29.6~130.9) Z=-0.718 0.473 ALP(U/L) 114.8(75.8~161.7) 104.4(71.0~155.5) Z=-0.661 0.509 TBil(μmol/L) 78.9(26.1~130.2) 83.5(57.3~133.1) Z=-0.932 0.352 白蛋白(g/L) 29.4(27.0~33.6) 26.9(23.5~29.6) Z=-2.643 0.008 血氨(μmol/L) 48(42~62) 64(69~80) Z=-5.331 0.001 凝血酶原时间(s) 16.8(14.5~17.9) 17.9(15.9~18.9) Z=-1.982 0.047 胆碱酯酶(U/L) 3567(3274~4148) 2399(2024~2781) Z=-6.594 0.001 表 2 单变量和多变量分析的诊断效能
Table 2. Diagnostic efficacy of univariate and multivariate analysis
变量 截断值 AUC 敏感度(%) 特异度(%) 约登指数 胆碱酯酶 <2966 0.925 86.1(70.5~95.3) 91.1(78.8~97.5) 0.772 血氨 >60 0.845 94.4(81.3~99.3) 75.6(60.5~87.1) 0.700 胆碱酯酶+血氨 >0.513 0.941 91.7(77.5~98.2) 84.4(70.5~93.5) 0.761 表 3 MHE和肝硬化患者Child-Pugh分级分析
Table 3. Child-Pugh grading analysis of patients with minimai hepatic encephalopathy and cirrhosis
统计变量 Child-Pugh A级(n=35) Child-Pugh B级(n=25) Child-Pugh C级(n=21) 统计值 P值 男/女(例) 24/11 15/10 14/7 χ2=0.493 0.782 年龄(岁) 49(43~55) 52(56.5~39.5) 55(43~57.5) H=1.840 0.398 胆碱酯酶(U/L) 3790(3423~4432) 2980(2756~3153)1) 1966(2236~2521)1)2) H=69.813 0.01 血氨(μmol/L) 45(37~56) 46(41~71) 65(60~74)1)2) H=25.126 0.01 注:与Child-Pugh A级比较,1)P<0.05;与Child-Pugh B级比较,2)P<0.05。 -
[1] REGA D, AIKO M, PEÑARANDA N, et al. Patients with minimal hepatic encephalopathy show altered thermal sensitivity and autonomic function[J]. J Clin Med, 2021, 10(2): 239. DOI: 10.3390/jcm10020239. [2] MONTAGNESE S, BAJAJ JS. Impact of hepatic encephalopathy in cirrhosis on quality-of-life issues[J]. Drugs, 2019, 79(Suppl 1): 11-16. DOI: 10.1007/s40265-018-1019-y. [3] YANG X, LIU W, DANG P, et al. Decreased brain noradrenaline in minimal hepatic encephalopathy is associated with cognitive impairment in rats[J]. Brain Res, 2022, 1793: 148041. DOI: 10.1016/j.brainres.2022.148041. [4] CÓRDOBA J, MÍNGUEZ B. Hepatic encephalopathy[J]. Semin Liver Dis, 2008, 28(1): 70-80. DOI: 10.1055/s-2008-1040322. [5] GU TM, ZHANG Y, JJIANG YP, et al. Efficacy of acarbose-linked ornithine aspartate in hepatic encephalopathy in cirrhotic patients[J]. Chin J Mod Appl Pharm, 2018, 35(10): 1538-1542. DOI: 10.13748/j.cnki.issn1007-7693.2018.10.023.顾体梅, 张勇, 江永平, 等. 阿卡波糖联合门冬氨酸鸟氨酸对肝硬化患者肝性脑病的疗效评估[J]. 中国现代应用药学, 2018, 35(10): 1538-1542. DOI: 10.13748/j.cnki.issn1007-7693.2018.10.023. [6] FICHET J, MERCIER E, GENÉE O, et al. Prognosis and 1-year mortality of intensive care unit patients with severe hepatic encephalopathy[J]. J Crit Care, 2009, 24(3): 364-370. DOI: 10.1016/j.jcrc.2009.01.008. [7] PESSIDJO DJOMATCHO L, KOWO MP, NDAM AN, et al. Normalisation of the psychometric encephalopathy score within the Cameroonian population[J]. BMC Gastroenterol, 2021, 21(1): 287. DOI: 10.1186/s12876-021-01858-7. [8] HA ZY, MATHEW S, YEONG KY. Butyrylcholinesterase: a multifaceted pharmacological target and tool[J]. Curr Protein Pept Sci, 2020, 21(1): 99-109. DOI: 10.2174/1389203720666191107094949. [9] TURECKY L, KUPCOVA V, DURFINOVA M, et al. Serum butyrylcholinesterase activities in patients with non-alcoholic fatty liver disease. Comparison with liver proteosynthetic function and liver fibrosis[J]. Bratisl Lek Listy, 2021, 122(10): 689-694. DOI: 10.4149/BLL_2021_110. [10] PAREKH PJ, BALART LA. Ammonia and its role in the pathogenesis of hepatic encephalopathy[J]. Clin Liver Dis, 2015, 19(3): 529-537. DOI: 10.1016/j.cld.2015.05.002. [11] ZIMMERMANN M, REICHERT AS. Rapid metabolic and bioenergetic adaptations of astrocytes under hyperammonemia-a novel perspective on hepatic encephalopathy[J]. Biol Chem, 2021, 402(9): 1103-1113. DOI: 10.1515/hsz-2021-0172. [12] PALOMERO-GALLAGHER N, ZILLES K. Neurotransmitter receptor alterations in hepatic encephalopathy: a review[J]. Arch Biochem Biophys, 2013, 536(2): 109-121. DOI: 10.1016/j.abb.2013.02.010. [13] FIATI KENSTON SS, SONG X, LI Z, et al. Mechanistic insight, diagnosis, and treatment of ammonia-induced hepatic encephalopathy[J]. J Gastroenterol Hepatol, 2019, 34(1): 31-39. DOI: 10.1111/jgh.14408. [14] XU XY, DING HG, LI WG, et al. Chinese guidelines on the management of liver cirrhosis[J]. Clin Hepatal, 2019, 35(11): 2408-2425. DOI: 10.3969/j.issn.1001-5256.2019.11.006.徐小元, 丁惠国, 李文刚, 等. 肝硬化诊治指南[J]. 临床肝胆病杂志, 2019, 35(11): 2408-2425. DOI: 10.3969/j.issn.1001-5256.2019.11.006. [15] Chinese Society of Gastroenterology, Chinese Medical Association, Chinese Society of Hepatology, Chinese Medical Association. Consensus on diagnosis and treatment of hepatic encephalopathy in China (Chongqing, 2013)[J/CD]. Chin J Front Med Sci (Electronic Version), 2014, 6(2): 81-93.中华医学会消化病学分会, 中华医学会肝病学分会. 中国肝性脑病诊治共识意见(2013年, 重庆)[J/CD]. 中国医学前沿杂志(电子版), 2014, 6(2): 81-93. [16] WANG JY, ZHANG NP, CHI BR, et al. Prevalence of minimal hepatic encephalopathy and quality of life evaluations in hospitalized cirrhotic patients in China[J]. World J Gastroenterol, 2013, 19(30): 4984-4991. DOI: 10.3748/wjg.v19.i30.4984. [17] HANSEN M, KJÆRGAARD K, ERIKSEN LL, et al. Psychometric methods for diagnosing and monitoring minimal hepatic encephalopathy -current validation level and practical use[J]. Metab Brain Dis, 2022, 37(3): 589-605. DOI: 10.1007/s11011-022-00913-w. [18] BAJAJ JS, WADE JB, SANYAL AJ. Spectrum of neurocognitive impairment in cirrhosis: Implications for the assessment of hepatic encephalopathy[J]. Hepatology, 2009, 50(6): 2014-2021. DOI: 10.1002/hep.23216. [19] HADJIHAMBI A, ARIAS N, SHEIKH M, et al. Hepatic encephalopathy: a critical current review[J]. Hepatol Int, 2018, 12(Suppl 1): 135-147. DOI: 10.1007/s12072-017-9812-3. [20] SORIANO G, BAJAJ JS. Grading the range of hepatic encephalopathy from overt to covert: Animals to the rescue![J]. Hepatology, 2017, 66(1): 10-12. DOI: 10.1002/hep.29186. [21] ELSAID MI, RUSTGI VK, LOO N, et al. The burden associated with thrombocytopenia and platelet transfusions among patients with chronic liver disease[J]. J Med Econ, 2020, 23(4): 378-385. DOI: 10.1080/13696998.2019.1699563. [22] TAN L, MENG Y, ZENG T, et al. Clinical diagnostic significance of prealbumin, cholinesterase and retinol binding protein in liver cirrhosis combined with encephalopathy[J]. Br J Biomed Sci, 2019, 76(1): 24-28. DOI: 10.1080/09674845.2018.1523673. [23] AMPUERO J, MONTOLIÚ C, SIMÓN-TALERO M, et al. Minimal hepatic encephalopathy identifies patients at risk of faster cirrhosis progression[J]. J Gastroenterol Hepatol, 2018, 33(3): 718-725. DOI: 10.1111/jgh.13917. [24] RIGGIO O, AMODIO P, FARCOMENI A, et al. A model for predicting development of overt hepatic encephalopathy in patients with cirrhosis[J]. Clin Gastroenterol Hepatol, 2015, 13(7): 1346-1352. DOI: 10.1016/j.cgh.2014.12.025. [25] MONTAGNESE S, RUSSO FP, AMODIO P, et al. Hepatic encephalopathy 2018: A clinical practice guideline by the Italian Association for the Study of the Liver (AISF)[J]. Dig Liver Dis, 2019, 51(2): 190-205. DOI: 10.1016/j.dld.2018.11.035. [26] GAO YY, ZHANG X, LI FH, et al. Measurement of glycosylated albumin and its application value in liver cirrhosis patients with different Child-Pugh classes[J]. Clin Hepatal, 2022, 38(2): 347-351. DOI: 10.3969/j.issn.1001-5256.2022.02.018.高艳颖, 张旭, 李凤慧, 等. 不同child-pugh分级肝硬化患者糖化白蛋白的测定及其应用价值[J]. 临床肝胆病杂志, 2022, 38(2): 347-351. DOI: 10.3969/j.issn.1001-5256.2022.02.018. [27] DJIAMBOU-NGANJEU H. Hepatic encephalopathy in liver cirrhosis[J]. J Transl Int Med, 2017, 5(1): 64-67. DOI: 10.1515/jtim-2017-0013. -



 PDF下载 ( 2173 KB)
PDF下载 ( 2173 KB)

 下载:
下载:


