急性胰腺炎患者血浆内皮细胞微粒水平的改变及其形成机制
DOI: 10.3969/j.issn.1001-5256.2022.09.027
Changes and formation mechanism of plasma endothelial microparticles in patients with acute pancreatitis
-
摘要:
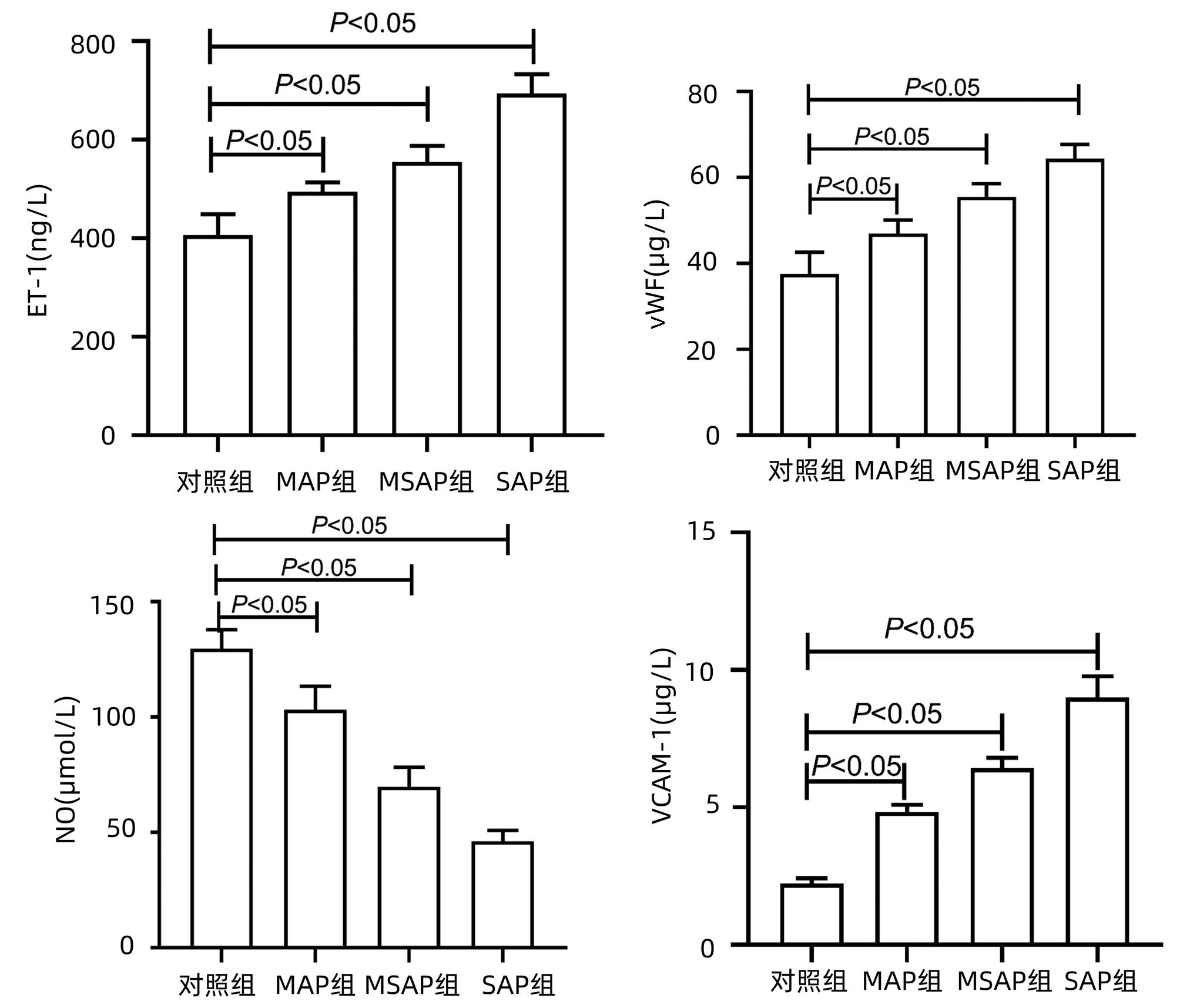
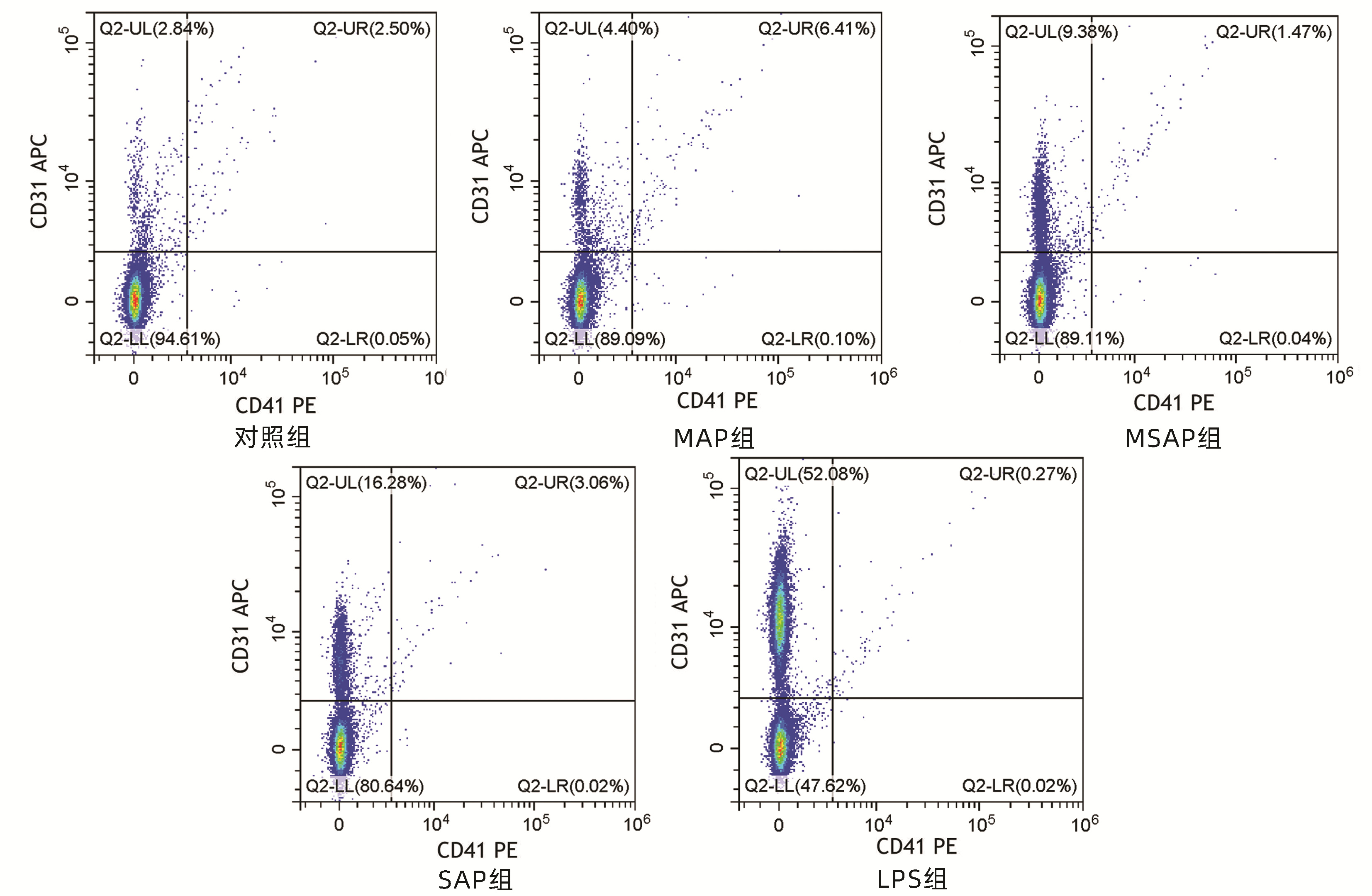
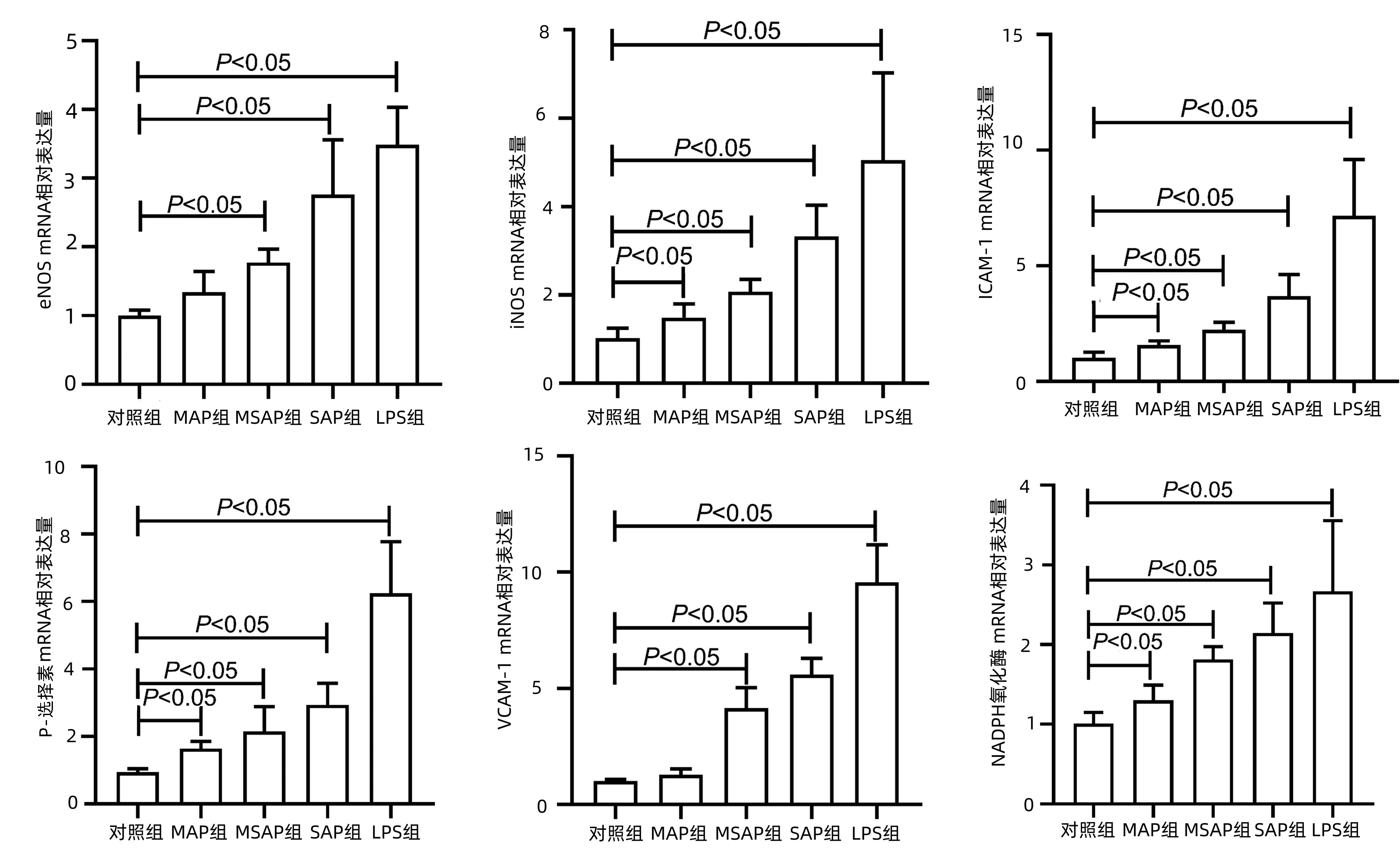
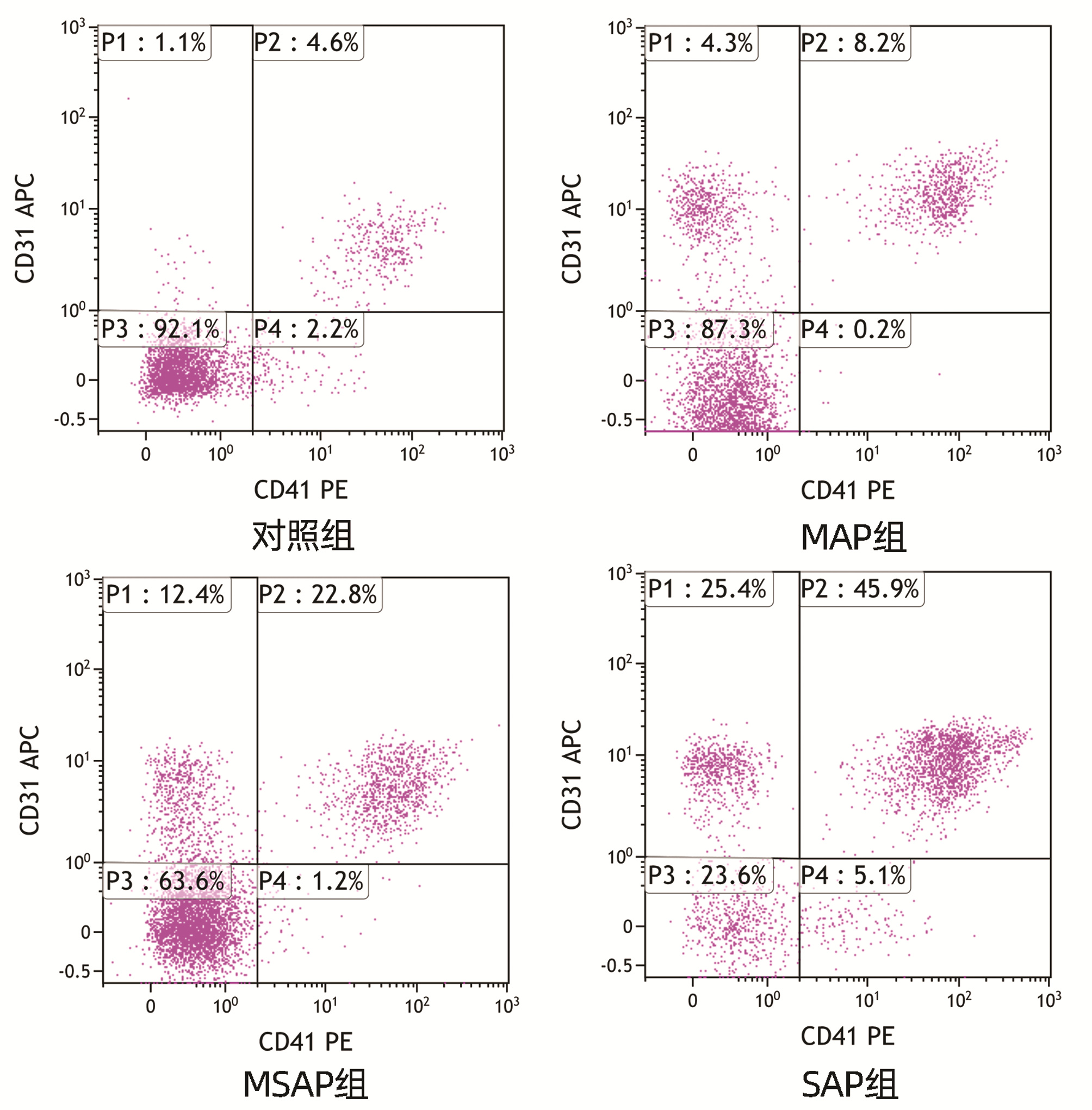
目的 探讨急性胰腺炎(AP)患者血浆内皮细胞微粒(EMP)水平的改变并初步观察其形成的机制。 方法 收集安徽医科大学第一附属医院2020年8月-2021年6月60例AP患者的血液标本, 分为轻症急性胰腺炎组(MAP组, n=23)、中度重症急性胰腺炎组(MSAP组, n=23)和重症急性胰腺炎组(SAP组, n=14), 取健康体检者20例为对照组。使用差速离心法获得贫血小板血浆, 流式细胞仪检测CD31+CD41-EMP水平, ELISA检测内皮素(ET-1)、血管性血友病因子(vWF)、一氧化氮(NO)和血管内皮细胞黏附分子-1(VCAM-1)水平。使用AP患者的血浆刺激HUVEC细胞, 使用流式细胞术和qRT-PCR分别检测EMP、细胞活性氧(ROS)、线粒体膜电位的变化及内皮型一氧化氮合酶(eNOS)、诱导型一氧化氮合酶(iNOS)、细胞间黏附分子-1(ICAM-1)、VCAM-1、NADPH氧化酶及P-选择素的表达。符合正态分布的计量资料多组间比较采用单因素方差分析, 进一步两两比较采用LSD-t检验; 不符合正态分布的计量资料组间和组内两两比较采用Kruskal-WallisH秩和检验。计数资料组间比较采用χ2检验。相关分析采用Pearson相关性检验。 结果 与对照组相比, MAP组、MSAP组、SAP组EMP水平均明显升高(P值均 < 0.05)。与MAP组、MSAP组比较, SAP组EMP水平明显升高(P值均 < 0.05)。AP患者EMP水平与APACHEⅡ评分、BISAP评分、Ranson评分、CT评分、CRP均呈正相关(r值分别为0.686 2、0.777 3、0.713 8、0.771 8、0.473 9, P值均 < 0.01)。与对照组相比, MAP组、MSAP组、SAP组的ET-1、vWF、VCAM-1水平明显升高, NO水平明显降低(P值均 < 0.05)。与对照组相比, MSAP和SAP组血浆可促进HUVEC中EMP大量释放(P值均 < 0.05)。与对照组比较, 除MAP组的VCAM-1和eNOS外, 其余各组的eNOS、iNOS、ICAM-1、P-选择素、VCAM-1、NADPH氧化酶的mRNA表达水平均显著升高(P值均 < 0.05)。与对照组相比, MAP组、MSAP组、SAP组、LPS组患者HUVEC中的ROS水平升高明显, 线粒体膜电位下降显著(P值均 < 0.05)。 结论 AP患者血浆EMP水平明显升高, 与胰腺炎严重程度相关, 且AP患者血浆可刺激HUVEC形成EMP, 机制可能与细胞氧化损伤相关。 Abstract:Objective To investigate the changes and formation mechanism of plasma endothelial microparticles (EMPs) in patients with acute pancreatitis (AP). Methods Blood samples were collected from 60 patients with AP who were treated in The First Affiliated Hospital of Anhui Medical University from August 2020 to June 2021, and these patients were divided into mild acute pancreatitis (MAP) group with 23 patients, moderate-severe acute pancreatitis (MSAP) group with 23 patients, and severe acute pancreatitis (SAP) group with 14 patients; 20 individuals who underwent physical examination were enrolled as control group.Differential centrifugation was used to obtain platelet-poor plasma, flow cytometry was used to measure the level of CD31+CD41-EMPs, and ELISA was used to measure the levels of endothelin-1(ET-1), von Willebrand factor (vWF), nitric oxide (NO), and vascular cell adhesion molecule-1(VCAM-1).HUVECs were stimulated by the plasma of AP patients, and then flow cytometry and qRT-PCR were used to measure the changes in EMPs, reactive oxygen species (ROS), and mitochondrial membrane potential and the expression of endothelial nitric oxide synthase (eNOS), inducible nitric oxide synthase (iNOS), intercellular adhesion molecule-1(ICAM-1), VCAM-1, NADPH oxidase, and P-selectin.A one-way analysis of variance was used for comparison of normally distributed continuous data between multiple groups, and the least significant difference t-test was used for further comparison between two groups.The Kruskal-Wallis H test was used for comparison of non-normally distributed continuous data between groups and within each group.The chi-square test was used for comparison of categorical data between groups, and the Pearson correlation test was used for correlation analysis. Results Compared with the control group, the MAP, MSAP, and SAP groups had a significant increase in the level of EMPs (all P < 0.05).Compared with the MAP and MSAP groups, the SAP group had a significant increase in the level of EMPs (both P < 0.05).In the patients with AP, the level of EMPs was negatively correlated with Acute Physiology and Chronic Health Evaluation Ⅱ score, Bedside Index for Severity in Acute Pancreatitis, Ranson score, CT score, and C-reactive protein (r=0.686 2, 0.777 3, 0.713 8, 0.771 8, and 0.473 9, all P < 0.01).Compared with the control group, the MAP, MSAP, and SAP groups had significant increases in the levels of ET-1, vWF, and VCAM-1 and a significant reduction in the level of NO (all P < 0.05).Compared with the control group, the MSAP and SAP groups had the plasma that promoted the release of a large amount of EMPs (both P < 0.05).Compared with the control group, all the other groups, except the MAP group in terms of VCAM-1 and eNOS, had significant increases in the mRNA expression levels of eNOS, iNOS, ICAM-1, P-selectin, VCAM-1, and NADPH oxidase (all P < 0.05).Compared with the HC group, the MAP, MSAP, and SAP groups and the LPS group had a significant increase in the level of ROS and a significant reduction in mitochondrial membrane potential in HUVECs (all P < 0.05). Conclusion There is a significant increase in the plasma level of EMPs in AP patients, which is correlated with the severity of pancreatitis.Meanwhile, the plasma of AP patients can promote the formation of EMPs in HUVECs in vitro, which may be associated with cell oxidative injury. -
Key words:
- Pancreatitis /
- Endothelial Microparticles /
- Endothelial Cells
-
表 1 qRT-PCR引物序列
Table 1. Primers of qRT-PCR
引物名称 引物序列(5′-3′) GAPDH F:AACAGCGACACCCACTCCTC R:CATACCAGGAAATGAGCTTGACAA eNOS F:GTGGCTGTCTGCATGGACCT R:CCACGATGGTGACTTTGGCT iNOS F:ATGGAACATCCCAAATACGA R:GTCGTAGAGGACCACTTTGT ICAM-1 F:CCTTCCTCACCGTGTACTGC R:AGCGTAGGGTAAGGTTCTTGC VCAM-1 F:ACTGGTGGCCTCCTGAATGG R:CTGTGTCTCCTGTCTCCGCT NADPH氧化酶 F:CTGTGGTGTTACTATCTGTATTTTCTC R:CTTGCTGCATTCAGTTCAACA P-选择素 F:CTGTTACCCTGGATTCTATGGGC R:GCTGCACTGCGAGTTAAAAGAG 注:GAPDH,内参。 表 2 AP患者临床资料
Table 2. Clinical data of acute pancreatitis patients
指标 对照组(n=20) MAP组(n=23) MSAP组(n=23) SAP组(n=14) 统计值 P值 性别(例) χ2=6.344 0.096 男 13 17 9 9 女 7 6 14 5 年龄(岁) 46.10±17.15 45.22±15.61 45.00±16.13 50.93±21.45 F=0.510 0.747 WBC(×109/L) 5.95±1.66 12.55±4.541) 15.15±6.241) 12.91±3.471) F=16.220 <0.01 中性粒细胞(×109/L) 3.35±0.97 10.66±4.301) 12.99±5.341) 11.39±3.501) F=23.350 <0.01 RBC(×1012/L) 4.51±0.36 4.87±0.631) 4.64±0.79 4.66±0.88 F=1.056 0.370 HB(g/L) 135.00(124.50~145.25) 149.00(131.00~164.00)1) 140.00(120.00~158.00) 138.50(125.75~172.25) H=5.929 0.115 HCT(%) 40.12±4.34 43.33±5.161) 40.62±6.26 42.01±8.54 F=1.261 0.294 CRP(mg/L) 0.53(0.43~0.65) 21.88(6.01~121.27)1) 60.31(5.82~196.19)1) 141.28(44.19~252.83)1) H=46.070 <0.01 Alb(g/L) 42.48±2.87 42.89±5.45 40.68±5.50 34.10±6.831) F=9.520 <0.01 TBil(μmol/L) 13.55(9.66~15.58) 15.85(10.89~22.85) 17.50(11.94~36.17)1) 25.08(12.83~33.32)1) H=7.348 0.062 ALT(U/L) 15.50(10.25~25.25) 43.00(33.00~70.00)1) 37.00(19.00~264.00)1) 30.50(19.75~45.50)1) H=19.529 <0.01 AST(U/L) 17.00(15.25~20.00) 26.00(22.00~53.00)1) 41.00(23.00~116.00)1) 38.50(25.25~78.25)1) H=26.067 <0.01 ALP(U/L) 81.00(68.00~97.75) 102.00(87.00~137.00)1) 101.00(84.00~141.00)1) 90.50(76.25~97.75) H=12.667 <0.01 LDH(U/L) 161.00(148.00~174.00) 207.00(177.00~244.00)1) 247.00(183.40~323.00)1) 521.50(220.00~733.25)1) H=48.475 <0.01 BUN(μmol/L) 5.18±1.28 5.55±1.36 4.56±2.02 7.05±2.991) F=5.054 0.003 CRE(μmol/L) 66.04±9.94 59.80±18.90 54.64±27.07 86.08±58.23 F=3.372 0.023 钾(mmol/L) 3.93±0.33 4.28±0.421) 3.80±0.31 4.08±0.90 F=3.635 0.017 钠(mmol/L) 140.95(139.40~142.18) 136.20(134.10~138.40)1) 136.60(131.80~138.50)1) 136.50(131.18~140.55)1) H=24.179 <0.01 钙(mmol/L) 2.30(2.23~2.40) 2.32(2.19~2.45) 2.14(2.03~2.23)1) 1.78(1.14~2.10)1) H=30.525 <0.01 血糖(mmol/L) 4.83(4.53~5.18) 7.42(6.33~13.16)1) 8.18(7.07~9.76)1) 8.99(6.63~11.56)1) H=31.061 <0.01 APACHE-Ⅱ评分≥3[例(%)] 16(69.6) 21(91.3) 14(100.0) χ2=7.485 <0.05 BISAP评分≥3[例(%)] 0 1(4.3) 10(71.4) χ2=34.528 <0.05 Ranson评分≥3[例(%)] 0 12(52.2) 12(85.7) χ2=28.944 <0.05 CT评分>3[例(%)] 5(21.7) 13(56.5) 14(100.0) χ2=21.568 <0.05 注:与对照组比较,1)P<0.05。 -
[1] GARG PK, SINGH VP. Organ failure due to systemic injury in acute pancreatitis[J]. Gastroenterology, 2019, 156(7): 2008-2023. DOI: 10.1053/j.gastro.2018.12.041. [2] SINGH P, GARG PK. Pathophysiological mechanisms in acute pancreatitis: Current understanding[J]. Indian J Gastroenterol, 2016, 35(3): 153-166. DOI: 10.1007/s12664-016-0647-y. [3] GE N, XIA Q, YANG ZH, et al. Vascular endothelial injury and apoptosis in rats with severe acute pancreatitis[J]. Gastroenterol Res Pract, 2015, 2015: 235017. DOI: 10.1155/2015/235017. [4] DUMNICKA P, MADUZIA D, CERANOWICZ P, et al. The interplay between inflammation, coagulation and endothelial injury in the early phase of acute pancreatitis: clinical implications[J]. Int J Mol Sci, 2017, 18(2): 354. DOI: 10.3390/ijms18020354. [5] de OLIVEIRA C, KHATUA B, NOEL P, et al. Pancreatic triglyceride lipase mediates lipotoxic systemic inflammation[J]. J Clin Invest, 2020, 130(4): 1931-1947. DOI: 10.1172/JCI132767. [6] MAHMOUD AM, WILKINSON FL, MCCARTHY EM, et al. Endothelial microparticles prevent lipid-induced endothelial damage via Akt/eNOS signaling and reduced oxidative stress[J]. FASEB J, 2017, 31(10): 4636-4648. DOI: 10.1096/fj.201601244RR. [7] PARKER B, Al-HUSAIN A, PEMBERTON P, et al. Suppression of inflammation reduces endothelial microparticles in active systemic lupus erythematosus[J]. Ann Rheum Dis, 2014, 73(6): 1144-1150. DOI: 10.1136/annrheumdis-2012-203028. [8] JALAL D, RENNER B, LASKOWSKI J, et al. Endothelial microparticles and systemic complement activation in patients with chronic kidney disease[J]. J Am Heart Assoc, 2018, 7(14): e007818. DOI: 10.1161/JAHA.117.007818. [9] ABBAS M, JESEL L, AUGER C, et al. Endothelial microparticles from acute coronary syndrome patients induce premature coronary artery endothelial cell aging and thrombogenicity: Role of the Ang Ⅱ/AT1 receptor/NADPH oxidase-mediated activation of MAPKs and PI3-kinase pathways[J]. Circulation, 2017, 135(3): 280-296. DOI: 10.1161/CIRCULATIONAHA.116.017513. [10] Al-QAISSI A, PAPAGEORGIOU M, DESHMUKH H, et al. Effects of acute insulin-induced hypoglycaemia on endothelial microparticles in adults with and without type 2 diabetes[J]. Diabetes Obes Metab, 2019, 21(3): 533-540. DOI: 10.1111/dom.13548. [11] BANKS PA, BOLLEN TL, DERVENIS C, et al. Classification of acute pancreatitis——2012: revision of the Atlanta classification and definitions by international consensus[J]. Gut, 2013, 62(1): 102-111. DOI: 10.1136/gutjnl-2012-302779. [12] POHL PH, LOZITO TP, CUPERMAN T, et al. Catabolic effects of endothelial cell-derived microparticles on disc cells: Implications in intervertebral disc neovascularization and degeneration[J]. J Orthop Res, 2016, 34(8): 1466-1474. DOI: 10.1002/jor.23298. [13] TOMKÖTTER L, ERBES J, TREPTE C, et al. The effects of pancreatic microcirculatory disturbances on histopathologic tissue damage and the outcome in severe acute pancreatitis[J]. Pancreas, 2016, 45(2): 248-253. DOI: 10.1097/MPA.0000000000000440. [14] LEROYER AS, TEDGUI A, BOULANGER CM. Role of microparticles in atherothrombosis[J]. J Intern Med, 2008, 263(5): 528-537. DOI: 10.1111/j.1365-2796.2008.01957.x. [15] PERNOMIAN L, MOREIRA JD, GOMES MS. In the view of endothelial microparticles: Novel perspectives for diagnostic and pharmacological management of cardiovascular risk during diabetes distress[J]. J Diabetes Res, 2018, 2018: 9685205. DOI: 10.1155/2018/9685205. [16] LI T, LUO N, DU L, et al. Tumor necrosis factor-α plays an initiating role in extracorporeal circulation-induced acute lung injury[J]. Lung, 2013, 191(2): 207-214. DOI: 10.1007/s00408-012-9449-x. [17] CLEMMER JS, XIANG L, LU S, et al. Hyperglycemia-mediated oxidative stress increases pulmonary vascular permeability[J]. Microcirculation, 2016, 23(3): 221-229. DOI: 10.1111/micc.12267. [18] BURGER D, TURNER M, XIAO F, et al. High glucose increases the formation and pro-oxidative activity of endothelial microparticles[J]. Diabetologia, 2017, 60(9): 1791-1800. DOI: 10.1007/s00125-017-4331-2. [19] CAO WL, XIANG XH, CHEN K, et al. Potential role of NADPH oxidase in pathogenesis of pancreatitis[J]. World J Gastrointest Pathophysiol, 2014, 5(3): 169-177. DOI: 10.4291/wjgp.v5.i3.169. [20] CHAN S, LIAN Q, CHEN MP, et al. Deferiprone inhibits iron overload-induced tissue factor bearing endothelial microparticle generation by inhibition oxidative stress induced mitochondrial injury, and apoptosis[J]. Toxicol Appl Pharmacol, 2018, 338: 148-158. DOI: 10.1016/j.taap.2017.11.005. [21] CHEN YH, CHEN ZW, LI HM, et al. AGE/RAGE-induced EMP release via the NOX-derived ROS pathway[J]. J Diabetes Res, 2018, 2018: 6823058. DOI: 10.1155/2018/6823058. -



 PDF下载 ( 3880 KB)
PDF下载 ( 3880 KB)

 下载:
下载: