经肝动脉化疗栓塞术联合微波消融与重复肝切除术治疗复发性肝细胞癌的效果比较
DOI: 10.3969/j.issn.1001-5256.2022.09.020
Clinical effect of transcatheter arterial chemoembolization combined with microwave ablation versus repeat resection in treatment of recurrent hepatocellular carcinoma
-
摘要:
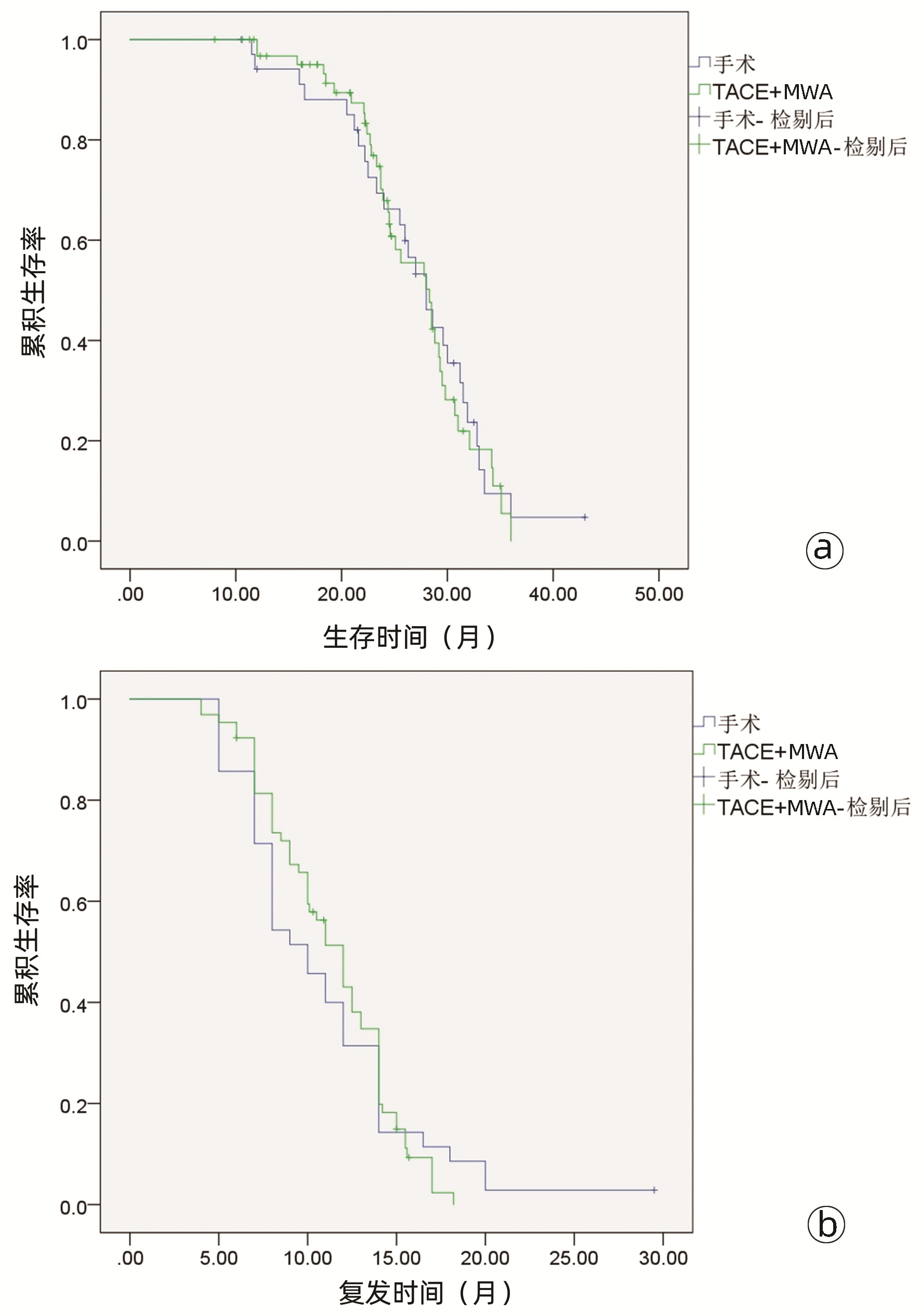
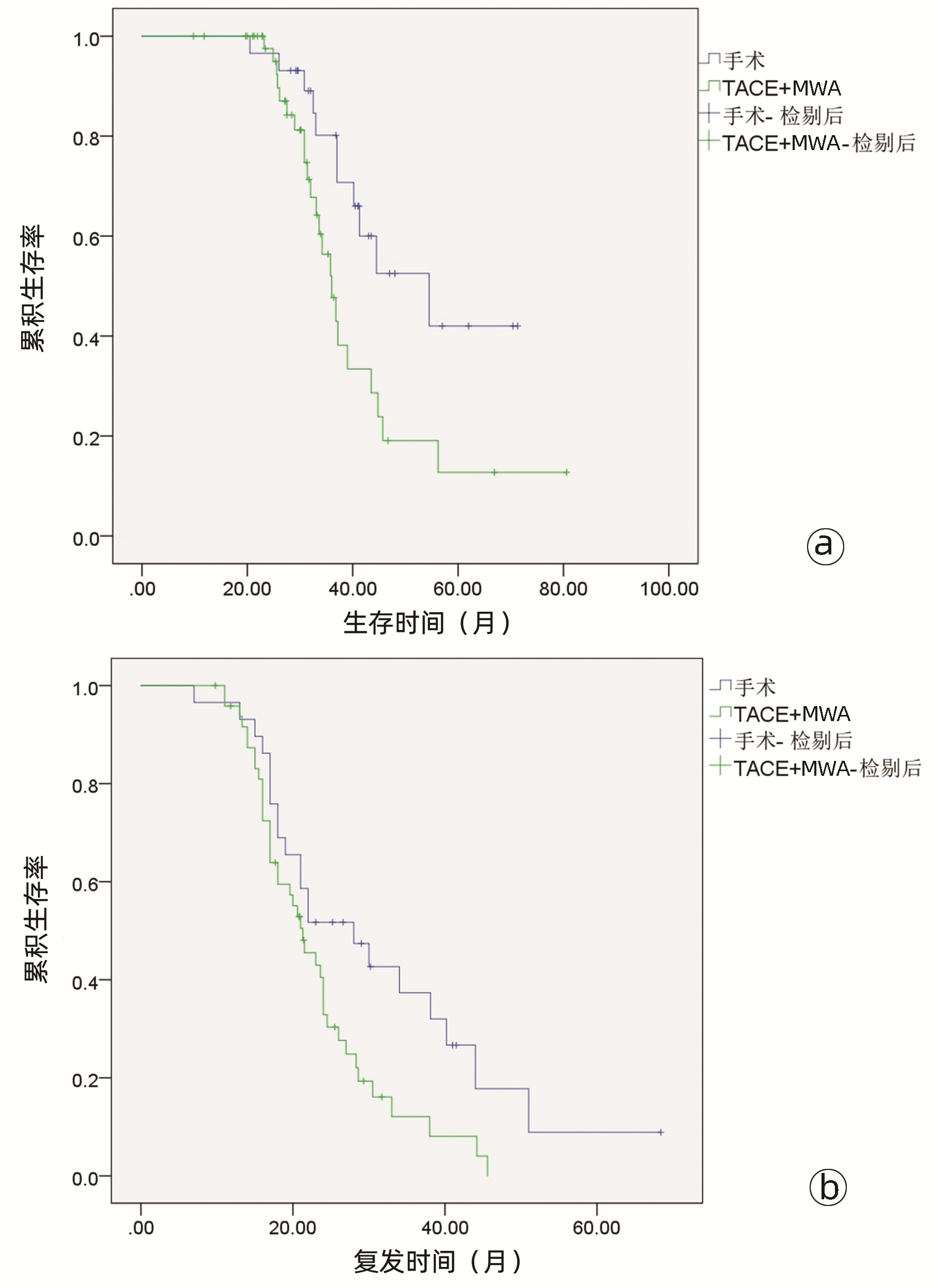
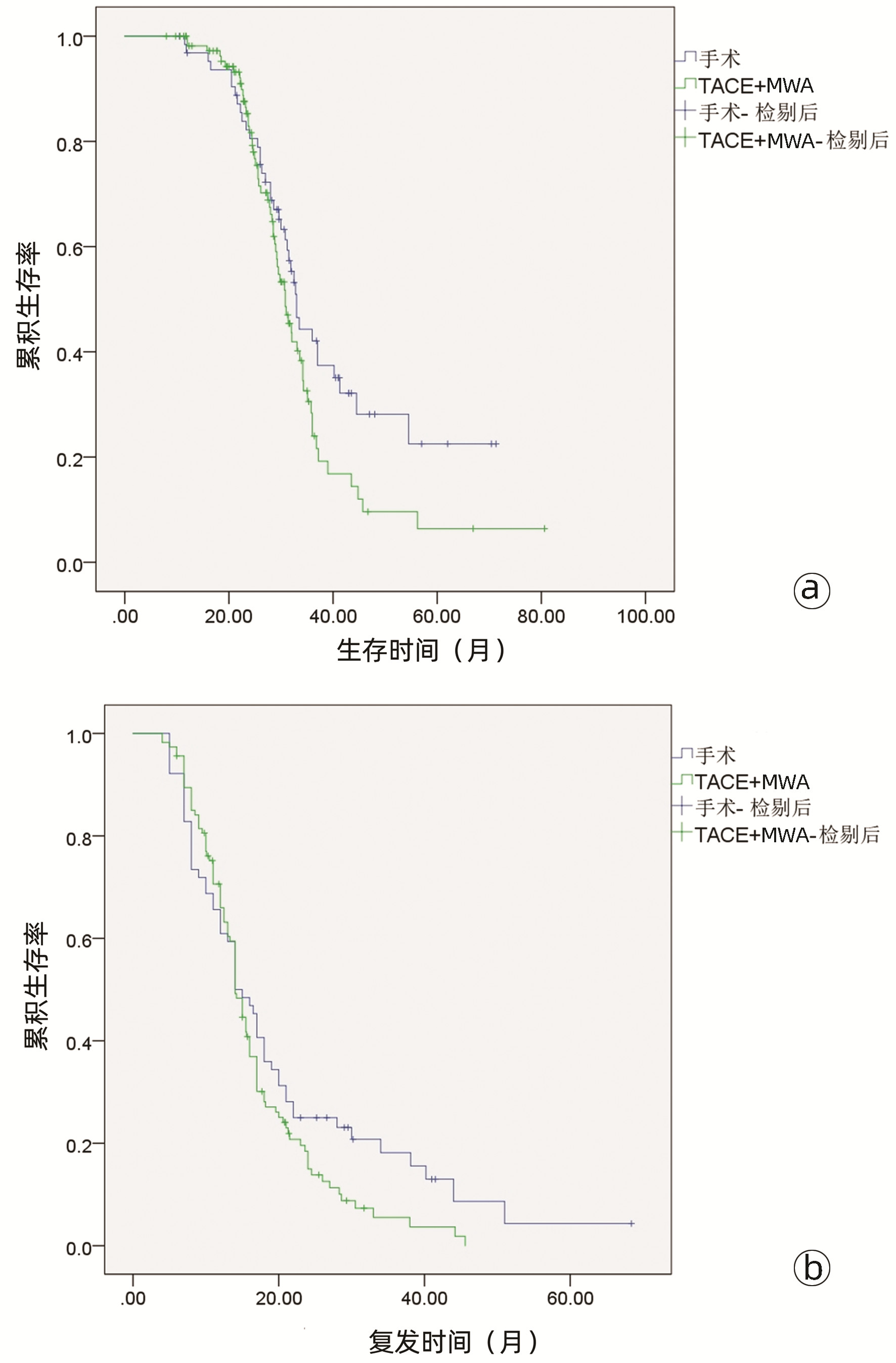
目的 比较经肝动脉化疗栓塞(TACE)联合微波消融(MWA)(TACE-MWA)与重复肝切除术(RR)治疗复发性肝细胞癌(RHCC)的效果。 方法 选取2015年6月1日—2020年9月30日内江市第二人民医院收治的178例RHCC患者,按治疗方式不同分为RR组(n=64例)和TACE-MWA组(n=114)。记录治疗前基线人口学资料、肝功能及肿瘤相关情况等。随访至2021年10月,比较两组患者术后总生存期(OS)和无复发生存期(RFS)。进行基于复发模式(复发时间和肿瘤大小)的亚组分析,并研究预后的影响因素。计量资料两组间比较采用独立样本t检验,计数资料两组间比较采用χ2检验;术后生存率用Kaplan-Meier法,两组间生存差异用Log-rank检验;使用Cox多因素分析方法探寻影响生存的独立危险因素。 结果 多因素分析显示,肿瘤直径、AFP水平、ALT、Alb和复发时间是OS的独立预后因素(P值均<0.05),AFP水平和复发时间是RFS的独立预后因素(P值均<0.05)。对于晚期复发(>2年)的RHCC,RR组与TACE-MWA组之间的中位OS有显著差异(54.0个月vs 36.0个月,χ2=6.171,P=0.013),中位RFS也有显著差异(28.0个月vs 21.0个月,χ2=5.211,P=0.022)。对于肿瘤直径≤5 cm的RHCC,两组的中位OS有显著差异(33.0个月vs 27.0个月,χ2=6.447,P=0.011)。 结论 对于早期复发或肿瘤直径>5 cm的RHCC,RR与TACE-MWA有相似的疗效,但对于晚期复发或肿瘤直径≤5 cm的RHCC,RR应为首选。 Abstract:Objective To investigate the clinical effect of transcatheter arterial chemoembolization (TACE) combined with microwave ablation (MWA) (TACE-MWA) versus repeat resection (RR) in the treatment of recurrent hepatocellular carcinoma (RHCC). Methods A total of 178 patients with RHCC who were admitted to The Second People's Hospital of Neijiang from June 2015 to September 2020 were enrolled, and according to the treatment modality, they were divided into RR group with 64 patients and TACE-MWA group with 114 patients. Baseline demographic data, liver function, and tumor conditions before treatment were recorded, and the patients were followed up to October 2021 to compare postoperative overall survival (OS) time and recurrence-free survival (RFS) time between the two groups. Subgroup analysis based on recurrence pattern (recurrence time and tumor size) was performed, and the influencing factors for prognosis were analyzed. The independent samples t-test was used for comparison of continuous data between groups, and the chi-square test was used for comparison of categorical data; the Kaplan-Meier method was used for postoperative survival rate, the Log-rank test was used for survival difference analysis, and a multivariate Cox regression analysis was used to investigate independent risk factors for survival. Results The multivariate analysis showed that tumor diameter, alpha-fetoprotein (AFP) level, alanine aminotransferase, albumin, and time to recurrence were independent prognostic factors for OS (all P < 0.05), and AFP level and time to recurrence were independent prognostic factors for RFS (both P < 0.05). For RHCC with late recurrence (> 2 years), there were significant differences between the two groups in median OS (54.0 months vs 36.0 months, χ2=6.171, P=0.013) and median RFS (28.0 months vs 21.0 months, χ2= 5.211, P=0.022). For RHCC with a tumor diameter of ≤5 cm, there was a significant difference in median OS between the two groups (33.0 months vs 27.0 months, χ2=6.447, P=0.011). Conclusion RR has a similar clinical effect to TACE-MWA in RHCC with early recurrence or a tumor diameter of > 5 cm, but RR should be the first choice for RHCC with late recurrence or a tumor diameter of ≤5 cm. -
表 1 两组基线资料比较
Table 1. Comparison of baseline data between the two groups
项目 RR组(n=64) TACE-MWA组(n=114) 统计值 P值 年龄(岁) 54.26±8.99 53.27±10.39 t=0.635 0.526 男性[例(%)] 55(85.9) 106(93.0) χ2=2.355 0.104 HBV感染[例(%)] 59(92.2) 103(90.4) χ2=0.169 0.681 TBil(μmol/L) 39.055±13.384 36.738±12.230 t=1.143 0.243 AST(U/L) 66.930±25.791 69.720±29.467 t=-0.633 0.527 ALT(U/L) 70.183±26.244 71.210±31.624 t=-0.221 0.817 Alb(g/L) 38.853±3.910 38.752±5.175 t=0.137 0.891 PLT(×109/L) 177.912±71.565 194.029±66.438 t=-1.510 0.133 INR 1.334±0.189 1.384±0.170 t=-1.723 0.088 肿瘤直径(cm) 5.121±2.709 4.582±2.089 t=1.480 0.171 肿瘤个数[例(%)] χ2=3.383 0.066 单发 49(76.6) 72(68.0) 多发 15(23.4) 42(42.0) AFP[例(%)] χ2=0.387 0.534 >400 ng/mL 25(39.1) 50(43.9) ≤400 ng/mL 39(60.9) 64(56.1) Child-Pugh分级[例(%)] χ2=1.369 0.242 A 47(73.4) 74(64.9) B 17(26.6) 40(35.1) BCLC分期[例(%)] χ2=0.472 0.492 A 48(75.0) 80(70.2) B 16(25.0) 34(29.8) 距离初次切除后的复发时间(年) χ2=0.090 0.764 ≤2 35(54.7) 65(57.0) >2 29(45.3) 49(43.0) 表 2 两组并发症比较
Table 2. Comparison of complications between the two groups
项目 RR组(n=64) TACE-MWA组(n=114) χ2值 P值 轻微并发症(等级Ⅰ/Ⅱ)[例(%)] 发热 21(32.8) 45(39.5) 0.780 0.377 疼痛 23(35.9) 32(28.1) 1.188 0.276 呕吐 13(20.3) 35(30.7) 2.246 0.134 胃肠道反应 14(21.9) 13(11.4) 3.493 0.062 严重并发症(等级Ⅲ/Ⅳ/Ⅴ)[例(%)] 12(18.8) 11(9.6) 3.017 0.082 胆瘘 1(1.6) 0 出血 1(1.6) 0 腹水/胸水 6(9.4) 3(2.6) 肝功能衰竭 4(6.3) 5(4.4) 治疗相关死亡 0 3(2.6) 表 3 影响RHCC患者OS的单因素和多因素分析
Table 3. Univariate and multivariate analysis of OS in patients with RHCC
项目 单因素分析 多因素分析 HR(95%CI) P值 HR(95%CI) P值 性别(男vs女) 0.674(0.359~1.264) 0.219 年龄(>60岁vs ≤60岁) 0.671(0.437~1.031) 0.069 HBV感染(是vs否) 0.712(0.369~1.373) 0.311 肿瘤直径(>5 cm vs ≤5 cm) 1.832(1.228~2.733) 0.003 2.299(1.504~3.513) <0.001 多发肿瘤(是vs否) 0.355(0.234~0.538) <0.001 0.611(0.370~1.007) 0.053 Child-Pugh分级(A vs B) 0.470(0.313~0.706) <0.001 0.693(0.451~1.065) 0.094 BCLC分期(A vs B) 0.424(0.279~0.642) <0.001 1.110(0.662~1.861) 0.691 AFP>400 ng/mL(是vs否) 0.228(0.143~0.362) <0.001 0.368(0.212~0.637) <0.001 TBil(>34.2 μmol/L vs ≤34.2 μmol/L) 1.038(0.694~1.551) 0.856 AST (>40 U/L vs ≤40 U/L) 0.995(0.602~1.646) 0.985 ALT (>40 U/L vs ≤40 U/L) 2.891(1.768~4.727) <0.001 3.395(1.978~5.827) <0.001 Alb (>35.0 g/L vs ≤35.0 g/L) 2.565(1.587~4.146) <0.001 2.824(1.718~4.641) <0.001 复发时间(>2年vs ≤2年) 5.854(3.599~9.522) <0.001 3.098(1.777~5.403) <0.001 治疗方式(RR vs TACE-MWA) 0.682(0.453~1.029) 0.068 表 4 影响RHCC患者RFS的单因素和多因素分析
Table 4. Univariate and multivariate analysis of RFS in patients with RHCC
项目 单因素分析 多因素分析 HR(95%CI) P值 HR(95%CI) P值 性别(男vs女) 0.822(0.486~1.388) 0.463 年龄(>60岁vs ≤60岁) 0.914(0.631~1.324) 0.635 HBV感染(是vs否) 0.853(0.492~1.479) 0.571 肿瘤直径(>5 cm vs ≤5 cm) 1.427(1.037~1.964) 0.029 1.136(0.816~1.581) 0.451 多发肿瘤(是vs否) 0.479(0.339~0.676) <0.001 0.907(0.612~1.344) 0.627 Child-Pugh分级(A vs B) 0.637(0.454~0.894) 0.009 0.771(0.547~1.089) 0.140 BCLC分期(A vs B) 0.467(0.330~0.660) <0.001 0.699(0.468~1.043) 0.080 AFP>400 ng/mL(是vs否) 0.321(0.223~0.461) <0.001 0.533(0.356~0.796) 0.002 TBil(>34.2 μmol/L vs ≤34.2 μmol/L) 1.044(0.757~1.440) 0.792 AST(>40 U/L vs ≤40 U/L) 1.318(0.886~1.961) 0.173 ALT(>40 U/L vs ≤40 U/L) 1.841(1.225~2.767) 0.003 1.382(0.911~2.097) 0.128 Alb(>35.0 g/L vs ≤35.0 g/L) 1.498(1.005~2.231) 0.047 1.142(0.760~1.716) 0.523 复发时间(>2年vs ≤2年) 6.158(4.179~9.076) <0.001 4.327(2.800~6.687) <0.001 治疗方式(RR vs TACE-MWA) 0.757(0.540~1.060) 0.105 -
[1] LURJE I, CZIGANY Z, BEDNARSCH J, et al. Treatment strategies for hepatocellular carcinoma—a multidisciplinary approach[J]. Int J Mol Sci, 2019, 20(6): 1465. DOI: 10.3390/ijms20061465. [2] TAKEISHI K, MAEDA T, TSUJITA E, et al. Predictors of intrahepatic multiple recurrences after curative hepatectomy for hepatocellular carcinoma[J]. Anticancer Res, 2015, 35(5): 3061-3066. https://kyushu-u.pure.elsevier.com/en/publications/predictors-of-intrahepatic-multiple-recurrences-after-curative-he [3] GUPTA S, KHAN S, KAWKA M, et al. Clinical utility of clonal origin determination in managing recurrent hepatocellular carcinoma[J]. Expert Rev Gastroenterol Hepatol, 2021, 15(10): 1159-1167. DOI: 10.1080/17474124.2021.1967144. [4] ZHENG J, CAI J, TAO L, et al. Comparison on the efficacy and prognosis of different strategies for intrahepatic recurrent hepatocellular carcinoma: A systematic review and Bayesian network meta-analysis[J]. Int J Surg, 2020, 83: 196-204. DOI: 10.1016/j.ijsu.2020.09.031. [5] RIZZO A, RICCI AD, BRANDI G. Immune-based combinations for advanced hepatocellular carcinoma: shaping the direction of first-line therapy[J]. Future Oncol, 2021, 17(7): 755-757. DOI: 10.2217/fon-2020-0986. [6] WANG X, LIANG H, LU Z. Efficacy of transarterial chemoembolization compared with radiofrequency ablation for the treatment of recurrent hepatocellular carcinoma after radiofrequency ablation[J]. Minim Invasive Ther Allied Technol, 2020, 29(6): 344-352. DOI: 10.1080/13645706.2019.1649286. [7] JIN YJ, LEE JW, LEE OH, et al. Transarterial chemoembolization versus surgery/radiofrequency ablation for recurrent hepatocellular carcinoma with or without microvascular invasion[J]. J Gastroenterol Hepatol, 2014, 29(5): 1056-1064. DOI: 10.1111/jgh.12507. [8] TUNG-PING POON R, FAN ST, WONG J. Risk factors, prevention, and management of postoperative recurrence after resection of hepatocellular carcinoma[J]. Ann Surg, 2000, 232(1): 10-24. DOI: 10.1097/00000658-200007000-00003. [9] PENG ZW, ZHANG YJ, LIANG HH, et al. Recurrent hepatocellular carcinoma treated with sequential transcatheter arterial chemoembolization and RF ablation versus RF ablation alone: a prospective randomized trial[J]. Radiology, 2012, 262(2): 689-700. DOI: 10.1148/radiol.11110637. [10] European Association for Study of Liver; European Organisation for Research and Treatment of Cancer. EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma[J]. Eur J Cancer, 2012, 48(5): 599-641. DOI: 10.1016/j.ejca.2011.12.021. [11] HEIMBACH JK, KULIK LM, FINN RS, et al. AASLD guidelines for the treatment of hepatocellular carcinoma[J]. Hepatology, 2018, 67(1): 358-380. DOI: 10.1002/hep.29086. [12] EISENHAUER EA, THERASSE P, BOGAERTS J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1)[J]. Eur J Cancer, 2009, 45(2): 228-247. DOI: 10.1016/j.ejca.2008.10.026. [13] DINDO D, DEMARTINES N, CLAVIEN PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey[J]. Ann Surg, 2004, 240(2): 205-213. DOI: 10.1097/01.sla.0000133083.54934.ae. [14] CHO JY, HAN HS, CHOI Y, et al. Association of remnant liver ischemia with early recurrence and poor survival after liver resection in patients with hepatocellular carcinoma[J]. JAMA Surg, 2017, 152(4): 386-392. DOI: 10.1001/jamasurg.2016.5040. [15] CALDERARO J, PETITPREZ F, BECHT E, et al. Intra-tumoral tertiary lymphoid structures are associated with a low risk of early recurrence of hepatocellular carcinoma[J]. J Hepatol, 2019, 70(1): 58-65. DOI: 10.1016/j.jhep.2018.09.003. [16] JUNG SM, KIM JM, CHOI GS, et al. Characteristics of early recurrence after curative liver resection for solitary hepatocellular carcinoma[J]. J Gastrointest Surg, 2019, 23(2): 304-311. DOI: 10.1007/s11605-018-3927-2. [17] ZHANG X, LI C, WEN T, et al. Appropriate treatment strategies for intrahepatic recurrence after curative resection of hepatocellular carcinoma initially within the Milan criteria: according to the recurrence pattern[J]. Eur J Gastroenterol Hepatol, 2015, 27(8): 933-940. DOI: 10.1097/MEG.0000000000000383. [18] PENG Z, CHEN S, WEI M, et al. Advanced recurrent hepatocellular carcinoma: Treatment with sorafenib alone or in combination with transarterial chemoembolization and radiofrequency ablation[J]. Radiology, 2018, 287(2): 705-714. DOI: 10.1148/radiol.2018171541. [19] LIU Y, REN Y, GE S, et al. Transarterial chemoembolization in treatment-naïve and recurrent hepatocellular carcinoma: a propensity-matched outcome and risk signature analysis[J]. Front Oncol, 2021, 11: 662408. DOI: 10.3389/fonc.2021.662408. [20] TANG C, SHEN J, FENG W, et al. Combination therapy of radiofrequency ablation and transarterial chemoembolization for unresectable hepatocellular carcinoma: A retrospective study[J]. Medicine (Baltimore), 2016, 95(20): e3754. DOI: 10.1097/MD.0000000000003754. [21] YUAN W, YANG MJ, XU J, et al. Radiofrequency ablation combined with transarterial chemoembolization for specially located small hepatocellular carcinoma[J]. Technol Cancer Res Treat, 2018, 17: 1533033818788529. DOI: 10.1177/1533033818788529. [22] YAN L, REN Y, QIAN K, et al. Sequential transarterial chemoembolization and early radiofrequency ablation improves clinical outcomes for early-intermediate hepatocellular carcinoma in a 10-year single-center comparative study[J]. BMC Gastroenterol, 2021, 21(1): 182. DOI: 10.1186/s12876-021-01765-x. [23] HASSANIN TM, FOUAD Y, HASSNINE A, et al. Quality of life after transcatheter arterial chemoembolization combined with radiofrequency ablation in patients with unresectable hepatocellular carcinoma compared with transcatheter arterial chemoembolization alone[J]. Asian Pac J Cancer Prev, 2021, 22(4): 1255-1261. DOI: 10.31557/APJCP.2021.22.4.1255. [24] JIANG C, CHENG G, LIAO M, et al. Individual or combined transcatheter arterial chemoembolization and radiofrequency ablation for hepatocellular carcinoma: a time-to-event meta-analysis[J]. World J Surg Oncol, 2021, 19(1): 81. DOI: 10.1186/s12957-021-02188-4. [25] MATSUDA M, FUJⅡ H, KONO H, et al. Surgical treatment of recurrent hepatocellular carcinoma based on the mode of recurrence: repeat hepatic resection or ablation are good choices for patients with recurrent multicentric cancer[J]. J Hepatobiliary Pancreat Surg, 2001, 8(4): 353-359. DOI: 10.1007/s005340170008. [26] HUANG ZY, LIANG BY, XIONG M, et al. Long-term outcomes of repeat hepatic resection in patients with recurrent hepatocellular carcinoma and analysis of recurrent types and their prognosis: a single-center experience in China[J]. Ann Surg Oncol, 2012, 19(8): 2515-2525. DOI: 10.1245/s10434-012-2269-7. [27] ZHENG X, REN Y, HU H, et al. Transarterial chemoembolization combined with radiofrequency ablation versus repeat hepatectomy for recurrent hepatocellular carcinoma after curative resection: A 10-year single-center comparative study[J]. Front Oncol, 2021, 11: 713432. DOI: 10.3389/fonc.2021.713432. [28] BRUIX J, TAKAYAMA T, MAZZAFERRO V, et al. Adjuvant sorafenib for hepatocellular carcinoma after resection or ablation (STORM): a phase 3, randomised, double-blind, placebo-controlled trial[J]. Lancet Oncol, 2015, 16(13): 1344-1354. DOI: 10.1016/S1470-2045(15)00198-9. [29] LUO P, WU S, YU Y, et al. Current status and perspective biomarkers in AFP negative HCC: Towards screening for and diagnosing hepatocellular carcinoma at an earlier stage[J]. Pathol Oncol Res, 2020, 26(2): 599-603. DOI: 10.1007/s12253-019-00585-5. [30] ZHENG Y, ZHU M, LI M. Effects of alpha-fetoprotein on the occurrence and progression of hepatocellular carcinoma[J]. J Cancer Res Clin Oncol, 2020, 146(10): 2439-2446. DOI: 10.1007/s00432-020-03331-6. [31] CHEN ZH, ZHANG XP, ZHOU TF, et al. Adjuvant transarterial chemoembolization improves survival outcomes in hepatocellular carcinoma with microvascular invasion: A systematic review and meta-analysis[J]. Eur J Surg Oncol, 2019, 45(11): 2188-2196. DOI: 10.1016/j.ejso.2019.06.031. -



 PDF下载 ( 3683 KB)
PDF下载 ( 3683 KB)

 下载:
下载: