二甲双胍对布加综合征小鼠模型肝纤维化的影响
DOI: 10.3969/j.issn.1001-5256.2022.09.017
-
摘要:
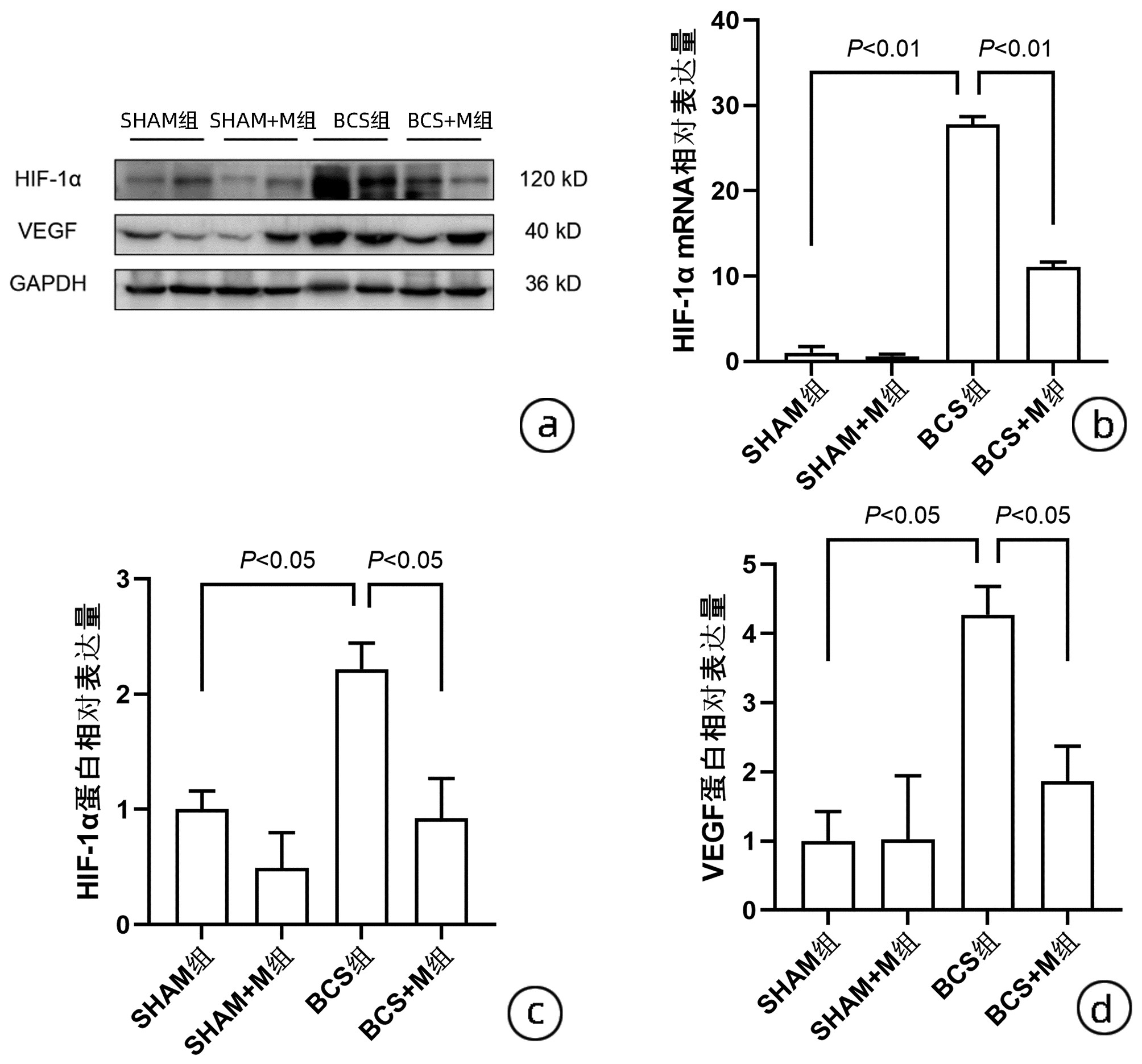
目的 探讨二甲双胍对于布加综合征小鼠模型肝脏纤维化的影响及其机制。 方法 C57雄性小鼠30只随机分为4组:假手术组(SHAM组)6只,假手术+二甲双胍组(SHAM+M组)5只,布加综合征模型组(BCS组)10只,布加综合征模型+二甲双胍组(BCS+M组)9只。模型组行下腔静脉部分结扎术,SHAM组不结扎,二甲双胍组造模同时饮水中给予0.1%二甲双胍。6周后处死小鼠取材,HE染色、天狼猩红染色观察小鼠肝脏组织病理和胶原沉积变化,免疫组化检测α-平滑肌肌动蛋白(α-SMA)、纤维蛋白原(Fibrinogen)表达情况,实时荧光定量PCR检测缺氧诱导因子-1α(HIF-1α)、Ⅰ型胶原(Collagen1)的mRNA表达,Western Blot检测HIF-1α、血管内皮生长因子(VEGF)、Fibrinogen、α-SMA、Collagen1蛋白的相对表达量。计量资料多组间比较采用单因素方差分析,进一步两两比较采用LSD-t检验。 结果 与SHAM组相比,病理染色结果显示BCS组肝脏明显纤维化,中央静脉旁肝细胞排列紊乱,肝窦扩张并伴有红细胞淤积和少量炎症细胞浸润,胶原沉积。BCS组HIF-1α、Collagen1 mRNA表达增加,α-SMA、Collagen1、HIF-1α、VEGF、Fibrinogen蛋白表达明显增加(P值均<0.05);与BCS组相比,BCS+M组肝脏纤维化减轻,红细胞淤积减少,胶原沉积减少,HIF-1α、Collagen1 mRNA表达降低,α-SMA、Collagen1、HIF-1α、VEGF、Fibrinogen蛋白表达降低(P值均<0.05)。 结论 二甲双胍能够改善由布加综合征所导致的淤血性肝纤维化,其机制可能为减少肝窦内微血栓,抑制HIF-1α/VEGF通路来发挥作用。 -
关键词:
- Budd-Chiari综合征 /
- 肝硬化 /
- 二甲双胍 /
- 小鼠, 近交C57BL
Abstract:Objective To investigate the effect of metformin on liver fibrosis in a mouse model of Budd-Chiari syndrome and its mechanism. Methods A total of 30 male C57 mice were randomly divided into sham-operation group (SHAM group) with 6 mice, sham operation+ metformin group (SHAM+M group) with 5 mice, Budd-Chiari model group (BCS group) with 10 mice, and Budd-Chiari model+metformin group (BCS+M group) with 9 mice. The mice in the model group were treated with partial ligation of the inferior vena cava, those in the SHAM group were not treated with ligation, and those in the metformin group were given 0.1% metformin in drinking water besides modeling. The mice were sacrificed after 6 weeks. HE staining and picrosirius red staining were used to observe liver histopathology and collagen deposition; immunohistochemistry was used to measure the expressions of α-smooth muscle actin (α-SMA) and fibrinogen; quantitative real-time PCR was used to measure the mRNA expression of hypoxia-inducible factor 1α (HIF-1α) and type Ⅰ collagen (collagen 1), and Western blot was used to measure the relative protein expression levels of HIF-1α, vascular endothelial growth factor (VEGF), fibrinogen, α-SMA, and collagen 1. A one-way analysis of variance was used for comparison of continuous data between multiple groups, and the least significant difference t-test was used for further comparison between two groups. Results Pathological staining showed that compared with the SHAM group, the BCS group had significant liver fibrosis, disordered arrangement of hepatocytes near the central vein, sinusoidal expansion with red blood cell deposition and a small amount of inflammatory cell infiltration, and collagen deposition. The BCS group had significant increases in the mRNA expression levels of HIF-1α and collagen 1 and the protein expression levels of α-SMA, collagen 1, HIF-1α, VEGF, and fibrinogen (all P < 0.05); compared with the BCS group, the BCS+M group had significant alleviation of liver fibrosis, red blood cell deposition, and collagen deposition and significant reductions in the mRNA expression levels of HIF-1α and collagen 1 and the protein expression levels of α-SMA, collagen 1, HIF-1α, VEGF, and fibrinogen (all P < 0.05). Conclusion Metformin can improve congestive liver fibrosis caused by Budd-Chiari syndrome, possibly by reducing microthrombus in hepatic sinusoid and inhibiting the HIF-1α/VEGF pathway. -
Key words:
- Budd-Chiari Syndrome /
- Liver Cirrhosis /
- Metformin /
- Mice, Inbred C57BL
-
表 1 qPCR引物序列
Table 1. The primers used for qPCR analysis
引物名称 序列(5′-3′) GAPDH F AGGTCGGTGTGAACGGATTTG GAPDH R TGTAGACCATGTAGTTGAGGTCA Collagen1 F GCTCCTCTTAGGGGCCACT Collagen1 R CCACGTCTCACCATTGGGG HIF-1α F ACCTTCATCGGAAACTCCAAAG HIF-1α R ACTGTTAGGCTCAGGTGAACT -
[1] WANG XY, XU WT, DANG XW, et al. An excerpt of Budd-Chiari syndrome: Consensus guidance of the Asian Pacific Association for the Study of the Liver (APASL)[J]. J Clin Hepatol, 2021, 37(11): 2555-2557. DOI: 10.3969/j.issn.1001-5256.2021.11.014.王雪莹, 许文涛, 党晓卫, 等. 《2021年亚太肝病学会共识指导: 布-加综合征》摘译[J]. 临床肝胆病杂志, 2021, 37(11): 2555-2557. DOI: 10.3969/j.issn.1001-5256.2021.11.014. [2] KHAN F, ARMSTRONG MJ, MEHRZAD H, et al. Review article: a multidisciplinary approach to the diagnosis and management of Budd-Chiari syndrome[J]. Aliment Pharmacol Ther, 2019, 49(7): 840-863. DOI: 10.1111/apt.15149. [3] SIMONETTO DA, YANG HY, YIN M, et al. Chronic passive venous congestion drives hepatic fibrogenesis via sinusoidal thrombosis and mechanical forces[J]. Hepatology, 2015, 61(2): 648-659. DOI: 10.1002/hep.27387. [4] FOGLIA B, NOVO E, PROTOPAPA F, et al. Hypoxia, hypoxia-inducible factors and liver fibrosis[J]. Cells, 2021, 10(7): 1764. DOI: 10.3390/cells10071764. [5] LI ZG, YANG XZ, LI XK, et al. Association of hypoxic microenvironment with the development and progression of liver diseases[J]. J Clin Hepatol, 2020, 36(8): 1891-1895. DOI: 10.3969/j.issn.1001-5256.2020.08.047.李志国, 杨先照, 李小科, 等. 缺氧微环境与肝病发生发展的关系[J]. 临床肝胆病杂志, 2020, 36(8): 1891-1895. DOI: 10.3969/j.issn.1001-5256.2020.08.047. [6] FLORY J, LIPSKA K. Metformin in 2019[J]. JAMA, 2019, 321(19): 1926-1927. DOI: 10.1001/jama.2019.3805. [7] LIU HY, CHENG G. Meta analysis of efficacy and safety of sitagliptin and glimepiride combined with metformin in the treatment of type 2 diabetes[J]. Clin J Med Offic, 2020, 48(2): 185-188. DOI: 10.16680/j.1671-3826.2020.02.20.刘慧莹, 程刚. 西格列汀、格列美脲分别联合二甲双胍治疗2型糖尿病有效性与安全性Meta分析[J]. 临床军医杂志, 2020, 48(2): 185-188. DOI: 10.16680/j.1671-3826.2020.02.20. [8] HE L. Metformin and systemic metabolism[J]. Trends Pharmacol Sci, 2020, 41(11): 868-881. DOI: 10.1016/j.tips.2020.09.001. [9] KULKARNI AS, GUBBI S, BARZILAI N. Benefits of metformin in attenuating the hallmarks of aging[J]. Cell Metab, 2020, 32(1): 15-30. DOI: 10.1016/j.cmet.2020.04.001. [10] CHEN GG, WOO P, NG S, et al. Impact of metformin on immunological markers: Implication in its anti-tumor mechanism[J]. Pharmacol Ther, 2020, 213: 107585. DOI: 10.1016/j.pharmthera.2020.107585. [11] FAN K, WU K, LIN L, et al. Metformin mitigates carbon tetrachloride-induced TGF-β1/Smad3 signaling and liver fibrosis in mice[J]. Biomed Pharmacother, 2017, 90: 421-426. DOI: 10.1016/j.biopha.2017.03.079. [12] NGUYEN G, PARK SY, LE CT, et al. Metformin ameliorates activation of hepatic stellate cells and hepatic fibrosis by succinate and GPR91 inhibition[J]. Biochem Biophys Res Commun, 2018, 495(4): 2649-2656. DOI: 10.1016/j.bbrc.2017.12.143. [13] SHE L, XU D, WANG Z, et al. Curcumin inhibits hepatic stellate cell activation via suppression of succinate-associated HIF-1α induction[J]. Mol Cell Endocrinol, 2018, 476: 129-138. DOI: 10.1016/j.mce.2018.05.002. [14] WELLS ML, FENSTAD ER, POTERUCHA JT, et al. Imaging findings of congestive hepatopathy[J]. Radiographics, 2016, 36(4): 1024-1037. DOI: 10.1148/rg.2016150207. [15] LI Z, DING Q, LING LP, et al. Metformin attenuates motility, contraction, and fibrogenic response of hepatic stellate cells in vivo and in vitro by activating AMP-activated protein kinase[J]. World J Gastroenterol, 2018, 24(7): 819-832. DOI: 10.3748/wjg.v24.i7.819. [16] ZHOU D, ZHANG LT, LI JF, et al. Mechanism of action of hypoxia-inducible factor 1α in the development and progression of liver fibrosis[J]. J Clin Hepatol, 2019, 35(7): 1604-1607. DOI: 10.3969/j.issn.1001-5256.2019.07.039.周丹, 张立婷, 李俊峰, 等. 缺氧诱导因子1α在肝纤维化发生发展中的作用机制[J]. 临床肝胆病杂志, 2019, 35(7): 1604-1607. DOI: 10.3969/j.issn.1001-5256.2019.07.039. -



 PDF下载 ( 6734 KB)
PDF下载 ( 6734 KB)


 下载:
下载: