整合素α4在美洲大蠊提取物黏糖氨酸抗肝纤维化中的作用
DOI: 10.3969/j.issn.1001-5256.2022.09.016
The role of integrin α4 in the anti-liver fibrosis effect of the sticky sugar amino acid extract of Periplaneta americana
-
摘要:
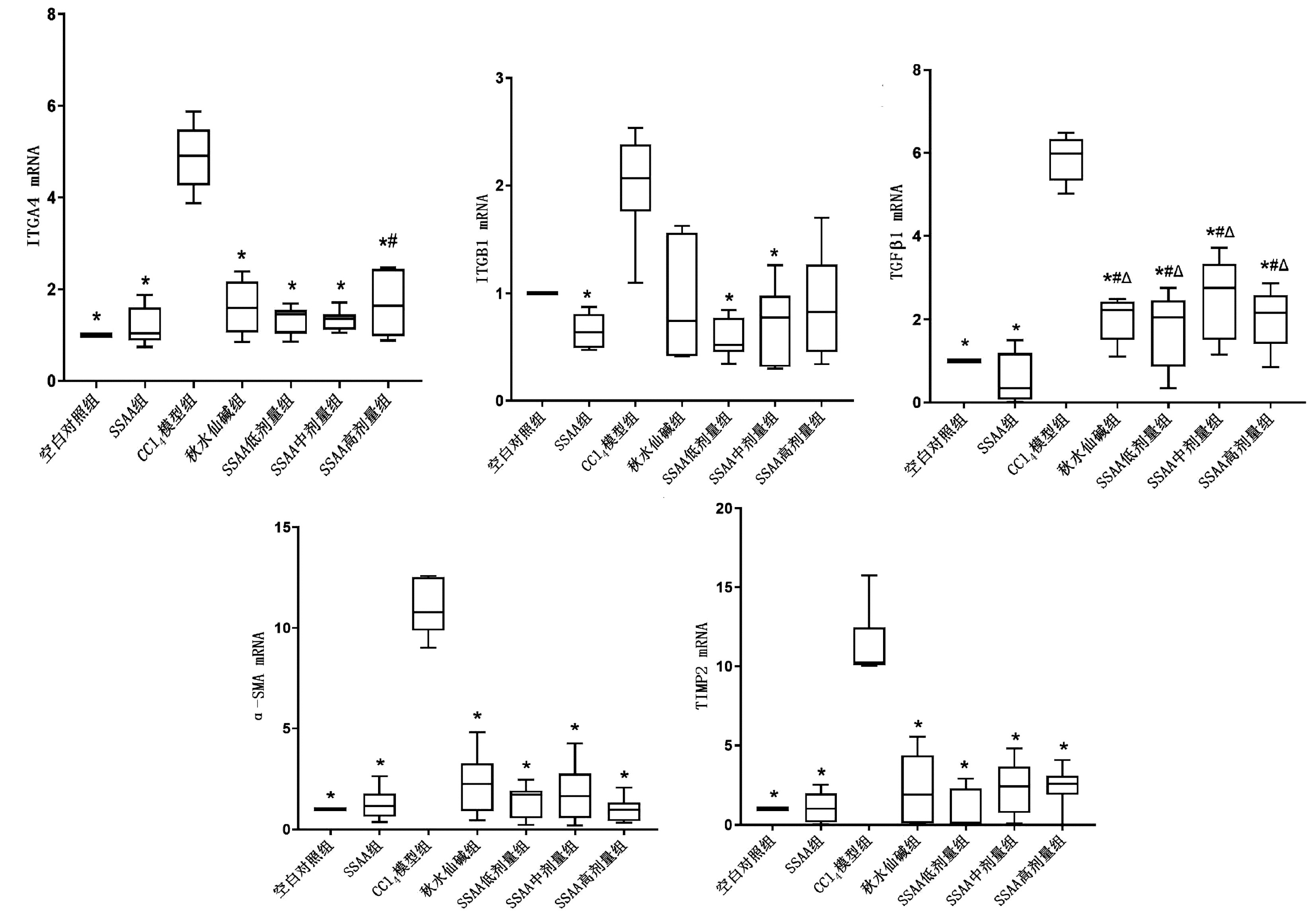
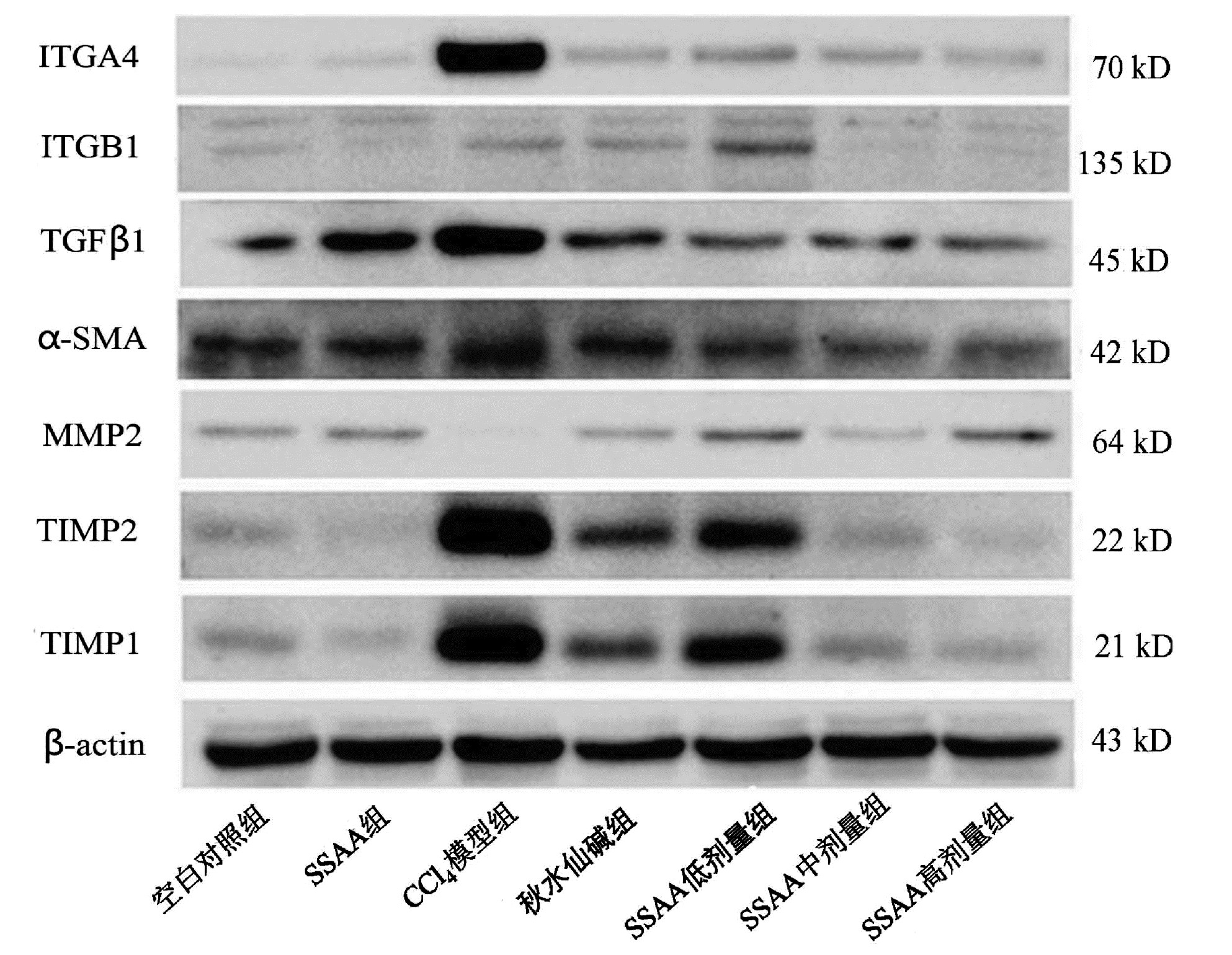
目的 本研究基于黏糖氨酸(SSAA)在大鼠中的抗肝纤维化作用,研究整合素α4(ITGA4)在肝纤维化中的作用机制。 方法 大鼠腹腔内注射CCl4诱导肝纤维化模型,在此基础上使用秋水仙碱或SSAA低、中、高剂量进行干预,以空白对照组、SSAA组作为对照。实验干预12周后,收集大鼠血清、肝脏标本,检测大鼠血清ALT、AST水平,HE染色、天蓝猩红染色观察肝组织病理情况;实时荧光定量PCR检测肝组织ITGA4、整合素β1(ITGB1)、TGFβ1、α-SMA、TIMP2的转录水平;Western Blot检测ITGA4、ITGB1、TGFβ1、α-SMA、MMP2、TIMP1、TIMP2的相对蛋白表达;免疫组化观察TGFβ1、α-SMA蛋白表达。计量资料多组间比较采用单因素方差分析,进一步两两比较采用LSD-t检验。 结果 CCl4模型组中AST、ALT显著升高,秋水仙碱或SSAA低、中、高剂量干预后能降低AST、ALT水平,各组与CCl4模型组相比差异均有统计学意义(P值均<0.05)。CCl4模型组中的HE染色和天蓝猩红染色结果显示肝小叶结构紊乱、胶原纤维增多,秋水仙碱或SSAA低、中、高剂量干预后肝小叶结构得到改善。CCl4模型组中的ITGA4、TGFβ1、α-SMA和TIMP2各指标转录水平均显著高于其余各组,秋水仙碱或SSAA干预后能降低各因子转录水平,与CCl4模型组相比差异均有统计学意义(P值均<0.05)。CCl4模型组中的ITGA4、TGFβ1、α-SMA、TIMP2和TIMP1蛋白表达高于其余各组,MMP2蛋白表达低于其余各组,在秋水仙碱或SSAA干预后均能抑制ITGA4、TGFβ1、α-SMA、TIMP2、TIMP1表达,促进MMP2表达。免疫组化结果提示CCl4模型组的TGFβ1、α-SMA的表达显著高于其余各组,秋水仙碱或SSAA干预后均能抑制表达。SSAA高剂量组在降低转氨酶、改善肝小叶结构和抑制致肝纤维化因子蛋白表达上效果最显著。 结论 肝脏中ITGA4的高表达与肝纤维化的发生相关,与TGFβ1、α-SMA表达升高一致,抑制ITGA4表达可为抗肝纤维化提供更多治疗靶点,同时拓展了SSAA的抗肝纤维化作用机制。 -
关键词:
- 肝硬化 /
- 黏糖氨酸 /
- 整合素类 /
- 大鼠, Sprague-Dawley
Abstract:Objective To investigate the mechanism of action of integrin α4 (ITGA4) in liver fibrosis based on the anti-liver fibrosis effect of sticky sugar amino acid (SSAA) in rats. Methods A rat model of liver fibrosis was induced by intraperitoneal injection of CCl4, and then colchicine and low-, middle-, and high-dose SSAA were used for intervention, with blank control group and SSAA group as control. After 12 weeks of experimental intervention, serum and liver samples were collected to measure the serum levels of alanine aminotransferase (ALT) and aspartate aminotransferase (AST), and HE staining and Sirius Red staining were used to observe the pathological conditions of liver tissue; quantitative real-time PCR was used to measure the transcriptional level of ITGA4, integrin β1 (ITGB1), transforming growth factor-β1 (TGFβ1), alpha-smooth muscle actin (α-SMA), and TIMP2 in liver tissue; Western blot was used to measure the relative protein expression levels of ITGA4, ITGB1, TGFβ1, α-SMA, MMP2, TIMP1, and TIMP2; immunohistochemistry was used to observe the protein expression of TGFβ1 and α-SMA. A one-way analysis of variance was used for comparison of continuous data between multiple groups, and the least significant difference t-test was used for comparison between two groups. Results There were significant increases in AST and ALT in the CCl4 model group, and intervention with colchicine or low-, middle-, and high-dose SSAA reduced the levels of AST and ALT, with a significant difference between the CCl4 model group and the other groups (all P < 0.05). HE staining and Sirius Red staining showed disordered structure of hepatic lobules and an increase in collagen fibers in the CCl4 model group, and the structure of hepatic lobules was improved after intervention with colchicine or low-, middle-, and high-dose SSAA. The CCl4 model group had significantly higher transcriptional levels of ITGA4, TGFβ1, α-SMA, and TIMP2 than the other groups, and there were significant reductions in the transcriptional levels of each factor after intervention with colchicine or SSAA, with a significant difference between the CCl4 model group and the other groups (all P < 0.05). The CCl4 model group had significantly higher protein expression levels of ITGA4, TGFβ1, α-SMA, TIMP2, and TIMP1 and a significantly lower protein expression level of MMP2 than the other groups, and intervention with colchicine or SSAA inhibited the expression of ITGA4, TGFβ1, α-SMA, TIMP2, and TIMP1 and promoted the expression of MMP2. Immunohistochemistry showed that the CCl4 model group had significantly higher expression levels of TGFβ1 and α-SMA than the other groups, which was inhibited by intervention with colchicine or SSAA. The high-dose SSAA group had the most significant effect in reducing aminotransferases, improving lobular structure, and inhibiting the protein expression of liver fibrosis factors. Conclusion The high expression of ITGA4 in the liver is associated with the development of liver fibrosis, which is consistent with the increases in the expression of TGFβ1 and α-SMA. Inhibiting the expression of ITGA4 can provide more therapeutic targets for liver fibrosis and expand the anti-liver fibrosis mechanism of SSAA. -
Key words:
- Liver Cirrhosis /
- Mucoglycosine /
- Integrins /
- Rats, Sprague-Dawley
-
表 1 PCR引物
Table 1. PCR primer
基因 引物序列(5′-3′) 长度(bp) ITGA4 F: ACTTCGCAAGGTTTTGTGCC 20 R: CCTGTGGGGAGTTGGACATT 20 ITGB1 F: TGTCCTACTGGTCCCGACAT 20 R: TTTTCACCCGTGTCCCACTT 20 TGFβ1 F: AGGGCTACCATGCCAACTTC 20 R: CCACGTAGTAGACGATGGGC 20 α-SMA F: CATCACCAACTGGGACGACA 20 R: TCCGTTAGCAAGGTCGGATG 20 TIMP2 F: ATGGCAACCCCATCAAGAGG 20 R: TCTTTCCTCCAACGTCCAGC 20 注:F,正向引物;R,反向引物。 表 2 各组ALT、AST水平
Table 2. Transaminase levels in each group
组别 ALT(U/L) AST(U/L) 空白对照组 41.7±8.93) 71.1±4.93) SSAA组 41.3±10.93) 69.0±10.93) CCl4模型组 236.6±131.2 568.3±416.2 秋水仙碱组 91.7±31.21)2)3) 139.7±48.33) SSAA低剂量组 127.9±43.81)2)3) 191.6±112.73) SSAA中剂量组 105.5±39.81)2)3)4) 141.8±97.23) SSAA高剂量组 81.5±36.53) 120.6±48.43) F值 30.324 12.249 P值 <0.001 <0.001 注:与空白对照组相比,1)P<0.05;与SSAA组相比,2)P<0.05;与CCl4模型组相比,3)P<0.05;与SSAA低剂量组相比,4)P<0.05。 -
[1] LI Y, PU S, LIU Q, et al. An integrin-based nanoparticle that targets activated hepatic stellate cells and alleviates liver fibrosis[J]. J Control Release, 2019, 303: 77-90. DOI: 10.1016/j.jconrel.2019.04.022. [2] PULKKA OP, MPINDI JP, TYNNINEN O, et al. Clinical relevance of integrin alpha 4 in gastrointestinal stromal tumours[J]. J Cell Mol Med, 2018, 22(4): 2220-2230. DOI: 10.1111/jcmm.13502. [3] HINTERMANN E, CHRISTEN U. The many roles of cell adhesion molecules in hepatic fibrosis[J]. Cells, 2019, 8(12): 1503. DOI: 10.3390/cells8121503. [4] SFERRA R, VETUSCHI A, POMPILI S, et al. Expression of pro-fibrotic and anti-fibrotic molecules in dimethylnitrosamine-induced hepatic fibrosis[J]. Pathol Res Pract, 2017, 213(1): 58-65. DOI: 10.1016/j.prp.2016.11.004. [5] SHAO T, CHEN Z, BELOV V, et al. [18F]-Alfatide PET imaging of integrin αvβ3 for the non-invasive quantification of liver fibrosis[J]. J Hepatol, 2020, 73(1): 161-169. DOI: 10.1016/j.jhep.2020.02.018. [6] BERNSMEIER C, van DER MERWE S, PÉRIANIN A. Innate immune cells in cirrhosis[J]. J Hepatol, 2020, 73(1): 186-201. DOI: 10.1016/j.jhep.2020.03.027. [7] SHAO MY, LAI Y. Effects of Ganlong Capsules combined with silibinin on the expression of Col-Ⅰ and TIMP-1 genes in liver tissue of hepatic fibrosis rats[J]. Advances in Veterinary Med, 2018, 39(10): 50-55. DOI: 10.16437/j.cnki.1007-5038.2018.10.010.邵明园, 赖泳. 肝龙胶囊联合水飞蓟宾对肝纤维化大鼠肝组织Col-Ⅰ和TIMP-1基因表达的影响[J]. 动物医学进展, 2018, 39(10): 50-55. DOI: 10.16437/j.cnki.1007-5038.2018.10.010. [8] HU YF. Efficacy and survival analysis of plasma exchange in patients with acute-on-chronic liver failure[D]. Zhengzhou: Zhengzhou University, 2020.呼怡菲. 血浆置换治疗慢加急性肝衰竭患者的疗效及生存分析[D]. 郑州: 郑州大学, 2020. [9] TSUCHIDA T, FRIEDMAN SL. Mechanisms of hepatic stellate cell activation[J]. Nat Rev Gastroenterol Hepatol, 2017, 14(7): 397-411. DOI: 10.1038/nrgastro.2017.38. [10] LACHOWSKI D, CORTES E, RICE A, et al. Matrix stiffness modulates the activity of MMP-9 and TIMP-1 in hepatic stellate cells to perpetuate fibrosis[J]. Sci Rep, 2019, 9(1): 7299. DOI: 10.1038/s41598-019-43759-6. [11] ZHOU J, LI R, ZHANG J, et al. Targeting interstitial myofibroblast-expressed integrin αvβ3 alleviates renal fibrosis[J]. Mol Pharm, 2021, 18(3): 1373-1385. DOI: 10.1021/acs.molpharmaceut.0c01182. [12] HUANG YZ, ZHAO L, ZHU Y, et al. Interrupting TGF-β1/CCN2/integrin-α5β1 signaling alleviates high mechanical-stress caused chondrocyte fibrosis[J]. Eur Rev Med Pharmacol Sci, 2021, 25(3): 1233-1241. DOI: 10.26355/eurrev_202102_24827. [13] STRUDWICK XL, ADAMS DH, PYNE NT, et al. Systemic delivery of anti-integrin αL antibodies reduces early macrophage recruitment, inflammation, and scar formation in murine burn wounds[J]. Adv Wound Care (New Rochelle), 2020, 9(12): 637-648. DOI: 10.1089/wound.2019.1035. [14] LIU W, SUN T, WANG Y. Integrin αvβ6 mediates epithelial-mesenchymal transition in human bronchial epithelial cells induced by lipopolysaccharides of Pseudomonas aeruginosa via TGF-β1-Smad2/3 signaling pathway[J]. Folia Microbiol (Praha), 2020, 65(2): 329-338. DOI: 10.1007/s12223-019-00728-w. [15] BONUS M, HÄUSSINGER D, GOHLKE H. Liver cell hydration and integrin signaling[J]. Biol Chem, 2021, 402(9): 1033-1045. DOI: 10.1515/hsz-2021-0193. [16] HIGHT-WARBURTON W, PARSONS M. Regulation of cell migration by α4 and α9 integrins[J]. Biochem J, 2019, 476(4): 705-718. DOI: 10.1042/BCJ20180415. [17] KUMMER C, PETRICH BG, ROSE DM, et al. A small molecule that inhibits the interaction of paxillin and alpha 4 integrin inhibits accumulation of mononuclear leukocytes at a site of inflammation[J]. J Biol Chem, 2010, 285(13): 9462-9469. DOI: 10.1074/jbc.M109.066993. [18] DAMMES N, GOLDSMITH M, RAMISHETTI S, et al. Conformation-sensitive targeting of lipid nanoparticles for RNA therapeutics[J]. Nat Nanotechnol, 2021, 16(9): 1030-1038. DOI: 10.1038/s41565-021-00928-x. [19] RAI RP, LIU Y, IYER SS, et al. Blocking integrin α4β7-mediated CD4 T cell recruitment to the intestine and liver protects mice from western diet-induced non-alcoholic steatohepatitis[J]. J Hepatol, 2020, 73(5): 1013-1022. DOI: 10.1016/j.jhep.2020.05.047. -



 PDF下载 ( 7805 KB)
PDF下载 ( 7805 KB)


 下载:
下载: