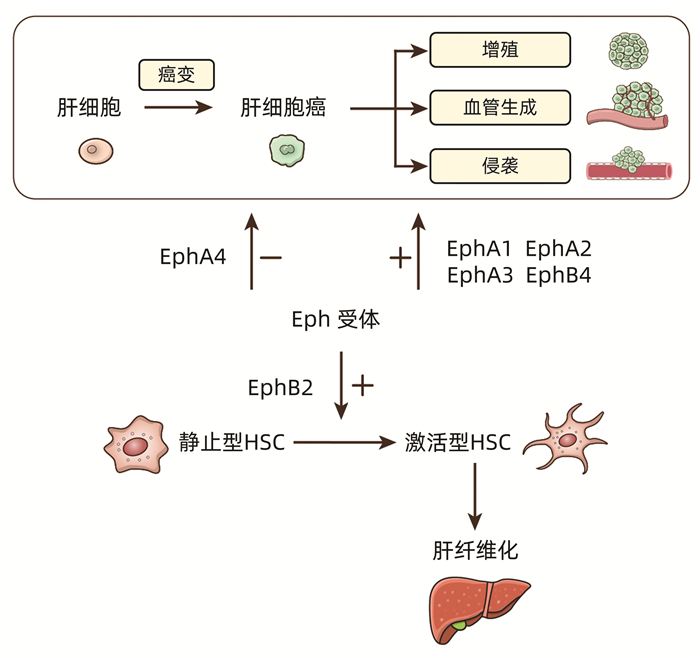
| [1] |
KANDA T, GOTO T, HIROTSU Y, et al. Molecular mechanisms driving progression of liver cirrhosis towards hepatocellular carcinoma in chronic hepatitis B and C infections: A review[J]. Int J Mol Sci, 2019, 20(6): 1358. DOI: 10.3390/ijms20061358.
|
| [2] |
WANG YC, DAI Y, XU GL, et al. Association between EphA1 and tumor microenvironment in gastric carcinoma and its clinical significance[J]. Med Sci Monit, 2020, 26: e923409. DOI: 10.12659/MSM.923409.
|
| [3] |
LI JY, XIAO T, YI HM, et al. S897 phosphorylation of EphA2 is indispensable for EphA2-dependent nasopharyngeal carcinoma cell invasion, metastasis and stem properties[J]. Cancer Lett, 2019, 444: 162-174. DOI: 10.1016/j.canlet.2018.12.011.
|
| [4] |
WANG L, PENG Q, SAI B, et al. Ligand-independent EphB1 signaling mediates TGF-β-activated CDH2 and promotes lung cancer cell invasion and migration[J]. J Cancer, 2020, 11(14): 4123-4131. DOI: 10.7150/jca.44576.
|
| [5] |
WANG Y, YU H, SHAN Y, et al. EphA1 activation promotes the homing of endothelial progenitor cells to hepatocellular carcinoma for tumor neovascularization through the SDF-1/CXCR4 signaling pathway[J]. J Exp Clin Cancer Res, 2016, 35: 65. DOI: 10.1186/s13046-016-0339-6.
|
| [6] |
ANDERTON M, van der MEULEN E, BLUMENTHAL MJ, et al. The role of the Eph receptor family in tumorigenesis[J]. Cancers (Basel), 2021, 13(2): 206. DOI: 10.3390/cancers13020206.
|
| [7] |
ZHANG XH, MA N, LYU CY, et al. Role of receptor tyrosine kinase Eph in hepatocellular carcinoma[J]. Chin J Surg Oncol, 2019, 11(2): 136. DOI: 10.3969/j.issn.1674-4136.2019.02.014.张晓华, 马宁, 吕成余, 等. 受体酪氨酸激酶Eph受体在肝细胞癌中作用的研究进展[J]. 中国肿瘤外科杂志, 2019, 11(2): 136. DOI: 10.3969/j.issn.1674-4136.2019.02.014.
|
| [8] |
HIRAI H, MARU Y, HAGIWARA K, et al. A novel putative tyrosine kinase receptor encoded by the eph gene[J]. Science, 1987, 238(4834): 1717-1720. DOI: 10.1126/science.2825356.
|
| [9] |
LIANG LY, PATEL O, JANES PW, et al. Eph receptor signalling: From catalytic to non-catalytic functions[J]. Oncogene, 2019, 38(39): 6567-6584. DOI: 10.1038/s41388-019-0931-2.
|
| [10] |
Unified nomenclature for Eph family receptors and their ligands, the ephrins. Eph Nomenclature Committee[J]. Cell, 1997, 90(3): 403-404. DOI: 10.1016/s0092-8674(00)80500-0.
|
| [11] |
PASQUALE EB. Eph receptor signalling casts a wide net on cell behaviour[J]. Nat Rev Mol Cell Biol, 2005, 6(6): 462-475. DOI: 10.1038/nrm1662.
|
| [12] |
PASQUALE EB. Eph receptors and ephrins in cancer: Bidirectional signalling and beyond[J]. Nat Rev Cancer, 2010, 10(3): 165-180. DOI: 10.1038/nrc2806.
|
| [13] |
BATLLE E, WILKINSON DG. Molecular mechanisms of cell segregation and boundary formation in development and tumorigenesis[J]. Cold Spring Harb Perspect Biol, 2012, 4(1): a008227. DOI: 10.1101/cshperspect.a008227.
|
| [14] |
GONG J, KÖRNER R, GAITANOS L, et al. Exosomes mediate cell contact-independent ephrin-Eph signaling during axon guidance[J]. J Cell Biol, 2016, 214(1): 35-44. DOI: 10.1083/jcb.201601085.
|
| [15] |
FALIVELLI G, LISABETH EM, RUBIO de la TORRE E, et al. Attenuation of eph receptor kinase activation in cancer cells by coexpressed ephrin ligands[J]. PLoS One, 2013, 8(11): e81445. DOI: 10.1371/journal.pone.0081445.
|
| [16] |
GOPAL U, BOHONOWYCH JE, LEMA-TOME C, et al. A novel extracellular Hsp90 mediated co-receptor function for LRP1 regulates EphA2 dependent glioblastoma cell invasion[J]. PLoS One, 2011, 6(3): e17649. DOI: 10.1371/journal.pone.0017649.
|
| [17] |
LARSEN AB, STOCKHAUSEN MT, POULSEN HS. Cell adhesion and EGFR activation regulate EphA2 expression in cancer[J]. Cell Signal, 2010, 22(4): 636-644. DOI: 10.1016/j.cellsig.2009.11.018.
|
| [18] |
WU B, ROCKEL JS, LAGARES D, et al. Ephrins and eph receptor signaling in tissue repair and fibrosis[J]. Curr Rheumatol Rep, 2019, 21(6): 23. DOI: 10.1007/s11926-019-0825-x.
|
| [19] |
IEGUCHI K, MARU Y. Roles of EphA1/A2 and ephrin-A1 in cancer[J]. Cancer Sci, 2019, 110(3): 841-848. DOI: 10.1111/cas.13942.
|
| [20] |
DUSABLON A, PARKS J, WHITEHURST K, et al. EphrinA1-Fc attenuates myocardial ischemia/reperfusion injury in mice[J]. PLoS One, 2017, 12(12): e0189307. DOI: 10.1371/journal.pone.0189307.
|
| [21] |
WIJERATNE D, RODGER J, STEVENSON A, et al. Ephrin-A2 affects wound healing and scarring in a murine model of excisional injury[J]. Burns, 2019, 45(3): 682-690. DOI: 10.1016/j.burns.2018.10.002.
|
| [22] |
LI Y, YAN H, WANG F, et al. Activation of EphA1-Epha receptor axis attenuates diabetic nephropathy in mice[J]. Biochem Biophys Res Commun, 2017, 486(3): 693-699. DOI: 10.1016/j.bbrc.2017.03.100.
|
| [23] |
LAGARES D, GHASSEMI-KAKROODI P, TREMBLAY C, et al. ADAM10-mediated ephrin-B2 shedding promotes myofibroblast activation and organ fibrosis[J]. Nat Med, 2017, 23(12): 1405-1415. DOI: 10.1038/nm.4419.
|
| [24] |
MIMCHE PN, BRADY LM, BRAY CF, et al. The receptor tyrosine kinase EphB2 promotes hepatic fibrosis in mice[J]. Hepatology, 2015, 62(3): 900-914. DOI: 10.1002/hep.27792.
|
| [25] |
MIMCHE PN, LEE CM, MIMCHE SM, et al. EphB2 receptor tyrosine kinase promotes hepatic fibrogenesis in mice via activation of hepatic stellate cells[J]. Sci Rep, 2018, 8(1): 2532. DOI: 10.1038/s41598-018-20926-9.
|
| [26] |
CHEN X, ZHANG D, WANG Y, et al. Synergistic antifibrotic effects of miR-451 with miR-185 partly by co-targeting EphB2 on hepatic stellate cells[J]. Cell Death Dis, 2020, 11(5): 402. DOI: 10.1038/s41419-020-2613-y.
|
| [27] |
IIDA H, HONDA M, KAWAI HF, et al. Ephrin-A1 expression contributes to the malignant characteristics of {alpha}-fetoprotein producing hepatocellular carcinoma[J]. Gut, 2005, 54(6): 843-851. DOI: 10.1136/gut.2004.049486.
|
| [28] |
FAN M, LIU Y, XIA F, et al. Increased expression of EphA2 and E-N cadherin switch in primary hepatocellular carcinoma[J]. Tumori, 2013, 99(6): 689-696. DOI: 10.1700/1390.15457.
|
| [29] |
YIN JP, YUE ZC, ZHUO SY. STAT3: A key molecule in the progression of liver cancer mediated by chronic inflammation[J]. J Clin Hepatol, 2020, 36(4): 948-952. DOI: 10.3969/j.issn.1001-5256.2020.04.054.音金萍, 岳紫晨, 卓少元. STAT3: 慢性炎症介导肝癌进程的关键分子[J]. 临床肝胆病杂志, 2020, 36(4): 948-952. DOI: 10.3969/j.issn.1001-5256.2020.04.054.
|
| [30] |
WANG H, HOU W, PERERA A, et al. Targeting EphA2 suppresses hepatocellular carcinoma initiation and progression by dual inhibition of JAK1/STAT3 and AKT signaling[J]. Cell Rep, 2021, 34(8): 108765. DOI: 10.1016/j.celrep.2021.108765.
|
| [31] |
LV XY, WANG J, HUANG F, et al. EphA3 contributes to tumor growth and angiogenesis in human gastric cancer cells[J]. Oncol Rep, 2018, 40(4): 2408-2416. DOI: 10.3892/or.2018.6586.
|
| [32] |
WANG W, JIA WD, HU B, et al. RAB10 overexpression promotes tumor growth and indicates poor prognosis of hepatocellular carcinoma[J]. Oncotarget, 2017, 8(16): 26434-26447. DOI: 10.18632/oncotarget.15507.
|
| [33] |
LU CY, YANG ZX, ZHOU L, et al. High levels of EphA3 expression are associated with high invasive capacity and poor overall survival in hepatocellular carcinoma[J]. Oncol Rep, 2013, 30(5): 2179-2186. DOI: 10.3892/or.2013.2679.
|
| [34] |
YAN Y, LUO YC, WAN HY, et al. MicroRNA-10a is involved in the metastatic process by regulating Eph tyrosine kinase receptor A4-mediated epithelial-mesenchymal transition and adhesion in hepatoma cells[J]. Hepatology, 2013, 57(2): 667-677. DOI: 10.1002/hep.26071.
|
| [35] |
CHEN G, WANG Y, ZHOU M, et al. EphA1 receptor silencing by small interfering RNA has antiangiogenic and antitumor efficacy in hepatocellular carcinoma[J]. Oncol Rep, 2010, 23(2): 563-570.
|
| [36] |
MIAO H, LI DQ, MUKHERJEE A, et al. EphA2 mediates ligand-dependent inhibition and ligand-independent promotion of cell migration and invasion via a reciprocal regulatory loop with Akt[J]. Cancer Cell, 2009, 16(1): 9-20. DOI: 10.1016/j.ccr.2009.04.009.
|
| [37] |
ZANTEK ND, AZIMI M, FEDOR-CHAIKEN M, et al. E-cadherin regulates the function of the EphA2 receptor tyrosine kinase[J]. Cell Growth Differ, 1999, 10(9): 629-638.
|
| [38] |
LI N, SHI K, LI W. TUSC7: A novel tumor suppressor long non-coding RNA in human cancers[J]. J Cell Physiol, 2018, 233(9): 6401-6407. DOI: 10.1002/jcp.26544.
|



 PDF下载 ( 2646 KB)
PDF下载 ( 2646 KB)


 下载:
下载: