Pathogenesis of drug-induced liver injury: Current understanding and future needs
-
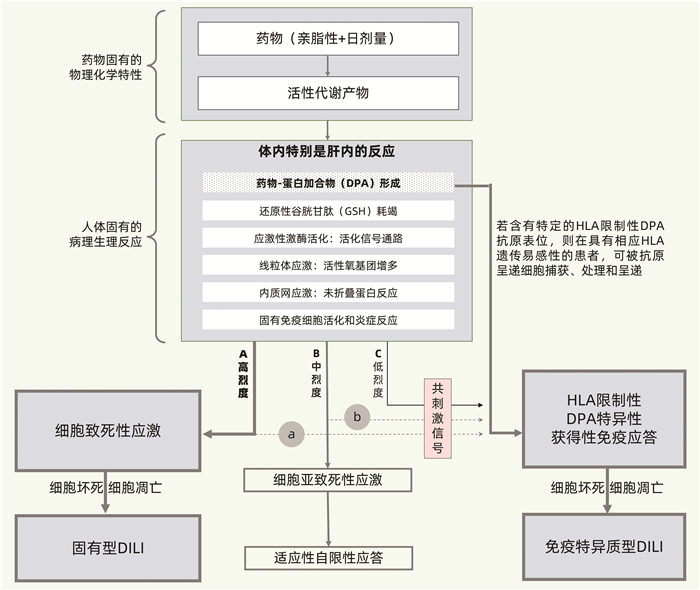
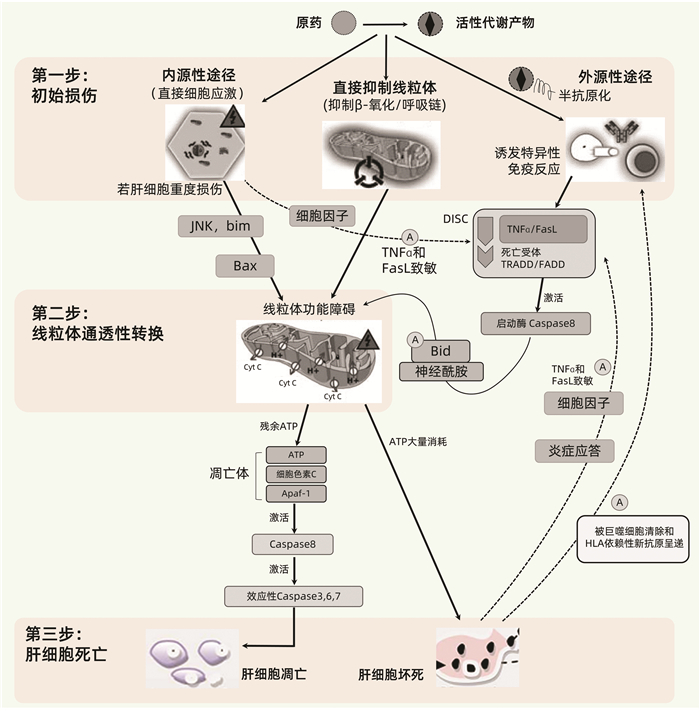
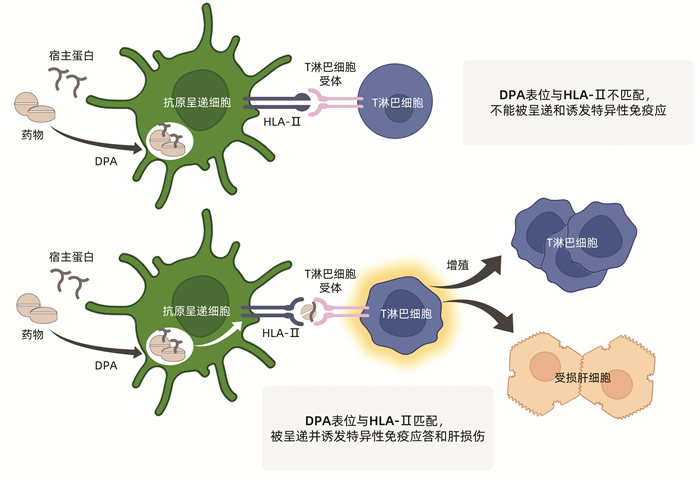
摘要: 药物性肝损伤(DILI)的危险因素涉及人体因素(包括一般性非遗传因素和特异质性遗传及免疫因素)、药物因素和环境因素。其发生机制可分为固有(直接)肝毒性、特异质肝毒性和间接肝毒性,此外还有某些药物对肝脏的致瘤与致癌性。不同类型肝毒性的发生机制既有明显的区别,又有多环节的内在关联。以线粒体通透性转换(MPT)为中心的三步机制进程模型和以肝细胞再生及肝组织修复能力为转归节点的两阶段机制进程模型,从不同视角展示了DILI的机制进程。阐明DILI复杂的发生机制,需要长期的病例积累和系统研究,这对于其科学预防、诊断和治疗具有十分重要的意义。
-
关键词:
- 化学性与药物性肝损伤 /
- 危险因素 /
- 发病机制
Abstract: The risk factors for drug-induced liver injury (DILI) involve host factors (including general non-genetic factors and idiosyncratic genetic and immune factors), drug-related factors, and environmental factors. The pathogenesis of DILI can be classified as intrinsic (or direct) hepatotoxicity, idiosyncratic hepatotoxicity, and indirect hepatotoxicity, as well as tumorigenicity and carcinogenicity of some drugs to the liver. The pathogenesis of different types of hepatotoxicity not only has significant differences, but also has internal correlation at multiple links. The three-step model centered on mitochondrial permeability transition (MPT) and a two-stage model with liver cell regeneration and liver tissue repair capacity as the determinants of different outcomes display the mechanism progress of DILI from different perspectives. Clarification of the complex pathogenesis of DILI needs long-term collection of clinical cases and systematic studies, which is of great significance for the scientific prevention, diagnosis, and treatment of DILI.-
Key words:
- Chemical and Drug-Induced Liver Injury /
- Risk Factors /
- Pathogenesis
-
表 1 HLA遗传多态性与特定药物所致DILI的相关性
HLA基因单倍型 相关药物(患者阳性率) 相关性或对照组阳性率 A*02∶01 阿莫西林-克拉维酸 相关 A*31∶01 卡巴咪嗪(17%) 正常对照组阳性率2% A*33∶01 噻氯匹定(80%),甲基多巴(50%),依那普利(50%),
非诺贝特(43%),特比萘芬(43%),舍曲林(40%),
红霉素(20%)正常对照组阳性率1% A*33∶03 噻氯匹定 相关 B*18∶01 阿莫西林-克拉维酸 相关 B*35∶01 何首乌(41.1%) 其他原因DILI阳性率11.9% 汉族MHC数据库阳性率2.7% B*35∶02 米诺环素(16%) 正常对照组阳性率0.6% B*57∶01 氟氯西林(84%~87%) 正常对照组阳性率6% DQA1*01∶02 卢米考昔 相关 DRB1*01∶01 奈韦拉平 相关 DRB1*15∶011)/DRB5*
01∶01/DQB1*06∶02阿莫西林-克拉维酸(57%~67%)
罗美昔布
氟烷正常对照组阳性率15%~20%
强相关
强相关DRB1*07∶012)/DQB1*
02∶02/DQA1*02∶01拉帕替尼
希美加群相关
相关DRB1*16∶01/DQB1* 05∶02 氟吡汀(25%) 正常对照组阳性率1% 注:1)与药物性胆汁淤积的发生强相关; 2)可使氟氯西林诱导DILI的风险增加81~100倍以上。 表 2 三种药物肝毒性类型的比较1)
项目 固有(直接)肝毒性 特异质肝毒性 间接肝毒性 发生机制 药物及其活性代谢产物的固有(直接)肝毒性,以及人体固有的病理生理性损伤反应 与人体遗传多态性相关的特异质性药物代谢障碍,或药物-宿主蛋白加合物特异性、HLA限制性获得性免疫应答 继发于药物或其活性代谢产物的生物学活性,多通过影响免疫系统而间接对肝脏产生毒性作用 发生率 对具体药物而言,常见 对具体单药而言,少见。对药物整体而言,常见 随具体药物生物学活性特点的不同,可从少见到较为常见 剂量相关 明显的剂量依赖性 不明显(但阈剂量通常也需达到50~100 mg/d) 有一定程度的剂量和疗程相关性 可预测性 多可预测 难以预测 部分可预测 可复制性 常可在动物模型复制 难以在动物模型复制 有可能在动物模型复制 潜伏期 通常很快(数日内) 相差较大:数日至数月乃至1年以上 一般较迟出现(数月) 临床表型 肝酶升高,急性肝坏死,肝窦阻塞综合征,急性脂肪肝,结节性再生,等 急性肝细胞损伤,淤胆性肝炎,混合性肝炎,单纯性胆汁淤积,慢性肝炎,等 急性肝炎,免疫介导的肝炎,脂肪肝,慢性肝炎,等 常见涉及药物 APAP、阿司匹林、甲氨蝶呤等抗肿瘤化疗药物、高效抗逆转录病毒药物、合成代谢类甾族激素、他汀类药物、环孢霉素、肝素、丙戊酸、烟酸、丁酸、可卡因、胺碘酮、他克林、消胆胺、含有吡咯双烷生物碱的草药,等 阿莫西林-克拉维酸、氟氯西林、头孢菌素类、大环内酯类、曲伐沙星等喹诺酮类、磺胺类药物、异烟肼、吡嗪酰胺、酮康唑、特比萘芬,呋喃妥因、米诺环素,別嘌呤醇、丙基硫氧嘧啶,双氯芬酸、来氟米特、噻氯匹定、拉帕替尼、帕唑帕尼、氟他米特、他汀类药物、非诺贝特、丹曲林、胺碘酮、氟烷、托伐普坦、赖诺普利、甲基多巴、波生坦、托卡朋、苯妥英、戒酒硫、菲尔安酯,等 免疫检查点抑制剂、抗肿瘤坏死因子单抗、抗CD20单抗、蛋白激酶抑制剂、糖皮质激素、某些抗肿瘤药物、影响和干扰物质及能量代谢的相关药物,等 注:1)根据参考文献[18-19]进行必要修改。 -
[1] YU YC, MAO YM, CHEN CW, et al. CSH guidelines for the diagnosis and treatment of drug-induced liver injury[J]. Hepatol Int, 2017, 11(3): 221-241. DOI: 10.1007/s12072-017-9793-2. [2] European Association for the Study of the Liver. EASL clinical practice guidelines: Drug-induced liver injury[J]. J Hepatol, 2019, 70(6): 1222-1261. DOI: 10.1016/j.jhep.2019.02.014. [3] CHALASANI NP, MADDUR H, RUSSO MW, et al. ACG Clinical Guideline: Diagnosis and management of idiosyncratic drug-induced liver injury[J]. Am J Gastroenterol, 2021, 116(5): 878-898. DOI: 10.14309/ajg.0000000000001259. [4] DEVARBHAVI H, AITHAL G, TREEPRASERTSUK S, et al. Drug-induced liver injury: Asia Pacific Association of study of liver consensus guidelines[J]. Hepatol Int, 2021, 15(2): 258-282. DOI: 10.1007/s12072-021-10144-3. [5] BJÖRNSSON ES, BERGMANN OM, BJÖRNSSON HK, et al. Incidence, presentation, and outcomes in patients with drug-induced liver injury in the general population of Iceland[J]. Gastroenterology, 2013, 144(7): 1419-1425, e1-e3; quiz e19-e20. DOI: 10.1053/j.gastro.2013.02.006. [6] TREEM WR, PALMER M, LONJON-DOMANEC I, et al. Consensus guidelines: Best practices for detection, assessment and management of suspected acute drug-induced liver injury during clinical trials in adults with chronic viral hepatitis and adults with cirrhosis secondary to hepatitis B, C and nonalcoholic steatohepatitis[J]. Drug Saf, 2021, 44(2): 133-165. DOI: 10.1007/s40264-020-01014-2. [7] LIU M, YANG XZ, YU YC. Current status of the pathogenesis, diagnosis and treatment of drug-induced cholestasis[J]. J Clin Hepatol, 2019, 35(2): 115-120. DOI: 10.3969/j.issn.1001-5256.2019.02.003.刘梦, 杨玄子, 于乐成. 药物性胆汁淤积的发病机制及诊疗现状[J]. 临床肝胆病杂志, 2019, 35(2): 252-257. DOI: 10.3969/j.issn.1001-5256.2019.02.003. [8] CHEN K, GUO R, WEI C. Synonymous mutation rs2515641 affects CYP2E1 mRNA and protein expression and susceptibility to drug-induced liver injury[J]. Pharmacogenomics, 2020, 21(7): 459-470. DOI: 10.2217/pgs-2019-0151. [9] CHEN R, WANG J, TANG SW, et al. CYP7A1, BAAT and UGT1A1 polymorphisms and susceptibility to anti-tuberculosis drug-induced hepatotoxicity[J]. Int J Tuberc Lung Dis, 2016, 20(6): 812-818. DOI: 10.5588/ijtld.15.0450. [10] ZHANG M, WANG S, WILFFERT B, et al. The association between the NAT2 genetic polymorphisms and risk of DILI during anti-TB treatment: A systematic review and meta-analysis[J]. Br J Clin Pharmacol, 2018, 84(12): 2747-2760. DOI: 10.1111/bcp.13722. [11] CHANHOM N, UDOMSINPRASERT W, CHAIKLEDKAEW U, et al. GSTM1 and GSTT1 genetic polymorphisms and their association with antituberculosis drug-induced liver injury[J]. Biomed Rep, 2020, 12(4): 153-162. DOI: 10.3892/br.2020.1275. [12] DALY AK, DAY CP. Genetic factors in the pathogenesis of drug-induced liver injury[M]// KAPLOWITZ N, DELEVE LD. Drug-induced liver disease, 3rd ed. San Diego: Academic Press, 2013: 215-225. [13] WATKINS PB, SELIGMAN PJ, PEARS JS, et al. Using controlled clinical trials to learn more about acute drug-induced liver injury[J]. Hepatology, 2008, 48(5): 1680-1689. DOI: 10.1002/hep.22633. [14] YANG WN, PANG LL, ZHOU JY, et al. Single-nucleotide polymorphisms of HLA and polygonum multiflorum-induced liver injury in the Han Chinese population[J]. World J Gastroenterol, 2020, 26(12): 1329-1339. DOI: 10.3748/wjg.v26.i12.1329. [15] YU YC, HE CL. Outcome of liver injury and liver tissue repair[J]. Chin Hepatol, 2010, 15(6): 460-464. DOI: 10.3969/j.issn.1008-1704.2010.06.023.于乐成, 何长伦. 肝损伤转归与肝组织修复[J]. 肝脏, 2010, 15(6): 460-464. DOI: 10.3969/j.issn.1008-1704.2010.06.023. [16] HUI CL, LEE ZJ. Hepatic disorders associated with exogenous sex steroids: MR imaging findings[J]. Abdom Radiol (NY), 2019, 44(7): 2436-2447. DOI: 10.1007/s00261-019-01941-4. [17] LU ZN, LUO Q, ZHAO LN, et al. The mutational features of aristolochic acid-induced mouse and human liver cancers[J]. Hepatology, 2020, 71(3): 929-942. DOI: 10.1002/hep.30863. [18] HOOFNAGLE JH, BJÖRNSSON ES. Drug-induced liver injury-types and phenotypes[J]. N Engl J Med, 2019, 381(3): 264-273. DOI: 10.1056/NEJMra1816149. [19] ZHUGE Y, LIU Y, XIE W, et al. Expert consensus on the clinical management of pyrrolizidine alkaloid-induced hepatic sinusoidal obstruction syndrome[J]. J Gastroenterol Hepatol, 2019, 34(4): 634-642. DOI: 10.1111/jgh.14612. [20] CHEN M, BORLAK J, TONG W. A Model to predict severity of drug-induced liver injury in humans[J]. Hepatology, 2016, 64(3): 931-940. DOI: 10.1002/hep.28678. [21] MEHENDALE HM. Tissue repair: An important determinant of final outcome of toxicant-induced injury[J]. Toxicol Pathol, 2005, 33(1): 41-51. DOI: 10.1080/01926230590881808. [22] ANAND SS, MURTHY SN, VAIDYA VS, et al. Tissue repair plays pivotal role in final outcome of liver injury following chloroform and allyl alcohol binary mixture[J]. Food Chem Toxicol, 2003, 41(8): 1123-1132. DOI: 10.1016/s0278-6915(03)00066-8. [23] ANAND SS, SONI MG, VAIDYA VS, et al. Extent and timeliness of tissue repair determines the dose-related hepatotoxicity of chloroform[J]. Int J Toxicol, 2003, 22(1): 25-33. DOI: 10.1080/10915810305074. [24] FONTANA RJ. Pathogenesis of idiosyncratic drug-induced liver injury and clinical perspectives[J]. Gastroenterology, 2014, 146(4): 914-928. DOI: 10.1053/j.gastro.2013.12.032. [25] HOU JX, YAN FQ, YU YC. The pathogenesis, diagnosis and treatment of drug-induced liver injury with extrahepatic adverse drug reactions[J]. J Clin Hepatol, 2020, 36(3): 497-500. DOI: 10.3969/j.issn.1001-5256.2020.03.003.侯俊兴, 严粉琴, 于乐成. 药物性肝损伤合并肝外药物不良反应的发生机制及诊疗问题[J]. 临床肝胆病杂志, 2020, 36(3): 497-500. DOI: 10.3969/j.issn.1001-5256.2020.03.003. [26] WANG DY, SALEM JE, COHEN JV, et al. Fatal toxic effects associated with immune checkpoint inhibitors: A systematic review and meta-analysis[J]. JAMA Oncol, 2018, 4(12): 1721-1728. DOI: 10.1001/jamaoncol.2018.3923. [27] POSTOW MA, SIDLOW R, HELLMANN MD. Immune-related adverse events associated with immune checkpoint blockade[J]. N Engl J Med, 2018, 378(2): 158-168. DOI: 10.1056/NEJMra1703481. [28] VADDEPALLY RK, KHAREL P, PANDEY R, et al. Review of indications of FDA-approved immune checkpoint inhibitors per NCCN guidelines with the level of evidence[J]. Cancers (Basel), 2020, 12(3): 738. DOI: 10.3390/cancers12030738. [29] PEERAPHATDIT TB, WANG J, ODENWALD MA, et al. Hepatotoxicity from immune checkpoint inhibitors: A systematic review and management recommendation[J]. Hepatology, 2020, 72(1): 315-329. DOI: 10.1002/hep.31227. [30] KOK B, LESTER E, LEE WM, et al. Acute liver failure from tumor necrosis factor-α antagonists: Report of four cases and literature review[J]. Dig Dis Sci, 2018, 63(6): 1654-1666. DOI: 10.1007/s10620-018-5023-6. [31] LOPETUSO LR, MOCCI G, MARZO M, et al. Harmful effects and potential benefits of anti-tumor necrosis factor (TNF)-α on the liver[J]. Int J Mol Sci, 2018, 19(8): 2199. DOI: 10.3390/ijms19082199. [32] ZOUBEK ME, PINAZO-BANDERA J, ORTEGA-ALONSO A, et al. Liver injury after methylprednisolone pulses: A disputable cause of hepatotoxicity. A case series and literature review[J]. United European Gastroenterol J, 2019, 7(6): 825-837. DOI: 10.1177/2050640619840147. [33] LOOMBA R, LIANG TJ. Hepatitis B reactivation associated with immune suppressive and biological modifier therapies: Current concepts, management strategies, and future directions[J]. Gastroenterology, 2017, 152(6): 1297-1309. DOI: 10.1053/j.gastro.2017.02.009. [34] KAPILA N, AL-KHALLOUFI K, BEJARANO PA, et al. Fibrosing cholestatic hepatitis after kidney transplantation from HCV-viremic donors to HCV-negative recipients: A unique complication in the DAA era[J]. Am J Transplant, 2020, 20(2): 600-605. DOI: 10.1111/ajt.15583. [35] YU YC, HE CL, WANG YM. Recent progress of fibrosing cholestatic hepatitis or immunosuppression-induced liver failure[J]. Infect Dis Info, 2011, 24(3): 185-188. DOI: 10.3969/j.issn.1007-8134.2011.03.018.于乐成, 何长伦, 王宇明. 纤维淤胆性肝炎/免疫抑制诱导性肝衰竭新进展[J]. 传染病信息, 2011, 24(3): 185-188. DOI: 10.3969/j.issn.1007-8134.2011.03.018. [36] RUSSMANN S, KULLAK-UBLICK GA, GRATTAGLIANO I. Current concepts of mechanisms in drug-induced hepatotoxicity[J]. Curr Med Chem, 2009, 16(23): 3041-3053. DOI: 10.2174/092986709788803097. [37] KRÄHENBUHL S, BRAUCHLI Y, KUMMER O, et al. Acute liver failure in two patients with regular alcohol consumption ingesting paracetamol at therapeutic dosage[J]. Digestion, 2007, 75(4): 232-237. DOI: 10.1159/000111032. [38] GALLUZZI L, VITALE I, AARONSON SA, et al. Molecular mechanisms of cell death: Recommendations of the nomenclature committee on cell death 2018[J]. Cell Death Differ, 2018, 25(3): 486-541. DOI: 10.1038/s41418-017-0012-4. [39] TANG D, KANG R, BERGHE TV, et al. The molecular machinery of regulated cell death[J]. Cell Res, 2019, 29(5): 347-364. DOI: 10.1038/s41422-019-0164-5. [40] COPPLE IM, GOLDRING CE, JENKINS RE, et al. The hepatotoxic metabolite of acetaminophen directly activates the Keap1-Nrf2 cell defense system[J]. Hepatology, 2008, 48(4): 1292-1301. DOI: 10.1002/hep.22472. [41] KALGUTKAR AS, GARDNER I, OBACH RS, et al. A comprehensive listing of bioactivation pathways of organic functional groups[J]. Curr Drug Metab, 2005, 6(3): 161-225. DOI: 10.2174/1389200054021799. [42] SÉGUIN B, UETRECHT J. The danger hypothesis applied to idiosyncratic drug reactions[J]. Curr Opin Allergy Clin Immunol, 2003, 3(4): 235-242. DOI: 10.1097/00130832-200308000-00001. [43] WANG X, TOMSO DJ, CHORLEY BN, et al. Identification of polymorphic antioxidant response elements in the human genome[J]. Hum Mol Genet, 2007, 16(10): 1188-1200. DOI: 10.1093/hmg/ddm066. [44] GRATTAGLIANO I, LAUTERBURG BH, PORTINCASA P, et al. Mitochondrial glutathione content determines the rate of liver regeneration after partial hepatectomy in eu- and hypothyroid rats[J]. J Hepatol, 2003, 39(4): 571-579. DOI: 10.1016/s0168-8278(03)00317-9. [45] FAUSTO N, CAMPBELL JS, RIEHLE KJ. Liver regeneration[J]. J Hepatol, 2012, 57(3): 692-694. DOI: 10.1016/j.jhep.2012.04.016. [46] MALATO Y, NAQVI S, SCHVRMANN N, et al. Fate tracing of mature hepatocytes in mouse liver homeostasis and regeneration[J]. J Clin Invest, 2011, 121(12): 4850-4860. DOI: 10.1172/JCI59261. [47] LIU L, YANNAM GR, NISHIKAWA T, et al. The microenvironment in hepatocyte regeneration and function in rats with advanced cirrhosis[J]. Hepatology, 2012, 55(5): 1529-1539. DOI: 10.1002/hep.24815. [48] YU YC, HE CL, HOU JL. Biomarkers in drug-induced liver injuries[J]. J Prac Hepatol, 2014, 17(6): 564-568. DOI: 10.3969/j.issn.1672-5069.2014.06.03.于乐成, 何长伦, 侯金林. 药物诱导性肝损伤生物标志物研究进展[J]. 实用肝脏病杂志, 2014, 17(6): 564-568. DOI: 10.3969/j.issn.1672-5069.2014.06.03. -



 PDF下载 ( 4898 KB)
PDF下载 ( 4898 KB)

 下载:
下载: